In this issue of Blood, Schmitt et al1 have identified the bromodomain-containing 4 (BRD4) protein as a suppressor of ferroptosis in diffuse large B-cell lymphoma (DLBCL). This study provides evidence that combination of bromodomain and extraterminal motif (BET) inhibitors and ferroptosis inducers synergistically kill germinal center B-cell–like (GCB) DLBCL.1
DLBCL is the most common subtype of non-Hodgkin lymphoma worldwide. Treatment of patients with DLBCL relies on an immunochemotherapy regimen based on rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP).2
Historically, gene expression profiling had identified 2 major subtypes of DLBCL: GCB and activated B-cell–like (ABC) lymphomas.3 More recently, 2 molecular classification schemes based on cooccurrence of genetic mutations MCD (cooccurrence of MYD88[L265P] and CD79B mutations), BN2 (cooccurrence of BCL6 fusions and NOTCH2 mutations), N1 (NOTCH1 mutations), EZB (cooccurrence of EZH2 mutations and BCL2 translocations), or C1-5 clusters revealed phenotypically different subtypes in their responses to immunochemotherapy and outcome, providing a potential diagnostic tool for precision-medicine approaches in DLBCL.4,5 Unfortunately, ≈40% of patients display refractory disease or short-term relapse,6 posing a critical therapeutic challenge. Therefore, identification of novel DLBCL vulnerabilities is of urgent medical need.
Ferroptosis is an iron-dependent form of regulated cell death that is driven by an overload of phospholipid peroxides and changes in cellular morphologic features, including shrinkage of the mitochondrial membrane. Notably, analysis of >100 cell lines demonstrated that DLBCL cell lines were particularly sensitive to the ferroptosis-inducing small-molecule erastin,7 revealing an unexpected therapeutic opportunity for DLBCL. More recently, Schmitt et al demonstrated that dimethyl fumarate (DMF), a US Food and Drug Administration–approved drug, promoted lipid peroxidation and efficiently induced ferroptotic cell death in GCB DLBCL cells.8
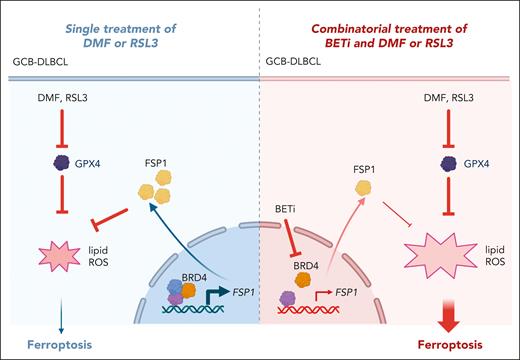
In the current study, Schmitt et al conducted a screen using a compound library targeting 140 epigenetic modulators alone or in combination with DMF. This analysis identified several BET inhibitors as enhancers of DMF-induced lipid peroxidation in GCB DLBCL. BRD4 is an important epigenetic transcriptional regulator in cancer,9 which promotes the expression of oncogenes via interaction with acetylated histones and assembly of superenhancers (SEs).10 Chemical inhibition of BRD4 via the small-molecule BET-inhibitor (BETi) JQ1 disrupts the communication between SEs and target oncogene promoters. Notably, only a few previous studies had focused on the relationship of BRD4 to ferroptosis.
Schmitt et al demonstrated that genetic silencing of BRD4 expression synergically induced ferroptosis when combined with DMF, revealing BRD4 as a key player in protecting cells from lipid peroxidation. Using RNA sequencing, the authors identified BRD4 as a repressor of ferroptosis-related genes, such as SLC7A11 (a cystine transporter key for glutathione biosynthesis) and AIFM2 (the ferroptosis suppressive protein 1 [FSP1]). Consistently, chromatin immunoprecipitation sequencing further proved SLC7A11 and AIFM2 as direct BRD4-target genes. Accordingly, chemical inhibition or genetic downregulation of FSP1 phenocopied the effects of BET inhibitors and DMF in induction of ferroptosis. More important, the combinatorial treatment of BETi and DMF was effective in a patient-derived xenograft mouse model of DLBCL, providing the rationale to assess the combinatorial effect of BET inhibitors and ferroptosis inducers on GCB DLBCL in the clinic (see figure).
Schematic representation of the pathways involved in the synergic effect of the small-molecules ferroptosis inducers together with BETi.
Schematic representation of the pathways involved in the synergic effect of the small-molecules ferroptosis inducers together with BETi.
This study also raises interesting questions that will be important to address in the future. What is the molecular mechanism by which GCB DLBCL cells are more sensitive to ferroptosis compared with ABC DLBCL? Similarly, why are DLBCL cell lines more sensitive to ferroptosis than other cancer cell lines, and is this relationship similarly observed in in vivo mouse models of cancer? Finally, it will be important to determine whether ferroptosis inducers could improve or even replace the current standard-of-care R-CHOP therapy for patients with GCB DLBCL.
Better understanding of the mechanisms of ferroptosis may be critical in building synergistic or synthetic lethal interactions with other drugs. As such, this study provides a proof of concept for ferroptosis activation as a novel strategy for improving therapeutic outcomes for patients with DLBCL.
Conflict-of-interest disclosure: The authors declare no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal