Our group has reported that transient CD19 antigen masking of leukemic cells with the CD19 monoclonal antibody (mAb), tafasitamab, resulted in reduction of CD19 chimeric antigen receptor T cell (CART19) apoptosis, enhanced CAR T proliferation, improved anti-tumor activity, diminished tumor pyroptosis, and reduced severity of cytokine release syndrome (CRS) in preclinical models. In this report, we aimed to study the mAb characteristics responsible for these observations.
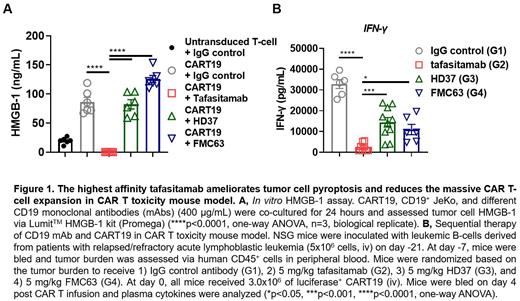
Specifically, we hypothesized that binding affinity of CD19 mAbs might affect the subsequent activation and apoptosis of CART19 as well as tumor cell pyroptosis in preclinical models. To test our hypothesis, we first determined the affinities of the CD19-targeting antibodies tafasitamab, HD37 and FMC63 to recombinant human CD19 protein. Tafasitamab demonstrated the strongest binding affinity among the three tested antibodies [tafasitamab ( K D: 1.0±0.2 nM), HD37 ( K D: 4.1±0.3 nM), FMC63 ( K D: 3.4±1.2 nM)]. Next, we studied CART19 apoptosis in the presence of these CD19 mAbs with different binding affinities. Here, we co-cultured CART19, CD19 + mantle cell lymphoma JeKo, and 400 μg/mL of CD19 mAb for 1 hour and measured CD3 + 7-AAD - Annexin V + T cells with flow cytometry. The number of apoptotic CART19 cells was significantly reduced by tafasitamab co-treatment compared to HD37, FMC63, or control IgG [control IgG (mean = 19.1, standard deviation (SD) = 0.8), tafasitamab (mean = 13.1, SD = 2.9), HD37 (mean = 17.8, SD = 0.8), FMC63 (mean = 18.4, SD = 1.0)]. We also performed an in vitro high mobility group box 1 (HMGB1) assay to assess if different affinities of the CD19 mAbs affect tumor cell pyroptosis triggered by CART19. Here, we co-cultured CART19, CD19 + JeKo, and 400 μg/mL of CD19 mAb for 24 hours and assessed HMGB1 on JeKo. In analogy to the apoptosis data, tafasitamab was the only mAb that prevented tumor cell pyroptosis, indicated by reduced HMGB1 expression (Fig 1A).
Given these results, we aimed to study the impact of CD19 occupancy with different CD19 mAbs prior to CART19 infusion on tumor pyroptosis in vivo. We utilized a mouse model that mimics CAR T toxicities. Immunocompromised NSG mice were inoculated via tail vein with 5x10 6 leukemic cells derived from patients with relapsed/refractory acute lymphoblastic leukemia. Two weeks later (day -7), tumor burden was assessed by peripheral blood sampling. Leukemic cells were defined as murine CD45 -, human CD45 + CD19 + CD20 + via flow cytometry, and volumetric absolute number was calculated. Mice were then randomized according to tumor burden to receive IgG control [Group 1 (G1)], tafasitamab (G2), HD37 (G3), or FMC63 (G4) (5 mg/kg, ip) at day -7. CRS was determined by weight loss, development of motor weakness, poor body posture, and elevated serum cytokine levels. At day 0, all mice received 3x10 6 luciferase + CART19 via tail vein. Mice were monitored with daily bioluminescent imaging (BLI) to track and quantify CART19 expansion and physical assessment to detect toxicity. BLI on day 4 revealed that CART19 expansion in the tafasitamab-treated group (G2, mean = 1x10 7, SD = 2.3x10 7) was significantly lower than in the control group (mean = 3.5x10 7, SD = 9.3x10 7), the HD37-treated group (G3, mean = 4x10 7, SD = 6.6x10 7), and the FMC63-treated group (G4, mean = 2x10 7, SD = 3.59x10 7), [G1 vs G2: 95% confidence interval (CI) 3.5x10 7 to 1.1x10 8, p <0.0001, G1 vs G3: 95% CI -7.5x10 7 to -1.1x10 8, p=0.005, G1 vs G4: 95% CI -4.5x10 7 to 1.9x10 7, p=0.71] indicating that tafasitamab pretreatment significantly reduced early activation of CART19 compared to IgG control and HD37 in this model. Mice were bled on day 4 to analyze plasma cytokine. Tafasitamab-treated mice (G2) secreted significantly less IFNγ (Fig 1B), IL10 (Mean G1: 78, G2: 2.52, G3: 12.2, G4: 13.9 pg/mL), MDC (Mean G1: 4072, G2: 445, G3: 921, G4: 1364 pg/mL), and MIP1β (Mean G1: 283, G2: 59.1, G3: 148, G4: 123 pg/mL) compared to G1, G3, and G4. Satellite mice were euthanized on day 1 and spleens were harvested. Western blot analysis of splenocytes revealed that G1, 3, and 4 showed significantly lower expression of apoptosis-inducing protein gasdermin E on leukemic cells compared to G2, indicating reduced tumor pyroptosis in G2.
In summary, application of the high affinity mAb tafasitamab prior to CART19 resulted in reduced CAR T apoptosis, diminished tumor cell pyroptosis, and reduction of CRS compared to lower affinity CD19 mAbs in preclinical models.
Disclosures
Sakemura:Janssen Pharmaceutical: Honoraria. Jaehrling:Morphosys: Current Employment. Ilieva:MorphoSys AG: Current Employment. Patra-Kneuer:Morphosys: Current Employment. Heitmüller:MorphoSys AG: Current Employment. Steidl:Morphosys: Current Employment. Kenderian:CapstanBio: Consultancy, Other: Scientific advisory board; LEAHLabs: Consultancy, Current equity holder in private company, Research Funding; Luminary therapeutics: Other: scientific advisory board ; Morphosys: Research Funding; MustangBio: Patents & Royalties; Kite/Gilead: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Juno/BMS: Other: Membership on an entity's board of directors or advisory committees, Research Funding; Humanigen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Patents & Royalties, Research Funding, Speakers Bureau; Lentigen: Research Funding; Sendero: Patents & Royalties; Torque: Consultancy; Tolero/Sumtomo: Research Funding; Mettaforge: Patents & Royalties.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal