Abstract
Introduction: Chimeric antigen receptor (CAR) T-cells have provided a therapeutic option for patients with relapsed or refractory (R/R) B-cell acute lymphoblastic leukemia (B-ALL) who have failed standard chemotherapy approaches. Despite initial responses, relapse rates after tisagenlecleucel approach 40% for pediatric patients necessitating a need to improve outcomes with this therapy. High pre-infusion disease burden (defined as ≥5% blasts on marrow or extramedullary disease) has been associated with inferior overall survival (OS) and event free survival (EFS) (Schultz 2021). However, it remains unclear if debulking disease will improve outcomes. Additionally, bridging chemotherapy used to debulk may introduce toxicity, without established benefit. Peripheral blast count represents high burden disease in the bone marrow. No current clinical standard exists guiding peripheral blast thresholds prior to apheresis. It remains unclear if having peripheral blasts at the time of apheresis is a surrogate marker for disease burden, or if peripheral blasts at time of apheresis collection has inherent independent predictive value.
Methods: We performed a retrospective analysis from the Pediatric Real-World CAR Consortium (PRWCC) consisting of 15 US institutions. All patients with data available regarding their peripheral blast count at time of apheresis were included. Primary outcomes were day 28 response, EFS (events; second malignancy, relapse, or death) and OS. Baseline factors of interest included age at diagnosis, sex, pre-infusion disease burden, prior lines of therapy, prior CD19 therapy, time from diagnosis to CAR infusion, CAR dose, prior stem cell transplantation (SCT), and timing of 1st relapse post SCT (Schultz 2021). The impact of peripheral blast count at apheresis was assessed using Cox models adjusting for all baseline factors of interest. Kaplan Meier (KM) curves were reported for time-to-death and time-to-first event.
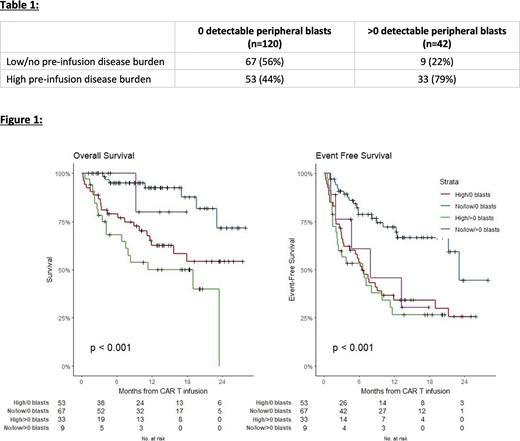
Results: Of 185 patients who were infused with tisagenlecleucel in the PRWCC database, 162 patients had peripheral blast data at time of apheresis available for analysis: 120 (74%) had no peripheral blasts on CBC and 42 (26%) had >0 blasts at apheresis. A greater proportion of patients with >0 blasts had high disease burden (79%) compared to those with no peripheral blasts (44%) (table 1). The median (interquartile range, IQR) from apheresis to CAR T infusion is 7 weeks (wks) (5.2-10 wks) and there was no difference in time to infusion for patients with no blasts (n=120): 7 wks (6-10) compared to those with >0 blasts (n=42): 6 wks (5-8.8). Day 28 response did not differ between those with no blasts (87% CR) and those with >0 blasts (83% CR) (p=0.83). With a median follow-up of 334 (143-536) days (>0 peripheral blasts; 248 (92, 520), no blasts; 353 (186, 549)), death occurred more often in patients with >0 blasts (43%) when compared to those with no blasts (22%). Patients who had >0 blasts had significantly inferior event-free survival (p=0.008) and overall survival (p=0.004) compared to those with no peripheral blasts at apheresis. Univariate analysis showed that for every 10% increase in blast percentage, the hazard of death increased 1.19-fold (95% confidence interval (CI) 1.05-1.35;p=0.006) and the hazard of events increased 1.10-fold (95% CI 0.99-1.23; p=0.077). High pre-infusion disease burden had a significantly higher hazard of death (p=0.002) and of event (p=0.002), compared to no detectable disease. After adjustment for baseline factors including high vs no vs low disease burden, multivariable analysis demonstrated higher blasts remained significantly associated with higher hazard of death (p=0.004) and event (p=0.04). KM curves were stratified by pre-infusion disease burden (high versus low/no) and peripheral blast count at apheresis (no blasts versus >0 blasts) (figure 1). Patients with low/no disease burden and no blasts at apheresis had the best EFS when compared to those with high disease burden (p<0.001). Although not statistically significantly, amongst patients with high pre-infusion disease burden, those with >0 blasts had slightly worse OS (p=0.13) and EFS (p=0.47) compared to those with 0 blasts.
Conclusion: Higher peripheral blasts at apheresis associates with inferior outcomes after tisagenlecleucel, even after multivariable analysis. Further study is needed to determine the mechanism of this association and if it is modifiable.
Disclosures
Fabrizio:Adaptimmune: Consultancy. Phillips:Novartis: Membership on an entity's Board of Directors or advisory committees; Incyte: Membership on an entity's Board of Directors or advisory committees. Stefanski:Novartis: Other: Ad Board, Speakers Bureau. Myers:Novartis: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Eliana Trial: Consultancy, Membership on an entity's Board of Directors or advisory committees. Brown:Bristol Myers Squibb: Current Employment. Qayed:Novartis: Honoraria; Vertex: Honoraria. Hermiston:Sobi: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees. Satwani:Takeda: Honoraria; Mesoblast: Honoraria. Curran:Novartis: Consultancy, Research Funding. Mackall:Medimmune Tech: Consultancy; BMS: Consultancy; Apricity: Consultancy, Divested equity in a private or publicly-traded company in the past 24 months; Nektar: Consultancy; Ensoma: Divested equity in a private or publicly-traded company in the past 24 months; Syncopation: Consultancy, Divested equity in a private or publicly-traded company in the past 24 months; Link: Consultancy, Divested equity in a private or publicly-traded company in the past 24 months; Mammoth: Divested equity in a private or publicly-traded company in the past 24 months; Immatics: Consultancy; GSK: Consultancy; Lyell Pharmaceuticals: Consultancy, Divested equity in a private or publicly-traded company in the past 24 months. Laetsch:Pfizer: Research Funding; Cellectis: Consultancy; Novartis: Consultancy, Research Funding; Bayer: Consultancy, Research Funding; Advanced Microbubbles: Current holder of stock options in a privately-held company; AI Therapeutics: Consultancy; Deciphera: Consultancy; GentiBio: Consultancy; Jazz Pharmaceuticals: Consultancy; Jumo Health: Consultancy; Massive Bio: Consultancy; Menarini: Consultancy; Pyramid Biosciences: Consultancy; y-mAbs Therapeutics: Consultancy. Verneris:Bmogen: Consultancy, Current equity holder in private company; Up-To-Date: Consultancy; Fate Therapeutics: Consultancy, Current equity holder in private company; Novartis: Membership on an entity's Board of Directors or advisory committees. Schultz:Novartis: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal