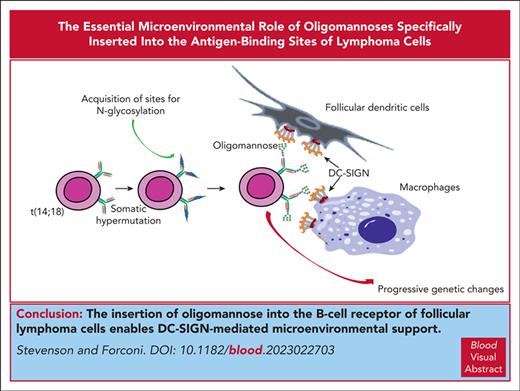
Visual Abstract
There are 2 mandatory features added sequentially en route to classical follicular lymphoma (FL): first, the t(14;18) translocation, which upregulates BCL2, and second, the introduction of sequence motifs into the antigen-binding sites of the B-cell receptor (BCR), to which oligomannose-type glycan is added. Further processing of the glycan is blocked by complementarity-determining region–specific steric hindrance, leading to exposure of mannosylated immunoglobulin (Ig) to the microenvironment. This allows for interaction with the local lectin, dendritic cell–specific ICAM-3–grabbing nonintegrin (DC-SIGN), expressed by tissue macrophages and follicular dendritic cells. The major function of DC-SIGN is to engage pathogens, but this is subverted by FL cells. DC-SIGN induces tumor-specific low-level BCR signaling in FL cells and promotes membrane changes with increased adhesion to VCAM-1 via proximal kinases and actin regulators but, in contrast to engagement by anti-Ig, avoids endocytosis and apoptosis. These interactions appear mandatory for the early development of FL, before the acquisition of other accelerating mutations. BCR-associated mannosylation can be found in a subset of germinal center B-cell–like diffuse large B-cell lymphoma with t(14;18), tracking these cases back to FL. This category was associated with more aggressive behavior: both FL and transformed cases and, potentially, a significant number of cases of Burkitt lymphoma, which also has sites for N-glycan addition, could benefit from antibody-mediated blockade of the interaction with DC-SIGN.
Introduction
The establishment of classic follicular lymphoma (FL) involves multiple B-cell opportunistic changes, beginning in the bone marrow, where t(14;18)(q32;q21) translocation upregulates antiapoptotic BCL2. The resulting progenitor cells can undergo the normal processes of antigen engagement and differentiation in the germinal center (GC) and may remain harmless. However, other events can occur that allow the cells to survive and proliferate in the hostile GC, where normal B cells remain only transiently. The strategy here is to harness and modify interactions with the microenvironment. Because the B-cell receptor (BCR) is a key sensor of the microenvironment, it is not surprising that this offers a major opportunity for subversion. The BCR-based subversion varies with the nature, behavior, and location of B-cell tumors, and in some cases, such as chronic lymphocytic leukemia, it clearly involves responding to autoantigens.1 FL, a GC-derived tumor, has evolved a different strategy.
The structural peculiarity of the BCR of classical FL cells has been well documented, and its mandatory nature reveals a feature required for lymphomagenesis.2 It arises as a simple modification of the surface immunoglobulin variable region that occurs during somatic hypermutation, introducing sequence motifs (Asn-Xaa-Ser/Thr, in which Xaa can be almost any amino acid except Pro) that are then available for the addition of glycan. In this way, FL cells capture a normal mutational mechanism, followed by positively selection of cells with these motifs. The same feature is also seen in some other GC tumors but rarely in normal GC B cells or other B-cell tumors, such as chronic lymphocytic leukemia or myeloma.3 Even more strange is that the introduced glycan terminates prematurely at the high-mannose stage, with a range of inserted mannoses from M5to M94 (Figure 1A). The halt on further processing is in stark contrast to the immunoglobulin (Ig)-constant region N-glycosylation sites in the same Ig molecule, which are fully processed to complex sugars.5 This leads to the odd situation in FL of expressing “naked” high mannoses in the antigen-binding site of the surface Ig receptor, a feature not generally found in cell-surface glycoproteins. The necessity of this peculiar modification of the BCR for FL is underpinned by the fact that surface Ig expression is retained in this lymphoma, even though 1 allele is lost because of the t(14;18) translocation.
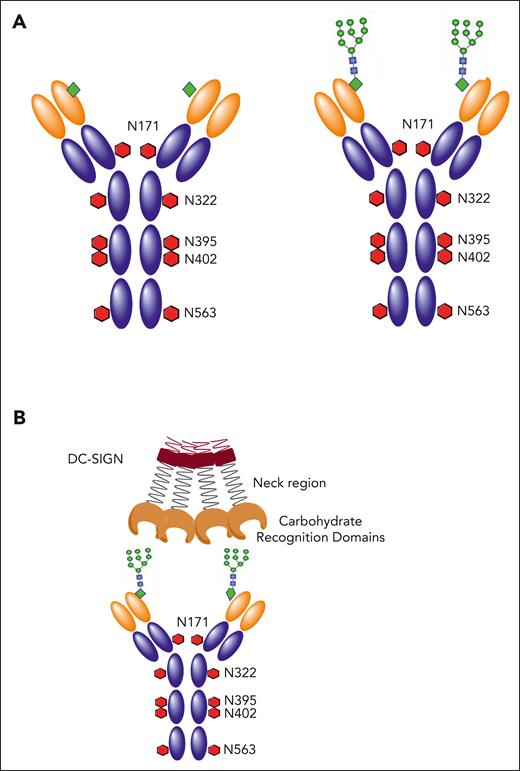
Natural and introduced N-glycosylation sites in human IgM. (A) Domain structure of healthy human IgM with natural GL sites for N-glycosylation (N-X-S/T) indicated as red hexagons at the amino acid positions in the constant region. Introduced sites in the variable region of cases of FL are shown as green diamonds. Available oligomannoses are specifically found in the variable region as branched mannoe linked to N-acetyl glucosamines (green circles). (B) DC-SIGN is a tetramer stabilized by the neck region. It interacts with oligomannoses introduced into the antigen-binding site of FL cells via its CRDs.
Natural and introduced N-glycosylation sites in human IgM. (A) Domain structure of healthy human IgM with natural GL sites for N-glycosylation (N-X-S/T) indicated as red hexagons at the amino acid positions in the constant region. Introduced sites in the variable region of cases of FL are shown as green diamonds. Available oligomannoses are specifically found in the variable region as branched mannoe linked to N-acetyl glucosamines (green circles). (B) DC-SIGN is a tetramer stabilized by the neck region. It interacts with oligomannoses introduced into the antigen-binding site of FL cells via its CRDs.
Knowing that the motifs were introduced during somatic hypermutation indicates that they are present at very early stages of lymphoma development, possibly even in the precursor cells that are detectable in healthy individuals.6 Potentially the presence of motifs could reveal the committed precursor cell,7 and motifs have been found in 2 of 2 cases of FL in situ.8,9 Among the vast interpatient heterogeneity of FL at the genetic/epigenetic level,7 the consistent presence of N-glycosylation sites in the BCR stands out as a common feature. The question then arises as to what is the purpose of the exposed mannoses and because sugars generally interact with lectins, which microenvironmental candidate can interact with mannosylated surface Ig (sIg-Mann)?
Dendritic cell–specific ICAM-3–grabbing nonintegrin (DC-SIGN; CD209) was an obvious candidate, because it is present in FL tissue10 and binds to mannoses.11 The nomenclature of DC-SIGN is misleading because it is not dendritic cell-specific and binds to other molecules besides ICAM-3. In fact, tetrameric DC-SIGN binds specifically to the Ig-Mann expressed on FL cell surfaces via its carbohydrate-recognition domains (CRDs; Figure 1B) and does not bind to normal B cells or other blood cells.12,13
This review makes the case for the significance of the sine qua non interaction between FL cells and DC-SIGN for lymphoma development/maintenance. It addresses the expression of DC-SIGN in vivo and the consequences of this interaction in vitro and suggests a therapeutic approach by antibody blockade, potentially at an early stage of the disease. For diffuse large B-cell lymphoma (DLBCL), the presence of acquired N-glycosylation sites (AGSs) in Ig complementarity-determining regions (CDRs) appears to reveal a subset derived from FL and could add prognostic value.
Localization of DC-SIGN in FL tissue
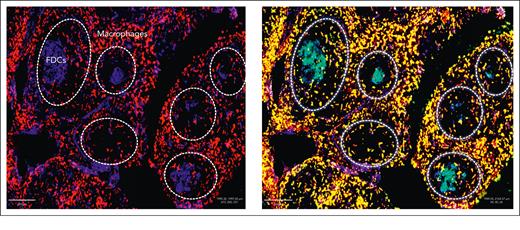
The expression of interleukin-4 in FL tissue14 sets the scene for the polarization of macrophages to the M2 type and explains the expression of DC-SIGN by these cells in FL, located mainly in the interfollicular zone.10,15 More recently, our collaborators have also detected DC-SIGN on follicular dendritic cells (FDCs) in FL (Figure 2)15 (R. Burack and Y. Zhang, unpublished data). Double staining of macrophages (red CD163) plus DC-SIGN (green CD209) gives a yellow color, and that of FDCs (blue CD23) plus DC-SIGN (green CD209) gives cyan Figure 2), showing that in this illustrative case, both macrophages and FDCs express DC-SIGN. The role of FDCs in FL is unclear, although it has been suggested that they may be critical for the early stages of lymphoma development, when the network that they form in healthy GCs is intact.16,17 In a normal immune response, FDCs bind to immune complexes via the Fc or complement receptors.18,19 These are taken up by cycling endosomes, which are then presented to B cells.20 Perhaps, more importantly, for FL, FDCs express a range of apoptosis regulators and are able to sense the local microenvironment as well as interact with normal B GC cells.19 The ability of FL cells to bind to FDCs via the sIg-Mann–DC-SIGN axis could allow the tumor to hijack a supportive interaction without the need for an antigen.
Expression of DC-SIGN in FL tissue. A representative case of FL is shown without (left) or with (right) staining with antiDC-SIGN by immunofluorescence. In the left panel, M2 macrophages, mainly in the interfollicular area, are stained red with anti-CD163, and FDCs in the neoplastic follicles outlined in dotted white lines are stained blue with anti-CD23. In the right panel, M2 macrophages costained with green anti-DC-SIGN appear yellow. FDCs costained with green anti-DC-SIGN appear cyan.
Expression of DC-SIGN in FL tissue. A representative case of FL is shown without (left) or with (right) staining with antiDC-SIGN by immunofluorescence. In the left panel, M2 macrophages, mainly in the interfollicular area, are stained red with anti-CD163, and FDCs in the neoplastic follicles outlined in dotted white lines are stained blue with anti-CD23. In the right panel, M2 macrophages costained with green anti-DC-SIGN appear yellow. FDCs costained with green anti-DC-SIGN appear cyan.
The frequency and distribution of FDCs in FL, among the infiltrating lymphoma cells, is complex and varies between cases. However, where FDCs persist, there appears to be a coordinated expression of CD23 and DC-SIGN, as found by our collaborators, Richard Burack and Yi Zhang (Figure 2). More details of the cellular composition of FL tissues and the expression of DC-SIGN are described in an accompanying review by Radtke and Roschewski.59 The main conclusion at this point is that FL cells can interact with both FDCs and M2 macrophages, both expressing DC-SIGN. Although cellular composition will vary between cases, there is always DC-SIGN available in the microenvironment of lymphoma tissues.
BCR Ig mannosylation in lymphoma
FL
The high incidence of AGS motifs for glycan addition to Ig heavy chain variable region (IGHV) of classical FL has been widely confirmed.21,22 In a tumor noted for its heterogeneity, AGSs are virtually universal and even the few cases with no sites in the IGHV could have them in the IGV of light chains, although these are usually found together with motifs in the heavy chains.3,23 Interestingly, AGSs do not accumulate in the nonfunctional IGHV of FL cells, confirming that they are positively selected for the functional allele.3 AGSs are found in both IgM+ (90%) and IgG+ cases (73.5%),22 with a lower incidence in the latter, pointing to possible heterogeneity in this subset. The location of the AGSs in the IGV sequences was known to be in the CDRs, with influences on location arising from appropriate germ line (GL) sequences and mutable hotspots, but was not derived from preferential IGHV usage.3
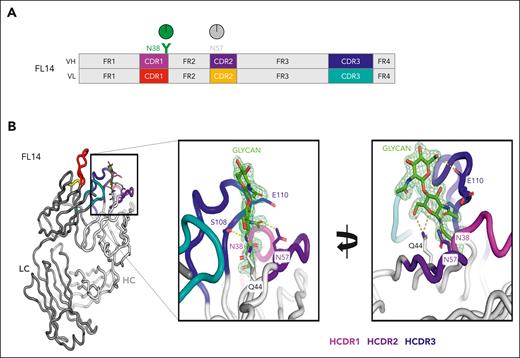
Crystallographic analysis of lymphoma-derived Fabs has revealed the importance of this positioning.13 It turns out that the CDR sequences selected in the FL cells act as a shield surrounding the added mannoses, apparently blocking access to the relatively large α-mannosidase 1 enzyme required for further processing of the glycan during Golgi transit (Figure 3). An illustrative case of FL selected for Fab crystallization had 2 heavy chain N-glycosylation sites, 1 in CDR1 at position N38 and 1 GL site in CDR2 at N57 of the IGH4-34 V region, which appears to never undergo N-glycosylation23 (Figure 3A). The crystal structure confirms the liquid chromatography–mass spectrometry analysis, which detected oligomannoses in CDR1 with no glycan addition at the GL site.13 Further analysis reveals that the oligomannosylated site at N38 is buried among the CDR loops, consistent with the inability of α-mannosidase enzymes to trim it for further processing (Figure 3B).
An illustrative example of a case of FL to reveal the nature and structure of the glycan-modified IGV region. (A) Site-specific glycan composition was determined by liquid chromatography–mass spectrometry, and oligomannoses at N38 in CDR-1 of the Ig heavy chain (HCDR1) are depicted in green. There were no sites in the Ig light chain. The gray circle at the GL position N57 indicates no detectable glycan addition. (B) Crystals of the derived Fab′ were analyzed at 1.65 Å resolution, and HCDRs are colored as indicated. The glycan is shown as 2 resolved N-acetyl glucosamine residues at position N38, which are attached to oligomannoses. More detail is available in the article by Chiodin et al.13 HC, heavy chain; LC, light chain.
An illustrative example of a case of FL to reveal the nature and structure of the glycan-modified IGV region. (A) Site-specific glycan composition was determined by liquid chromatography–mass spectrometry, and oligomannoses at N38 in CDR-1 of the Ig heavy chain (HCDR1) are depicted in green. There were no sites in the Ig light chain. The gray circle at the GL position N57 indicates no detectable glycan addition. (B) Crystals of the derived Fab′ were analyzed at 1.65 Å resolution, and HCDRs are colored as indicated. The glycan is shown as 2 resolved N-acetyl glucosamine residues at position N38, which are attached to oligomannoses. More detail is available in the article by Chiodin et al.13 HC, heavy chain; LC, light chain.
Even if the loops appear to lie closest to N-acetyl glucosamine sugars, this is sufficient to affect the steric access of α-mannosidases to proximal oligomannoses.24 This block on processing creates a “mannose paradox,” in which oligomannoses are hidden from α-mannosidase enzymes but remain visible to lectins.25 It seems that FL cells share a strategy with some pathogens to retain oligomannoses to capture DC-SIGN for different purposes. For FL, shielding by the CDRs from further modification can be considered another step in the multifactorial balance struck during lymphomagenesis.
The steric influence is clearly confirmed by the presence of fully processed oligosaccharides in the constant region of the same Ig molecule,4,5 demonstrating that the biosynthetic route for Igs of the FL BCR passes through normal Golgi stacks. The focus of oligomannose insertion into “suitable” acceptor IGV positions is made even clearer from the fact that sites in the framework regions of some cases of FL (usually additional to CDR sites) or, in certain subsets of DLBCL, commonly do not have oligomannose termination.13 There may be additional advantages to the insertion of oligomannoses into the BCR of FL cells because their presence actually blocks access of the original antigen.26 Hypothetically, such a block would prevent stimulation by antigen, which would drive unwanted differentiation.
Therefore, the CDR location is required for the consequential restriction of oligomannosylation of the BCR in FL and can be distinguished from the addition of glycan to the framework IGV regions of certain autoantibodies, such as the anti-citrullinated protein antibodies found in rheumatoid arthritis.27 These commonly acquire AGSs in IGV sequences, which are largely located in the framework regions and are fully glycosylated with the addition of terminal sialic acids.27 This underlines the importance of positioning of the AGS, which is strikingly different from that in FL. The function of glycans in autoimmune diseases remains speculative; however, in chronic autoimmune conditions, an influence on the selection of certain autoantibodies has been proposed.28
The essential nature of oligomannose insertion for FL is supported by the finding that, despite ongoing somatic hypermutation, AGS are retained by all individual emerging clones during the course of the disease.29 In the rare case in which a particular motif may be lost, another emerges at a different position. AGSs are also present in FL cells located at separate tissue sites and following transformation,29 raising the question of the incidence and positioning of AGSs in DLBCL.
DLBCL
From an earlier analysis, AGSs were detectable in 13 of 32 cases of DLBCL,3 but the more recent subdivision of this lymphoma based on transcriptomic analysis30 has now allowed for the correlation of AGS incidence among large numbers of cases of newly defined subtypes.13 As might be anticipated from the fact that GC B-cell–like DLBCL (GCB-DLBCL) is known to be closer to FL,30 motifs were highly associated with this category (>60%) and were rarer in activated B cell (ABC)-DLBCL (>13%) or the unclassified subtype (>24%). Critically, the vast majority (87%) of motifs in GCB-DLBCL were located in CDRs, as in FL, whereas >50% of the few found in ABC-DLBCL were located in framework regions.13 Interestingly, in the minor subset of FL, which is t(14;18) negative, AGSs were found only in the framework regions.31 This location and the tendency to overuse the IGHV4-34 gene connects these cases to ABC-DLBCL.32,33 With regard to IGHV4-34, as mentioned already, there is a GL AGS sequence in IGHV CDR2, but because this is not N-glycosylated, it can be discounted as a glycan-mediated influence.23
Among the genetically defined subgroups of GCB-DLBCL, 74% of AGS+ cases with a CDR location were classified with mutations in EZH2 and BCL2 translocations (EZB).13 This is expected, given that EZB cases are defined by a BCL2 translocation and mutations in epigenetic regulators, which are characteristic features of FL. Adding in the presence of AGSs draws all the features of FL together, consistent with the origin of a proportion of, but not all, EZB cases from FL. Equally, some cases with AGSs did not align with the classification as EZB. Although there is a strong overlap with the EZB category, the presence of AGSs in CDRs could allow for greater definition of the FL-derived subtype. This could be of clinical value because preliminary assessment of DLBCL revealed that cases within the EZB subtype, which mirrored FL having AGSs in the CDRs, progressed more rapidly than those without these AGSs.13 This distinction remained even when cases with MYC translocation were removed, although the numbers were small and further studies are required.
Recently, a transcriptionally defined microenvironmental influence has been revealed as potentially acting on the genetic subdivisions of DLBCL, with possible clinical relevance.34 It will be very interesting to map the AGS patterns onto the new categories, and this is in progress.
BL
Burkitt lymphoma (BL) is generally considered to originate from the dark zone of the GC. It comprises 2 main categories: endemic BL, mainly found in sub-Saharan Africa and highly associated with Epstein Barr virus; and sporadic BL, which is less commonly associated with Epstein Barr virus and includes cases associated with immunodeficiency, such as HIV/AIDS.35 Both categories carry chromosomal translocations involving the MYC oncogene on chromosome 8 and the Ig locus, with the most common being t(8;14).35 However, as for FL, despite the loss of 1 allele, most cases retain the expression of surface Ig, suggesting an involvement of the BCR in a pathogenesis very different from that of FL. In a few cases of sporadic BL, a drive by modified autoantigens has been reported,36 but in others, there is evidence of an antigen-independent tonic stimulation.37
A survey of the IGHV genes for N-glycosylation sites (AGSs) revealed a high incidence in endemic BL (14 of 17 cases) and a significant but lesser incidence of sporadic BL (10 of 23 cases), which tends to have lower levels of somatic hypermutation.38 In terms of the location of sites, ∼50% of each BL category was in the CDRs, pointing to a similarity to FL, possibly in lectin dependence. The presence of an AGS adds discrimination between tumor types, illustrated by the heterogeneity within DLBCL and by the very low incidence of AGS in mucosa-associated lymphoid tissue lymphoma, likely derived from marginal zone B cells.38 It will be of interest to probe for oligomannosylation in the BCR of BL cases to assess the potential influence of a microenvironmental lectin on these aggressive tumors.
DC-SIGN
The major function of DC-SIGN is to recognize pathogens. It is part of the “spy network” of protection against infection39 and is expressed by subepithelial dendritic cells, FDCs (Figure 2), and M2-polarized macrophages. DC-SIGN binds to oligomannoses expressed by a range of infectious organisms, including viruses, such as HIV-1, mycobacteria, and fungi. In HIV-1 infection, DC-SIGN-expressing dendritic cells ferry virus bound via the highly mannosylated gp120 from the mucosa to T cells for delivery via CD4/CCR5. In this regard, it acts as a transreceptor, which does not activate ferrying cells directly, although it can modulate other activation pathways, potentially leading to immunosuppression.40,41 Evidently, this relatively quiet anchoring and supportive function of DC-SIGN-expressing cells is captured by lymphoma cells for their own ends.
Structurally, DC-SIGN is a type 2 (C-terminus exposed outside the lipid bilayer) membrane protein that forms stable tetramers. It is a lectin, which binds target sugars via 4 calcium-dependent CRDs39 linked to oligomerized neck regions (Figure 1B). The clustered domains generate a multivalent glycan-binding surface, with each CRD possessing a Glu-Pro-Asn motif that preferentially binds to oligomannose-type saccharides.39
Similar to other lectins, DC-SIGN has a relatively low affinity for target oligomannoses, relying on multivalency for increasing avidity. Understanding the operation of sIg-Mann interactions with DC-SIGN requires new thinking because, in contrast to most protein receptor-ligand interactions, binding does not function as a deterministic on-off switch but rather as a progressive tuning of a cell-cell interaction (Figure 4A).39
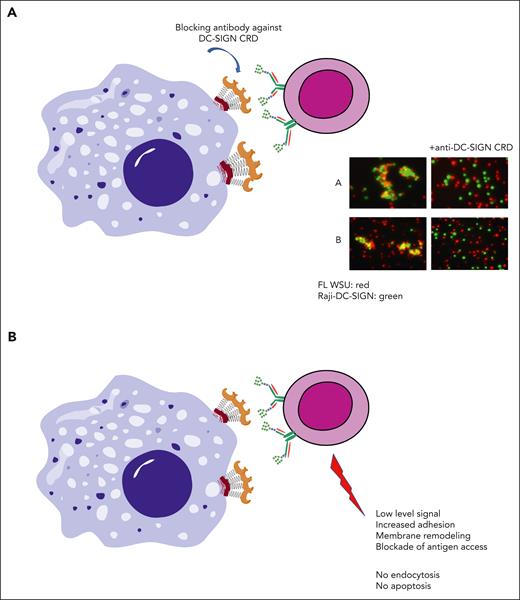
Consequences of interaction of DC-SIGN with mannosylated BCR on FL cells. (A) The clustering of DC-SIGN–expressing cells with oligomannoses in the BCR of FL cells can be blocked by an anti–DC-SIGN antibody against the CRD.13 (B) Interaction of FL cells expressing sIg-Mann with DC-SIGN in vitro generates a low-level BCR signal, membrane remodeling, increased adhesion, and blockade of antigen access12,15,26,42 but no detectable endocytosis or apoptosis.13,15
Consequences of interaction of DC-SIGN with mannosylated BCR on FL cells. (A) The clustering of DC-SIGN–expressing cells with oligomannoses in the BCR of FL cells can be blocked by an anti–DC-SIGN antibody against the CRD.13 (B) Interaction of FL cells expressing sIg-Mann with DC-SIGN in vitro generates a low-level BCR signal, membrane remodeling, increased adhesion, and blockade of antigen access12,15,26,42 but no detectable endocytosis or apoptosis.13,15
Interaction of DC-SIGN with lymphoma cells
It is difficult to mimic in vitro the low affinity–high avidity interactions that are likely to occur between cells in vivo. However, the tetrameric DC-SIGN–Fc soluble reagent binds specifically to FL-expressed Ig-Mann5 and induces the activation of upstream BCR signaling responses,5,12,42 comparable with those induced by anti-IgM, but at a lower level. Despite the weaker response, DC-SIGN is able to induce a similar downstream transcriptional response to anti-IgM in cell lines and primary FL cells.42 The pathways activated included those associated with B-cell survival, proliferation, and cell-cell communication. In addition, the engagement of DC-SIGN mediates intracellular cross talk via BCR, downregulating the expression of chemokine receptors, such as CXCR4.42
FL cell–expressed sIg-Mann can also directly interact with DC-SIGN+ cells, either via a transduced cell line or cells matured from monocytes by granulocyte-macrophage colony-stimulating factor/interleukin-4, forming clusters that can be broken by a monocloncal antibody (MoAb) directed at the CRDs13,43 (Figure 4A). More recently, we found that engagement of the BCR by DC-SIGN induces the adhesion of FL cells to the integrin ligand VCAM-1 via proximal kinases and actin regulators.15 Because FDCs express VCAM-1,44 which is retained in the FL,16 this could promote the beneficial adhesion of FL cells to FDCs.
Despite some similarities, there is a critical difference between the effects of anti-IgM and DC-SIGN in FL cells. The binding of high-affinity anti-IgM gives a strong signal to B cells and leads to endocytosis and death, the latter only partly ameliorated by BCL-xL.45 A similar BCR-induced death occurs in B cells from chronic lymphocytic leukemia and is not prevented by BCL-2.46 A parallel phenomenon is seen in a mouse model in which healthy memory B cells are silenced by an engaging high-affinity antigen in the absence of T cells.47 These findings illustrate that B cells are vulnerable to BCR-induced death unless protected by T cells, possibly reflecting a mechanism that controls antibody production.
In contrast, DC-SIGN binding activates BCR pathways in FL cells but does not induce endocytosis or death,13,15 thereby providing an ideal protected environment with a “Goldilocks,” intermediate signaling level for tumor cells to survive and proliferate (Figure 4B). The Goldilocks principle is common in biology and refers to a balancing act, in which structural stability and functional properties are poised at values that are just right for the environment the cell faces. Tumor cells are no exception to this.
However, although the binding of DC-SIGN to sIg-Mann on FL cells does not induce endocytosis, it does modulate BCR function. Pretreatment of either FL-derived cell lines or primary FL cells with DC-SIGN appears to paralyze the BCR so that it is unable to respond to stimulation via anti-IgM.42 This is not due to blockade but seems, instead, to be caused by a nanoscale reorganization of surface IgM-Mann, rendering it less able to signal.48 The effect is rapidly reversible in vitro so unlikely to be mediated by transcriptional changes. Lectin-mediated modulation of receptor function is reminiscent of the effects of galectin on T-cell function and has been explained there as due to the formation of a galectin lattice which “freezes” receptor function.49 The effect of DC-SIGN may be another strategy for protecting FL cells from BCR-mediated stimulation by antigen.
Concluding remarks
The mandatory insertion of mannoses into the antigen-binding site of FL cells points to the importance of this post-translational feature in lymphomagenesis. The sequence of events that incorporate this change in the nature and function of the BCR is shown in Figure 5. Insertion of oligomannose occurs early and appears to be a critical step toward full-blown FL. Together with the upregulation of BCL2, this allows the cell to persist and accumulate other mutations, often in epigenetic regulators, which then drive the cell to further escape from control.50 Some of these mutations in chromatin-modifying genes, such as CREBBP, occur before overt disease.51 A multistep pathway is likely to occur in all relatively indolent tumors, but this is well illustrated in FL. The BCR modification occurs in all tissues analyzed, although the motif can differ, in line with independent events at separate sites. However, it is not just a preliminary event, given that it persists over time, even being carried through transformation. Because every case of FL has a different IGV sequence, the AGSs will vary in number and position, raising the question of whether this variation grades the ability of the FL cell to interact with the microenvironment. A correlation between the ability to bind DC-SIGN in vitro and clinical behavior would be a simple starting point to answer this question.12 There could also be an association with transformation to a subset of DLBCL, thereby adding to the predictive power of the genetic background.52
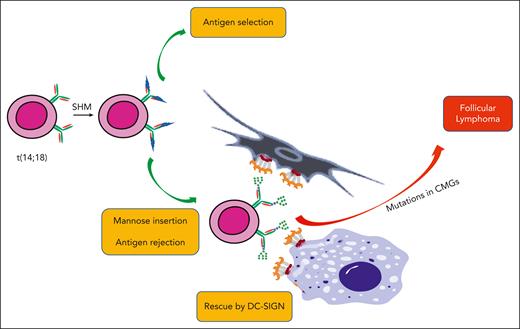
The potential influence of mandatory oligomannoses inserted into the BCR during the development of FL. The first event in the bone marrow is t(14;18) translocation, which upregulates BCL2. These B cells then, apparently, undergo somatic hypermutation (SHM) and antigen selection. There is a likely bifurcation of cells that are able to bind antigen and those that have acquired oligomannoses in the BCR, which cannot undergo antigen selection. These “rejects” may be lost or could be rescued by DC-SIGN on FDCs and/or macrophages, forming a population awaiting mutations in chromatin-modifying genes (CMGs) to drive lymphomagenesis.
The potential influence of mandatory oligomannoses inserted into the BCR during the development of FL. The first event in the bone marrow is t(14;18) translocation, which upregulates BCL2. These B cells then, apparently, undergo somatic hypermutation (SHM) and antigen selection. There is a likely bifurcation of cells that are able to bind antigen and those that have acquired oligomannoses in the BCR, which cannot undergo antigen selection. These “rejects” may be lost or could be rescued by DC-SIGN on FDCs and/or macrophages, forming a population awaiting mutations in chromatin-modifying genes (CMGs) to drive lymphomagenesis.
The function of this structural change in the BCR is beginning to emerge: the inserted oligomannoses have multiple potential roles including protective persistent interaction with DC-SIGN, blockade of antigen access, and downmodulation of BCR function. Although these functions have been documented in vitro, it now turns out that they match in vivo observations. DC-SIGN is plentiful in FL tissues and is expressed by M2-polarized macrophages, largely in the interfollicular zone, but also by FDCs, first on the FDC network and then on scattered fragments as the disease progresses. The tissue localization is presented in another article in this Review Series by Andrea Radtke and Mark Roschewski,59 and, via a collaboration with Richard Burack and Yi Zhang, we have similar unpublished findings, with a preliminary example shown in Figure 2. An urgent question is whether the presence and number of either of these cell types correlate with disease behavior. The role of macrophages has been much debated in FL, with initial findings that increased numbers correlated with poorer prognosis but a later reversal of this correlation, probably because of the need for macrophages to mediate the effect of anti-CD20.53,54 In terms of DC-SIGN, it is not yet possible to know whether FDCs or macrophages are the most important supporting cells for FL, and it could be both.
A speculative summary of how the documented effects of DC-SIGN could enable lymphomagenesis is shown in Figure 5. It is clear that B cells that carry the t(14;18) translocation are perfectly able to undergo somatic hypermutation and presumably antigen engagement. For normal B cells and likely for cells expressing BCL-2, the introduction of sites for N-glycosylation occurs during somatic hypermutation, but these sites are not generally retained. The reason could be that oligosaccharides could block antigen access, thereby opposing antigen selection and leading to the death of unselected cells. However, given the generation of sites in the CDRs that are “just right” for termination at oligomannoses, these cells free themselves of reliance on antigen by capturing DC-SIGN–expressing FDCs and benefiting from their antiapoptotic support. It could, therefore, be seen as a rescue of B cells unable to bind antigen and, therefore, officially be rejected. Furthermore, help from M2-macrophages would be on offer and could operate on cells in transit through the tissue. The mechanism of support appears to include a nourishing low-level BCR signal coupled with protection against strong stimulation, which in normal B cells would drive differentiation/death. Downregulation of major histocompatibility complex class II in FL would similarly protect against the provision of T-cell help, again holding the cells back from unwanted differentiation.55 The scene would be set for the accumulation of mutations that drive lymphoma development.56
Although the focus here is on the critical interactions between the FL BCR and DC-SIGN–expressing cells, there are many other influences operating in the FL tissue, which are discussed in detail in an accompanying review in this issue of Blood from Karin Tarte’s laboratory.57 Unraveling the biology should reveal new potential targets to add to current therapy, and there is a clinical aspect of the DC-SIGN interaction that is worth exploring. Antibodies against the CRD of DC-SIGN are available already, having been developed for other purposes58 and more are being produced. The aim would be to simply block the sIg-Mann–DC-SIGN interaction seemingly required by FL cells, ideally at an early stage of the disease, although retention of sites suggests that reliance on DC-SIGN continues throughout the disease. DLBCL is another story, and although there is a clear subset that carries oligomannosylated CDR-located sites, it remains to be seen whether this is just a useful marker or an antibody would be effective in this case. For FL, which is surrounded by an overwhelmingly heterogeneous population of T cells and stromal cells, as will be described in other reviews in this series, we conclude that the tumor cells cannot proceed without oligomannosylated BCRs.
Acknowledgments
This article contains contributions from many investigators, especially Giorgia Chiodin, from the Francesco Forconi group. F.K.S. acknowledges the contributions of the unpublished data from Richard Burack and Yi Zhang. The tissue architecture and location of DC-SIGN have been discussed by Andrea Radtke, an author of another article in this Review Series, from the group of Lou Staudt, who has also contributed to the discussion. The authors also acknowledge the major contributions from their glycobiology collaborators, Max Crispin and Joel Allen, and Patrick Duriez in Southampton; and for crystallographic analysis by T.A. Bowden of the Oxford Glycobiology Institute. The authors thank Lynsey Block for invaluable help in preparing the manuscript.
This work was supported by Blood Cancer UK (F.F.), Cancer Research UK BTERP (F.F. and Mark Cragg), Cancer Research UK Accelerator ECRIM-M3 (F.F.), and the Eyles Cancer Immunology fellowship (Giorgia Chiodin).
Authorship
Contribution: F.K.S. wrote the review and F.F. contributed new data.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Freda K. Stevenson, School of Cancer Sciences, University of Southampton, Southampton SO16 6YD, United Kingdom; email: fs@soton.ac.uk.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal