TO THE EDITOR:
Sézary syndrome (SS) is an aggressive subtype of cutaneous T-cell lymphoma, clinically presenting with erythroderma, lymphadenopathy, and atypical T cells (Sézary cells) in the skin, lymph nodes, and peripheral blood.1,2 As disease progresses, patients with SS develop severe immunodeficiency orchestrated by tumor cells and the tumor microenvironment (TME). This immunodeficiency is worsened by therapy and is responsible for a high incidence of life-threatening infections.3 Overexpression of CD39 and/or CD73 in malignant circulating SS T cells has previously been reported.4,5 Here we follow up on our preliminary data by investigating the functional role of the CD39/CD73 ectoenzymes in generating immunosuppressive extracellular adenosine (ADO) in patients with SS.6,7
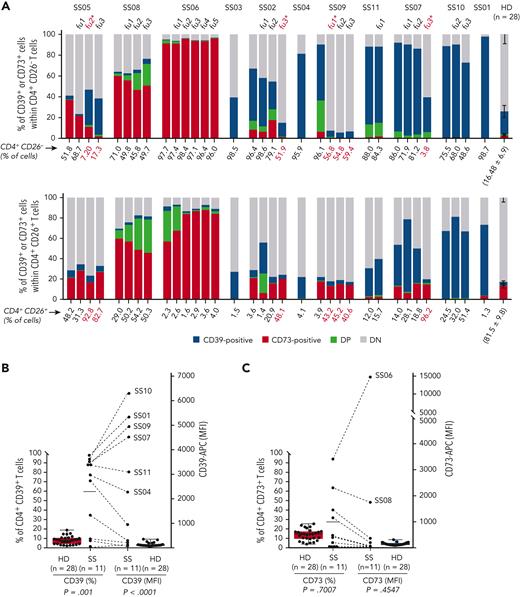
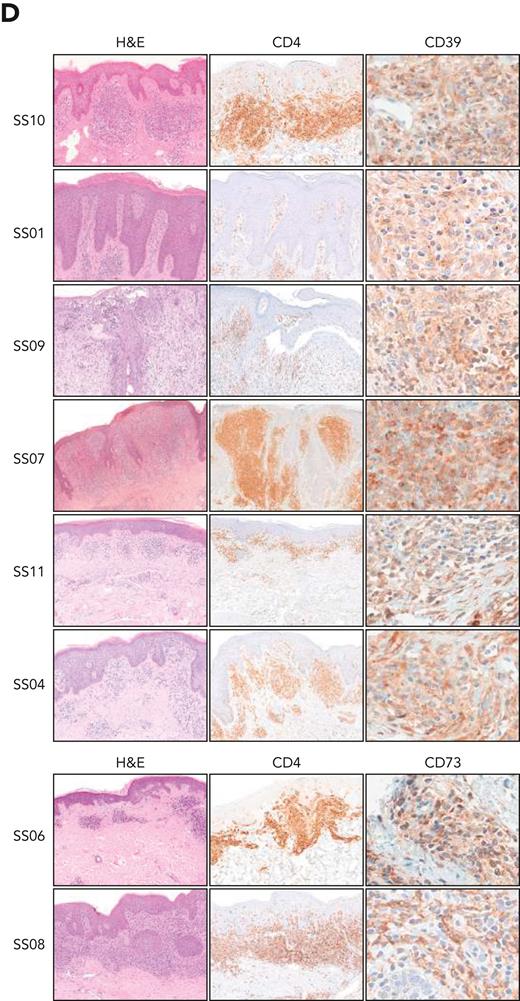
Our patient cohort consisted of 11 patients with SS (supplemental Table 1, available on the Blood website). At first encounter and at each subsequent follow-up visit flow cytometry analysis was performed on CD4+ T cells (containing the malignant SS cells) divided into CD26– and CD26+ subsets.8 Compared with 28 healthy donors (HDs), 8 patients with SS showed a higher percentage of CD26–/CD39+ cells and 3 showed a higher percentage of CD26–/CD73+ cells. A CD4+/CD39+/CD73+ cell subpopulation was visible in some patients. The expression of CD39 and CD73 was altered in all CD4+ T cells in most of the patients with SS and remained unchanged during follow-up, regardless of the therapy administered, with the exception of patients being treated with the anti-CCR4 (C-C chemokine receptor 4) antibody mogamulizumab, which promotes antibody-dependent cytotoxicity of the malignant T cells9 (Figure 1A; supplemental Table 2). Combined evaluation of the percentage of CD4+ T cells expressing CD39 or CD73 and the surface protein density, measured as median fluorescence intensity (MFI), revealed 6 patients exhibiting a high frequency of CD39+ T cells with high CD39 MFI (CD39high) and 2 exhibiting a high frequency of CD73+ T cells with high CD73 MFI (CD73high) (eg, percentage of CD39+ or CD73+ cells above the mean percentage in patients with SS and MFI 10 times higher than the mean MFI of CD39 or CD73 in HDs) (Figure 1B and C). In these patients, high CD39 or CD73 expression was also present in skin-infiltrating lymphocytes (Figure 1D), indicating that the expression of CD39 and CD73 is an intrinsic feature of each patient with SS. The genetic control of CD39 expression in T cells by a single-nucleotide polymorphism in the ENTPD1 (CD39) gene10,11 supports this assumption.
Heightened expression of CD39 and CD73 in T cells from patients with Sézary syndrome. (A) Histograms show the expression of CD39 and CD73 in CD26– and CD26+ CD4+ T cells from the peripheral blood of patients with Sézary syndrome (SS, n = 11) and healthy donors (HDs, n = 28) defined by flow cytometry immunophenotyping at the time of inclusion in this study and at each subsequent follow-up (fu), indicated by progressive numbers. Red-colored fu∗ indicates CD39 and CD73 expression 2 months after the first treatment with mogamulizumab. The number below each histogram is the percentage of CD26-negative (top) or CD26-positive (bottom) CD4+ T cells at the time of inclusion in this study and at each subsequent follow-up. The number below the HD histograms is the mean ± SD of the percentage of CD26-negative (top) or CD26-positive (bottom) CD4+ T cells in healthy donors. (B and C) Expression of CD39 (B) and CD73 (C) in CD4+ T cells from patients with SS and from HDs. The percentage of positive cells (left y-axis) and the median fluorescence intensity (MFI, right y-axis) of CD39 (B) and CD73 (C) in CD4+ T cells from patients with SS and from HDs are shown. Dashed lines joint the percentage of CD39+ or CD73+ cells with the corresponding MFI in CD4+ T cells of each patient. Each dot represents a single subject. The horizontal line indicates the mean percentage of positive cells in patients with SS (percentage of CD39+ cells, mean ± SD = 60.1 ± 38.64; percentage of CD73+ cells, mean ± SD = 27.76 ± 30.97). An MFI value 10 times higher than the mean MFI of HDs (MFI of CD39, mean ± SD = 85.01 ± 73.47; MFI of CD73, mean ± SD = 97.96 ± 51.9) was chosen as the cutoff to discriminate patients with high or low CD39 or CD73 expression. A two-tailed Mann-Whitney t test was used to compare both expression and MFI of CD39 and CD73 between patients with SS and HDs. (D) Immunohistochemical staining of skin biopsies from patients with SS. Shown are representative skin sections from patients with high CD39 expression in circulating CD4+ T cells (top) and from patients with high CD73 expression in circulating CD4+ T cells (bottom). CD39 staining was mainly cytoplasmic and membranous, whereas CD73 staining showed diffuse membranous expression with a focal dotlike pattern. Hematoxylin and eosin (H&E) and CD4 staining highlight lymphocytes infiltrating the skin lesions. Original magnifications: ×10 (H&E); ×10 (CD4); ×40 (CD39 and CD73). APC, allophycocyanin; DN, double-negative cells; DP, double-positive cells.
Heightened expression of CD39 and CD73 in T cells from patients with Sézary syndrome. (A) Histograms show the expression of CD39 and CD73 in CD26– and CD26+ CD4+ T cells from the peripheral blood of patients with Sézary syndrome (SS, n = 11) and healthy donors (HDs, n = 28) defined by flow cytometry immunophenotyping at the time of inclusion in this study and at each subsequent follow-up (fu), indicated by progressive numbers. Red-colored fu∗ indicates CD39 and CD73 expression 2 months after the first treatment with mogamulizumab. The number below each histogram is the percentage of CD26-negative (top) or CD26-positive (bottom) CD4+ T cells at the time of inclusion in this study and at each subsequent follow-up. The number below the HD histograms is the mean ± SD of the percentage of CD26-negative (top) or CD26-positive (bottom) CD4+ T cells in healthy donors. (B and C) Expression of CD39 (B) and CD73 (C) in CD4+ T cells from patients with SS and from HDs. The percentage of positive cells (left y-axis) and the median fluorescence intensity (MFI, right y-axis) of CD39 (B) and CD73 (C) in CD4+ T cells from patients with SS and from HDs are shown. Dashed lines joint the percentage of CD39+ or CD73+ cells with the corresponding MFI in CD4+ T cells of each patient. Each dot represents a single subject. The horizontal line indicates the mean percentage of positive cells in patients with SS (percentage of CD39+ cells, mean ± SD = 60.1 ± 38.64; percentage of CD73+ cells, mean ± SD = 27.76 ± 30.97). An MFI value 10 times higher than the mean MFI of HDs (MFI of CD39, mean ± SD = 85.01 ± 73.47; MFI of CD73, mean ± SD = 97.96 ± 51.9) was chosen as the cutoff to discriminate patients with high or low CD39 or CD73 expression. A two-tailed Mann-Whitney t test was used to compare both expression and MFI of CD39 and CD73 between patients with SS and HDs. (D) Immunohistochemical staining of skin biopsies from patients with SS. Shown are representative skin sections from patients with high CD39 expression in circulating CD4+ T cells (top) and from patients with high CD73 expression in circulating CD4+ T cells (bottom). CD39 staining was mainly cytoplasmic and membranous, whereas CD73 staining showed diffuse membranous expression with a focal dotlike pattern. Hematoxylin and eosin (H&E) and CD4 staining highlight lymphocytes infiltrating the skin lesions. Original magnifications: ×10 (H&E); ×10 (CD4); ×40 (CD39 and CD73). APC, allophycocyanin; DN, double-negative cells; DP, double-positive cells.
Analysis of CD39 and CD73 in the first available skin biopsy from each patient confirmed that high expression of CD39 and/or CD73 in the infiltrating lymphocytes was already appreciable at diagnosis (namely, patients SS10, SS09, SS07, SS11, and SS04) (supplemental Table 3), ruling out possible therapy-mediated effects.
As CD39 expression is increased on T-cell receptor stimulation,12 we monitored the expression of CD39 and CD73 in T cells stimulated via CD3/CD28 at intervals up to 7 days. In CD4+ SS cells, the frequency of CD39+ cells was unmodified, regardless of baseline levels, whereas CD39 density slightly increased over time. Both CD39+ cell frequency and CD39 density were augmented in HDs (supplemental Figure 1A). CD73 expression was unaltered on activation both in patients with SS and in HDs (supplemental Figure 1B). In CD8+ cells, CD39 expression increased both in patients with SS and in HDs, as expected. CD8+CD73+ cells showed low frequency at baseline in patients with SS but increased on activation, whereas it decreased in HDs12 (supplemental Figure 1C and D). T-cell activation was confirmed by enhanced expression of CD25 and CD38 both in HDs and in patients with SS even if with considerable interindividual variability, especially in CD4+ SS cells13 (supplemental Figure 1E-H). These findings underscore that CD39 or CD73 expression in CD4+ T cells represents an immunophenotypic signature of each patient with SS, not influenced by cell activation.
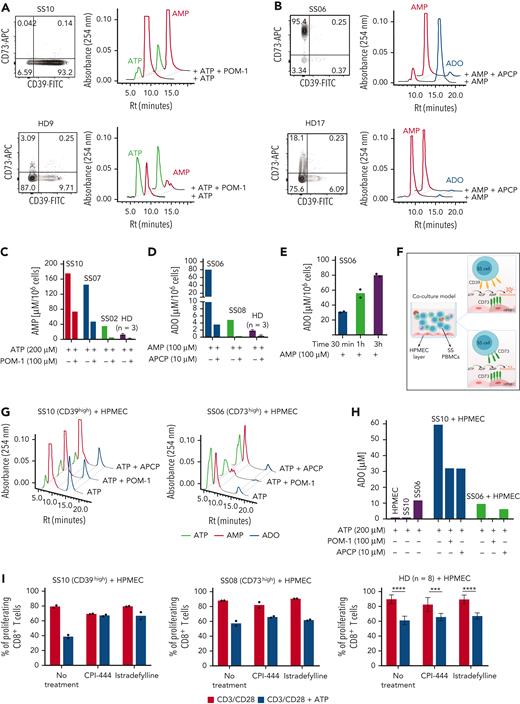
To assess the ectonucleotidase activity of CD39+ and CD73+ circulating SS lymphocytes, CD4+ T cells isolated from patients with SS and HDs by immune-magnetic negative selection were exposed to adenosine triphosphate (ATP) or adenosine monophosphate (AMP), and nucleotide consumption and ADO production were measured in the supernatant by a high-performance liquid chromatography–based assay.14 CD4+CD39+ SS cells hydrolyzed (and almost exhausted) ATP with generation of higher amounts of AMP, compared with HD cells. The amount of AMP was consistent with the expression levels of CD39 and was reduced by the CD39-specific inhibitor sodium polyoxotungstate, proving CD39-mediated ATPase activity (Figure 2A and C). CD4+CD73+ SS cells were able to metabolize AMP, generating large amounts of ADO; in contrast, CD4+ cells from HDs generated trace amounts of ADO, in agreement with their low CD73 expression (Figure 2B). ADO concentration increased with exposure time to AMP (Figure 2D), was strictly related to CD73 expression levels, and was blocked by the CD73-specific inhibitor adenosine 5′-(α,β-methylene)diphosphate (Figure 2E), confirming that the CD73+ SS cells exert AMPase activity.
Ectonucleotidase activity of CD39+ and CD73+ T cells from patients with SS, and functional implications of CD39high or CD73high T cells. (A and B) Flow cytometry dot plots showing CD73 and CD39 expression in CD4+ T cells from a representative CD39high patient with SS (A), a representative CD73high patient with SS (B), and 2 different HDs. Panels on the right show high-performance liquid chromatography (HPLC) profile of the peaks of extracellular ATP and AMP (A) or AMP and ADO (B) generated by CD4+ T cells on ATP or AMP supply, respectively. Cells were treated with or without 100 μM sodium polyoxotungstate (POM-1, a CD39 inhibitor) or 10 μM adenosine 5′-(α,β-methylene)diphosphate (APCP, a CD73 inhibitor) for 1 hour, before incubation for 3 hours with 200 μM ATP or 100 μM AMP, as indicated. (C) Quantification of AMP concentrations in the supernatant of CD4+ T cells from 3 patients with SS with different levels of CD39 expression and from HDs (n = 3), under the indicated experimental conditions. (D) Quantification of ADO concentration in the supernatant of CD4+ T cells from 2 patients with SS with different levels of CD73 expression and from HDs (n = 3) under the indicated experimental conditions. Results are expressed as micromoles of AMP per 106 cells or of ADO per 106 cells. In (C) and (D), data are expressed as mean ± SD. (E) Time-dependent increase in ADO concentration in the supernatant of CD4+ T cells from patient SS06 (CD73+). Histograms indicate ADO concentrations on AMP supply for 30 minutes, 1 hour, and 3 hours. Data are shown as the mean of technical replicates. (F) Schematic representation of a coculture system comprising human pulmonary microvascular endothelial cells (HPMECs) and peripheral blood mononuclear cells (PBMCs) used as an in vitro model to evaluate the degradation of ATP to ADO by CD39 and CD73 expressed by different cell types in the system. (G) HPLC profiles showing ATP, AMP, and ADO peaks on ATP addition. CD4+ T cells from a representative CD39high patient with SS (top) or from a representative CD73high patient with SS (bottom) were seeded onto HPMEC monolayers and exposed to extracellular ATP (200 μM) for 3 hours in the presence or absence of the indicated doses of POM-1 or APCP inhibitor. (H) Quantification of ADO concentration in the coculture system under the indicated experimental conditions. Results from 1 CD39high patient (SS10) and 1 CD73high patient (SS06) are shown. (I) CFSE-labeled PBMCs were activated for 5 days with CD3/CD28 antibodies in the presence of 500 μM ATP alone or combined with CPI-444 (10 μM) or istradefylline (0.1 μM). After immunolabeling, proliferation of CD8+ T cells was determined by flow cytometry. Data are shown for 1 representative CD39high patient with SS of 3 analyzed (mean of technical replicates), 1 representative CD73high patient with SS of 2 analyzed (mean of technical replicates), and for HDs (n = 8; mean ± SD). A two-way analysis of variance with the Šidák multiple comparisons test was done to compare untreated with ATP-treated cells under the indicated conditions in HDs: ∗∗∗∗P < .0001; ∗∗∗P < .001. CFSE, carboxyfluorescein diacetate succinimidyl ester; FITC, fluorescein isothiocyanate; Rt, retention time.
Ectonucleotidase activity of CD39+ and CD73+ T cells from patients with SS, and functional implications of CD39high or CD73high T cells. (A and B) Flow cytometry dot plots showing CD73 and CD39 expression in CD4+ T cells from a representative CD39high patient with SS (A), a representative CD73high patient with SS (B), and 2 different HDs. Panels on the right show high-performance liquid chromatography (HPLC) profile of the peaks of extracellular ATP and AMP (A) or AMP and ADO (B) generated by CD4+ T cells on ATP or AMP supply, respectively. Cells were treated with or without 100 μM sodium polyoxotungstate (POM-1, a CD39 inhibitor) or 10 μM adenosine 5′-(α,β-methylene)diphosphate (APCP, a CD73 inhibitor) for 1 hour, before incubation for 3 hours with 200 μM ATP or 100 μM AMP, as indicated. (C) Quantification of AMP concentrations in the supernatant of CD4+ T cells from 3 patients with SS with different levels of CD39 expression and from HDs (n = 3), under the indicated experimental conditions. (D) Quantification of ADO concentration in the supernatant of CD4+ T cells from 2 patients with SS with different levels of CD73 expression and from HDs (n = 3) under the indicated experimental conditions. Results are expressed as micromoles of AMP per 106 cells or of ADO per 106 cells. In (C) and (D), data are expressed as mean ± SD. (E) Time-dependent increase in ADO concentration in the supernatant of CD4+ T cells from patient SS06 (CD73+). Histograms indicate ADO concentrations on AMP supply for 30 minutes, 1 hour, and 3 hours. Data are shown as the mean of technical replicates. (F) Schematic representation of a coculture system comprising human pulmonary microvascular endothelial cells (HPMECs) and peripheral blood mononuclear cells (PBMCs) used as an in vitro model to evaluate the degradation of ATP to ADO by CD39 and CD73 expressed by different cell types in the system. (G) HPLC profiles showing ATP, AMP, and ADO peaks on ATP addition. CD4+ T cells from a representative CD39high patient with SS (top) or from a representative CD73high patient with SS (bottom) were seeded onto HPMEC monolayers and exposed to extracellular ATP (200 μM) for 3 hours in the presence or absence of the indicated doses of POM-1 or APCP inhibitor. (H) Quantification of ADO concentration in the coculture system under the indicated experimental conditions. Results from 1 CD39high patient (SS10) and 1 CD73high patient (SS06) are shown. (I) CFSE-labeled PBMCs were activated for 5 days with CD3/CD28 antibodies in the presence of 500 μM ATP alone or combined with CPI-444 (10 μM) or istradefylline (0.1 μM). After immunolabeling, proliferation of CD8+ T cells was determined by flow cytometry. Data are shown for 1 representative CD39high patient with SS of 3 analyzed (mean of technical replicates), 1 representative CD73high patient with SS of 2 analyzed (mean of technical replicates), and for HDs (n = 8; mean ± SD). A two-way analysis of variance with the Šidák multiple comparisons test was done to compare untreated with ATP-treated cells under the indicated conditions in HDs: ∗∗∗∗P < .0001; ∗∗∗P < .001. CFSE, carboxyfluorescein diacetate succinimidyl ester; FITC, fluorescein isothiocyanate; Rt, retention time.
To simulate the scenario occurring in vivo, peripheral blood mononuclear cells from CD39high (SS10) or CD73high (SS06) patients were seeded onto microvascular endothelial cells with ATP (Figure 2F). Neither human pulmonary microvascular endothelial cells (HPMECs) nor CD39high SS10 cells were able to degrade ATP to ADO (Figure 2H), whereas HPMECs hydrolyzed AMP, producing ADO (supplemental Figure 2A and B). However, when CD39high SS10 cells and HPMECs were cocultured, large amounts of ADO were produced on ATP addition. The amount of ADO was reduced by both sodium polyoxotungstate and 5′-(α,β-methylene)diphosphate. Notably, CD73high SS06 cells produced small amounts of ADO from ATP (not increased by contact with HPMECs), likely due to CD39 expressed in peripheral blood mononuclear cell subpopulations (Figure 2G and H). Hence, the interaction between CD39high SS cells and endothelial cells leads to overproduction of ADO; in contrast, the interaction of CD73high SS cells and endothelial cells does not. The higher levels of AMP in plasma from CD39+ patients with SS compared with both CD73+ patients with SS and HDs are consistent with this hypothesis (supplemental Figure 2C). Overall, this finding hints at a different local “adenosine halo” surrounding immune cells in CD39high compared with CD73high patients with SS, potentially shaping immunosuppression in the TME in CD39high but not in CD73high patients with SS.
To further substantiate this assumption, T-cell proliferation assays were performed in the presence of HPMECs with or without 500 μM ATP (simulating the ATP concentration in the TME15-17), and proliferation of CD8+ T cells was evaluated because proliferation of CD4+ T cells (mostly consisting of the tumor clone) is severely impaired in vitro.18 ATP decreased proliferation of both CD8+ SS and HD T cells. Strikingly, CPI-444 and istradefylline (antagonist inhibitors of ADORA2A [encoding adenosine receptor subtype A2a, or A2aR], the most important ADO receptor in regulating lymphocyte activation19) almost completely prevented ATP-mediated suppression of CD8+ cells from CD39high patients but only marginally influenced proliferation of CD8+ cells from CD73high patients, much in the same way as in HDs (Figure 2I). Hence, conversion of ATP into ADO by CD39/CD73 and ADO binding to A2aR occurring in the TME of CD39high patients with SS contribute to immunosuppression. In the TME of CD73high patients with SS, ADO concentration is low and minimally contributes to ATP-mediated immunosuppression, which apparently relies on a different mechanism, as well as in HDs. One could speculate that an alternative ATP degradation product other than ADO20 or a pathway other than A2aR signaling21 could prevail in suppressing proliferation of CD8+ T cells from CD73high patients with SS and HDs. This is a key aspect that deserves further investigation.
In conclusion, our study demonstrates that aberrant expression of CD39 or CD73 in circulating and skin-homing CD4+ T cells is a hallmark of each patient with SS, from diagnosis, that persists during disease progression and has important functional implications. Indeed, in vivo, CD39+ patients show significantly higher plasma levels of AMP than do CD73+ patients. In CD39+ patients, the CD39/CD73/adenosine axis promotes immunosuppression in vitro. These findings support the view that high CD39 expression in SS cells, combined with loss of CD26 (a docking site for adenosine deaminase that inactivates adenosine, preventing its binding to A2aR on immune cells22) and with abundant expression of CD73 by several cell types, including endothelial cells (especially dermal lymphatic endothelial cells23), provides the optimal milieu to sustain high concentrations of adenosine causing immunosuppression. The potential double effect of the CD39/CD73/adenosine axis, which on the one hand suppresses normal T-cell function and on the other hand slows down SS-cell proliferation, as recently suggested,10 is a critical issue that deserves further investigation, also considering the heterogeneity of the disease.24
Overall, these results, obtained on a relatively small group of patients, suggest that the inhibition of CD39 and/or A2aR combined with other immune-mediated therapies might mitigate immunosuppression in patients with SS with high CD39 expression. However, the existence of a correlation between CD39 expression and survival of patients with SS10 questions the benefit of CD39-targeted therapies and requires careful evaluation of the clinical significance of CD39 in a large number of patients undergoing homogeneous therapies, before definitive conclusions can be drawn.
Acknowledgments
The authors thank the patients and the healthy volunteers who participated in this study and E. Ferrero (University of Turin, Turin, Italy) for helpful discussion and critical review of the manuscript.
This work was supported by a Cutaneous Lymphoma Catalyst Research Grant contract (P.Q.). Y.Y. is the recipient of a fellowship from the Cutaneous Lymphoma Foundation.
Authorship
Contribution: Y.Y. and S.M. performed experiments and analyzed data; E.O. designed the study, performed experiments, and analyzed data; C.B. designed and performed HPLC experiments; P.Q., S.R., and M.F. provided patient samples and relevant clinical information; R.S. and M.R.R. provided skin sections, analyzed immunohistochemical data, and contributed to data interpretation; C.T. performed the immunohistochemical staining; A.F. and P.Q. designed the study, analyzed and interpreted data, and wrote the manuscript; and all authors read and approved the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Ada Funaro, Laboratory of Immunogenetics, Department of Medical Sciences, University of Turin, Via Santena 19, 10126 Turin, Italy; e-mail: ada.funaro@unito.it; and Pietro Quaglino, Dermatologic Clinic, Department of Medical Sciences, University of Turin, Via Cherasco 23, 10121 Turin, Italy; e-mail: pietro.quaglino@unito.it.
References
Author notes
For original data, please contact Ada Funaro or Pietro Quaglino via email at: ada.funaro@unito.it, pietro.quaglino@unito.it.
The online version of this article contains a data supplement.
There is a Blood Commentary on this article in this issue.
Y.Y. and S.M. contributed equally to this study and are joint first authors.
A.F. and P.Q. are joint senior authors.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal