TO THE EDITOR:
Bone marrow (BM) involvement impacts stage and prognosis in lymphoma, remaining an important factor in risk-stratification scores.1,2 Positron emission tomography (PET)/computed tomography (CT) has demonstrated high sensitivity to detect focal skeletal lesions compatible with BM involvement in patients with Hodgkin lymphoma (HL) and diffuse large B-cell lymphoma (DLBCL), obviating confirmatory biopsies in most patients.3-5 PET/CT is commonly integrated in the staging workup and assessment of treatment response across different lymphomas, though the ability of fluorodeoxyglucose (FDG) avidity to detect marginal zone lymphoma (MZL) remains questionable.6-8 Furthermore, whether PET/CT accurately predicts BM involvement in MZL is largely unknown. The Lugano classification supports BM evaluation by PET/CT in HL and DLBCL but acknowledges limited data in other histologies.7
MZL comprises 3 subtypes, each with unique biology, clinical features, and incidence of BM involvement.9 Extranodal MZL (EMZL) of mucosa-associated lymphoid tissue may arise at any site but exhibits site-specific differences with respect to etiology, genomics, and clinical course.10,11 EMZL affects the BM in approximately 10% of cases. FDG avidity of EMZL varies, depending on the involved extranodal site.8,12 Nodal MZL (NMZL) commonly presents with disseminated lymphadenopathy without evidence of extranodal or splenic disease, affecting BM in 30% to 40% of patients.10,12 NMZL frequently demonstrates FDG avidity (76%).13 Contrary to prior subtypes, splenic MZL (SMZL) typically presents with splenomegaly and peripheral blood and BM involvement (67% to100%), but peripheral lymphadenopathy is unusual.10,12 PET/CT is capable of identifying SMZL in 83% of cases.13
In this study, we assessed the predictive value of PET/CT to detect BM involvement and early prognostic implications in MZL using data from the Lymphoma Epidemiology of Outcomes (LEO) cohort. The LEO cohort is a prospective cohort from 8 academic cancer centers in which patients are enrolled within 6 months of lymphoma diagnosis and followed. All the patients signed an informed consent to participate in the LEO study, which was approved by the institutional review board of each of the participating centers. We extracted baseline data of patients with MZL enrolled between May 2015 and February 2020 and followed through July 2022. Using BM biopsy as the gold standard to define BM involvement, we reviewed radiology reports to detect cases for which evidence for BM involvement was based on PET/CT. Radiologists reading the PET/CTs had extensive expertise in reading these studies in patients with lymphoma. Radiologists and investigators on this study reviewing PET/CT reports were blinded to BM biopsy results. For review of the reports for this study, a strict data extraction methodology to document BM involvement by PET/CT was established defining test results as positive, negative, indeterminate, not done or unavailable. Patients with diffuse or focal BM involvement were defined as positive test results with BM involvement. Patients with indeterminate PET/CT findings or PET/CT not done or unavailable were excluded from the analysis. We defined event-free survival (EFS) as time from diagnosis to progression, retreatment, or death due to any cause and overall survival (OS) as time from diagnosis to death due to any cause estimated by Kaplan-Meier.
Of 706 patients with MZL enrolled in LEO, 311 (44%) with data on BM status by both biopsy and PET/CT were included in the analysis. We did not observe significant baseline differences between included and excluded patients, except for more frequent advanced stage (III-IV) in the analyzed cohort (53.7% vs 39.7%; P = .003) (supplemental Table 1). Characteristics of this cohort included a median age of 64 (range 24-89) years with most patients younger than 70 years (n = 217, 69.8%), non-Hispanic (n = 264, 84.9%), White (n = 276, 88.7%), without B symptoms (n = 246, 79.1%), and with normal lactate dehydrogenase (n = 185, 59.5%). The most common MZL subtype was EMZL (n = 208, 66.9%) followed by NMZL (n = 61, 19.6%) and SMZL (n = 42, 13.5%). Stage distribution was as follows: early stage (I-II; n = 144, 46.3%) and advanced stage (III-IV; n = 167, 53.7%). BM biopsy was positive in 99 (31.8%) and negative in 212 (68.2%) patients (Table 1).
Baseline patient characteristics
| Variable . | Total . | BM biopsy . | ||||
|---|---|---|---|---|---|---|
| Negative . | Positive . | |||||
| n . | % . | n . | % . | n . | % . | |
| Total | 311 | 100.0 | 212 | 100.0 | 99 | 100.0 |
| Age | ||||||
| <70 | 217 | 69.8 | 156 | 73.6 | 61 | 61.6 |
| ≥70 | 94 | 30.2 | 56 | 26.4 | 38 | 38.4 |
| Race | ||||||
| White | 276 | 88.7 | 183 | 86.3 | 93 | 93.9 |
| Black | 21 | 6.8 | 17 | 8.0 | 4 | 4.0 |
| Other | 6 | 1.9 | 5 | 2.4 | 1 | 1.0 |
| Not available | 8 | 2.6 | 7 | 3.3 | 1 | 1.0 |
| Ethnicity | ||||||
| Hispanic/Latino | 43 | 13.8 | 32 | 15.1 | 11 | 11.1 |
| Non-Hispanic | 264 | 84.9 | 178 | 84.0 | 86 | 86.9 |
| Not available | 4 | 1.3 | 2 | 0.9 | 2 | 2.0 |
| Stage | ||||||
| I-II | 144 | 46.3 | 144 | 67.9 | — | — |
| III-IV | 167 | 53.7 | 68 | 32.1 | 99 | 100.0 |
| LDH | ||||||
| Normal | 185 | 59.5 | 131 | 61.8 | 54 | 54.5 |
| Elevated | 56 | 18.0 | 27 | 12.7 | 29 | 29.3 |
| Not available | 70 | 22.5 | 54 | 25.5 | 16 | 16.2 |
| B symptoms | ||||||
| No | 246 | 79.1 | 175 | 82.5 | 71 | 71.7 |
| Yes | 60 | 19.3 | 33 | 15.6 | 27 | 27.3 |
| Not available | 5 | 1.6 | 4 | 1.9 | 1 | 1.0 |
| BM PET/CT | ||||||
| Negative | 273 | 87.8 | 210 | 99.1 | 63 | 63.6 |
| Positive | 38 | 12.2 | 2 | 0.9 | 36 | 36.4 |
| Variable . | Total . | BM biopsy . | ||||
|---|---|---|---|---|---|---|
| Negative . | Positive . | |||||
| n . | % . | n . | % . | n . | % . | |
| Total | 311 | 100.0 | 212 | 100.0 | 99 | 100.0 |
| Age | ||||||
| <70 | 217 | 69.8 | 156 | 73.6 | 61 | 61.6 |
| ≥70 | 94 | 30.2 | 56 | 26.4 | 38 | 38.4 |
| Race | ||||||
| White | 276 | 88.7 | 183 | 86.3 | 93 | 93.9 |
| Black | 21 | 6.8 | 17 | 8.0 | 4 | 4.0 |
| Other | 6 | 1.9 | 5 | 2.4 | 1 | 1.0 |
| Not available | 8 | 2.6 | 7 | 3.3 | 1 | 1.0 |
| Ethnicity | ||||||
| Hispanic/Latino | 43 | 13.8 | 32 | 15.1 | 11 | 11.1 |
| Non-Hispanic | 264 | 84.9 | 178 | 84.0 | 86 | 86.9 |
| Not available | 4 | 1.3 | 2 | 0.9 | 2 | 2.0 |
| Stage | ||||||
| I-II | 144 | 46.3 | 144 | 67.9 | — | — |
| III-IV | 167 | 53.7 | 68 | 32.1 | 99 | 100.0 |
| LDH | ||||||
| Normal | 185 | 59.5 | 131 | 61.8 | 54 | 54.5 |
| Elevated | 56 | 18.0 | 27 | 12.7 | 29 | 29.3 |
| Not available | 70 | 22.5 | 54 | 25.5 | 16 | 16.2 |
| B symptoms | ||||||
| No | 246 | 79.1 | 175 | 82.5 | 71 | 71.7 |
| Yes | 60 | 19.3 | 33 | 15.6 | 27 | 27.3 |
| Not available | 5 | 1.6 | 4 | 1.9 | 1 | 1.0 |
| BM PET/CT | ||||||
| Negative | 273 | 87.8 | 210 | 99.1 | 63 | 63.6 |
| Positive | 38 | 12.2 | 2 | 0.9 | 36 | 36.4 |
LDH, lactate dehydrogenase.
Across all MZL subtypes, biopsy-proven and PET/CT-suggested BM involvement was present in 11.6% (n = 36 of 311), and positive BM biopsy with negative PET/CT was observed in 20.3% (n = 63 of 311) (EMZL = 9.6%, n = 20 of 208; NMZL = 31.1%, n = 19 of 61; and SMZL = 57.1%, n = 24 of 42). For EMZL, 15.9% (n = 33) demonstrated pathologically confirmed BM involvement, and among those patients PET/CT was positive in 39.4% (n = 13) and negative in 60.6% (n = 20). For NMZL, 50.8% (n = 31) demonstrated pathologically confirmed BM involvement, and among those patients PET/CT was positive in 38.7% (n = 12) and negative in 61.3% (n = 19). Most patients (83.3%, n = 35) with SMZL demonstrated pathologically confirmed BM involvement, with positive PET/CT in 31.4% (n = 11) and negative in 68.6% (n = 24) (supplemental Table 2). Considering BM result from biopsy as the standard, PET/CT-based sensitivity and specificity in EMZL, NMZL, and SMZL were 39.4% and 98.9%, 38.7% and 100%, and 31.4% and 100%, respectively. PET/CT-based positive and negative predictive values in EMZL, NMZL, and SMZL were 86.7% and 89.6%, 100% and 61.2%, and 100% and 22.6%, respectively (supplemental Table 3). Similar parameters for BM biopsy if we would assume PET/CT as gold standard in this population are described in supplemental Table 4).
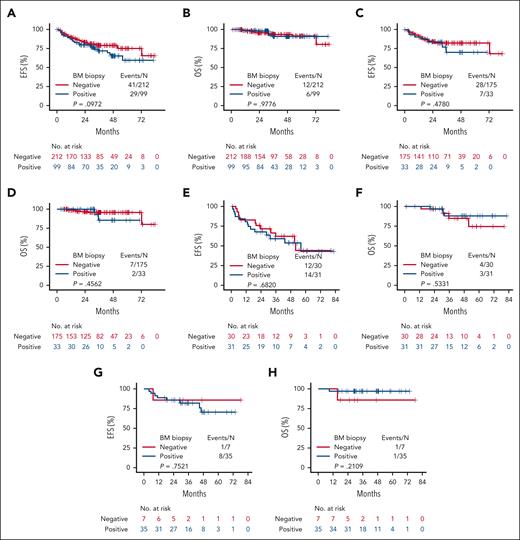
With a median follow-up of 35.4 months, we observed 70 EFS events and 18 OS events across all MZL subtypes. The 2-year EFS and OS were 81.7% (95% confidence interval [CI] 76.8%-85.7%) and 96.8% (95% CI, 93.9%-98.3%), respectively. The 2-year EFS and OS for EMZL were 84.7% (95% CI, 78.7%-89.1%) and 97.2% (95% CI, 93.5%-98.8%); for NMZL they were 69.6% (95% CI, 56.1%-79.6%) and 96.5% (95% CI, 86.9%-99.1%); and for SMZL they were 85.6% (95% CI, 70.7%-93.3%) and 95.2% (95% CI, 82.3%-98.8%), respectively. Patients exhibiting pathological BM involvement (n = 99) did not show significantly worse EFS (P = .097) or OS (P = .978) compared with those without BM disease (Figure 1A-B). Similarly, BM involvement was not associated with progression, relapse, and survival events within 24 months compared with those with negative biopsy (22.2% vs 19.9%; P = .66). Across all MZL subtypes, there were no significant differences in EFS (EMZL P = .48, NMZL P = .68, and SMZL P = .75, respectively) or OS (EMZL P = .46, NMZL P = .53, and SMZL P = .21, respectively) comparing patients with BM disease to those without BM disease (Figure 1C-H).
Survival analyses. EFS and OS across all MZL subtypes (A-B), in EMZL (C-D), in NMZL (E-F), and in SMZL (G-H) by BM involvement.
Survival analyses. EFS and OS across all MZL subtypes (A-B), in EMZL (C-D), in NMZL (E-F), and in SMZL (G-H) by BM involvement.
To our knowledge, this represents the first large national study evaluating the predictive value of PET/CT to detect BM involvement across MZL subtypes. Key findings of this analysis include the following: (1) the sensitivity of PET/CT to identify BM involvement in MZL was low, especially in patients with SMZL; (2) the negative predictive value was reasonable in EMZL but significantly lower in NMZL and SMZL; and (3) the presence of BM involvement does not correlate with early events and shorter survival in any subtype.
Over the last several years, growing data support the use of PET/CT to assess BM in HL and DLBCL. In patients with HL, focal bone lesions observed in PET/CT highly correlated with BM involvement with a sensitivity above 85% and negative predictive value of 99%.4,14 Most importantly, the presence of positive BM biopsy does not seem to affect treatment decision.4,15 Similar findings have been observed in DLBCL, in which PET/CT demonstrated high efficacy to identify BM involvement (sensitivity > 71% and negative predictive value of 95%).5,16-18 Furthermore, patients with BM involvement by PET/CT but not BM biopsy demonstrated similar survival to those with stage IV disease without marrow involvement.19 These data have supported a paradigm shift in the clinical use of BM biopsies, with many clinicians assessing BM involvement based solely on PET/CT results. The National Comprehensive Cancer Network guidelines do not recommend BM biopsy in patients with DLBCL with focal bone lesions and broadly do not recommend BM biopsy in HL staged with PET/CT.6,20
In low-grade lymphomas, most studies in this area have concentrated on follicular lymphoma, in which PET/CT demonstrated a sensitivity of 57% and negative predictive value of 81%.21 Furthermore, BM status does not appear to affect survival in FL.22 Although these values are better than those reported in our present study, they are significantly lower than prior observations in HL and DLBCL. The overall role of PET/CT in MZL remains unclear, with accumulating data supporting its use in SMZL and NMZL.8,13,23 In EMZL, FDG avidity seems to be dependent on the extranodal site with better detection rates in lung and head and neck compared with ocular adnexa and stomach.24 Further investigations should tackle detection rate by location. Prior studies reported a concordance between PET/CT and BM involvement between 12% and 37%.13,25 In the present study we demonstrate low sensitivity of PET/CT to detect BM involvement in MZL, highlighting the limitations of this test to fully capture advanced stage. One limitation of our study is the lack of data on the pattern of BM involvement (focal vs diffuse), and future studies should address this question. Acknowledging the relatively limited follow-up time, unspecific criteria for treatment selection, and the small number of patients in each subgroup may decrease the statistical power of the survival analysis, we did not observe significantly different early events that were shown to correlate with inferior outcomes,26,27 as well as survival rates based on BM status. These observations suggest that staging BM biopsies may not be needed in routine clinical practice. However, in clinical trials, in which BM status may affect treatment selection and accurate response to therapy needs to be collected, biopsies are still required because of the low sensitivity of PET/CT to detect involvement.
Acknowledgments
This work was funded by National Institutes of Health, National Cancer Institute grants P50 CA97274 and U01 CA195568. J.P.A. is supported by Peykoff Initiative from the Lymphoma Research Foundation and the Dwoskin Family Foundation. I.S.L. is supported by grant 1R01CA233945 and U01 CA195568 from the National Cancer Institute, the Intramural Funding Program from the University of Miami Sylvester Comprehensive Cancer Center, the Dwoskin Family Foundation, the Anthony Rizzo Family Foundation, and the Jaime Erin Follicular Lymphoma Research Consortium.
Authorship
Contribution: J.P.A. and I.S.L. designed and performed the research, analyzed data, and wrote the paper; and I.M.R., J.L.K., M.C.L., D.C., W.Z., S.H., T.M.H., P.M., J.R.C., C.S., B.S.K., J.B.C., J.W.F., J.R.C., and C.R.F. performed research, analyzed data, and wrote the paper.
Conflict-of-interest disclosure: J.P.A. received research funding from and is a consultant for ADC Therapeutics; and has an immediate family member who has served on the advisory boards of Puma Biotechnology, Inovio Pharmaceuticals, Agios Pharmaceuticals, Forma Therapeutics, and Foundation Medicine. J.L.K. received research funding from Atara BioTherapeutics, Oncternal Therapeutics, and Viracta Therapeutics and is a consultant for Gamida Cell, Janssen Pharmaceuticals, Morphosys, and TG Therapeutics. D.C. has received honoraria from Eisai and AstraZeneca. P.M. is a consultant for ADCT, AstraZeneca, BeiGene, BMS, Daiichi Sankyo, Epizyme, Genentech, Janssen Pharmaceuticals, Regeneron, and Takeda. J.R.C. received research funding from BMS, Genentech, and Genmab; is a consultant for BMS; and is on the safety monitoring board for Protagonist (none of these were related to the manuscript). B.S.K. received funding from AstraZeneca, Genentech, Abbvie, Celgene/BMS, BeiGene, and Hutchmed; is a consultant for AstraZeneca, ADT Therapeutics, Roche, Genentech, Abbvie, MEI, AcertaPharma, Pharmacyclics, Celgene/BMS, BeiGene, Kite, Janssen Pharmaceuticals, Incyte, Hutchmed, Seattle Genetics, TG Therapeutics, and Genmab; and is on the speakers bureau of Research To Practice. J.B.C. received research funding from BMS/Celgene, Lilly Oncology/Eli Lilly, BeiGene, Takeda, Genentech, HutchMed, AstraZeneca, and Novartis; and is a consultant for Aptitude Health, Kite Pharma/Gilead, Lilly Oncology/Eli Lilly, BeiGene, Janssen Pharmaceuticals, HutchMed, and AstraZeneca. J.R.C. received research funding from BMS/Celgene, Genentech, Genmab, and NanoString; and has membership on an entity’s board of directors or advisory committees at Genmab and Protagonist. C.R.F. received research funding from Iovance, Cellectis, EMD, Guardant, Eastern Cooperative Oncology Group, Burroughs Wellcome Fund, Ziopharm, V Foundation, Cancer Prevention and Research Institute of Texas: CPRIT scholar in cancer research, National Cancer Institute, Janssen Pharmaceuticals, Amgen, Allogene, Adaptimmune, Kite, Morphosys, Acerta, Takeda, Sanofi, Genentech/Roche, Bayer, Celgene, Gilead, TG Therapeutics, Abbvie, 4D, Xencor, Pharmacylics, and Pfizer; is a consultant for Denovo Biopharma, Genmab, Spectrum, Foresight Diagnostics, BeiGene, Genentech/Roche, Bayer, SeaGen, Celgene, Gilead, and Pharmacyclics/Janssen Pharmaceuticals, and is a current holder of stock options in privately held companies NPower and Foresight Diagnostics. I.S.L. has served on the advisory board of Adaptive Biotechnologies. The remaining authors declare no competing financial interests.
Correspondence: Izidore S. Lossos, Department of Medicine, Division of Hematology, University of Miami Miller School of Medicine, 1475 NW 12th Ave (D8-4), Miami, FL 33136; e-mail: ilossos@med.miami.edu.
References
Author notes
Requests for data sharing may be submitted to the corresponding author (ilossos@med.miami.edu).
The online version of this article contains a data supplement.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal