Abstract
Introduction
Secondary MDS (sMDS) is defined by MDS arising from another bone marrow disorder or related to previous therapy [chemotherapy (CT), radiation, or immunosuppression], and comprises about 10% of MDS cases. The rate of sMDS following CLL (including treatment-naïve CLL) has been reported as high as 3.5%. Developing in elderly populations, both CLL and MDS are associated with clonal hematopoiesis (CH), and chemoimmunotherapy (CIT) treatment for CLL likely contributes to the occurrence of MDS. There is little published data on the characteristics of patients (pts) who develop sMDS with antecedent/concurrent CLL, particularly in the era where kinase inhibitor therapy has displaced CIT. Therefore, we sought to characterize a cohort of CLL pts who develop sMDS in the era of modern treatments.
Methods
We performed a retrospective chart review at The Ohio State University between 01/01/2016 and 02/10/2021 of pts with a diagnosis of CLL who subsequently or concurrently were diagnosed with MDS. Clinical, next generation sequencing (NGS), and outcome data were collected. NGS test refers to a 50 gene hematologic neoplasm mutation panel. Overall survival (OS) was calculated from MDS diagnosis and estimated using Kaplan-Meier method.
Results
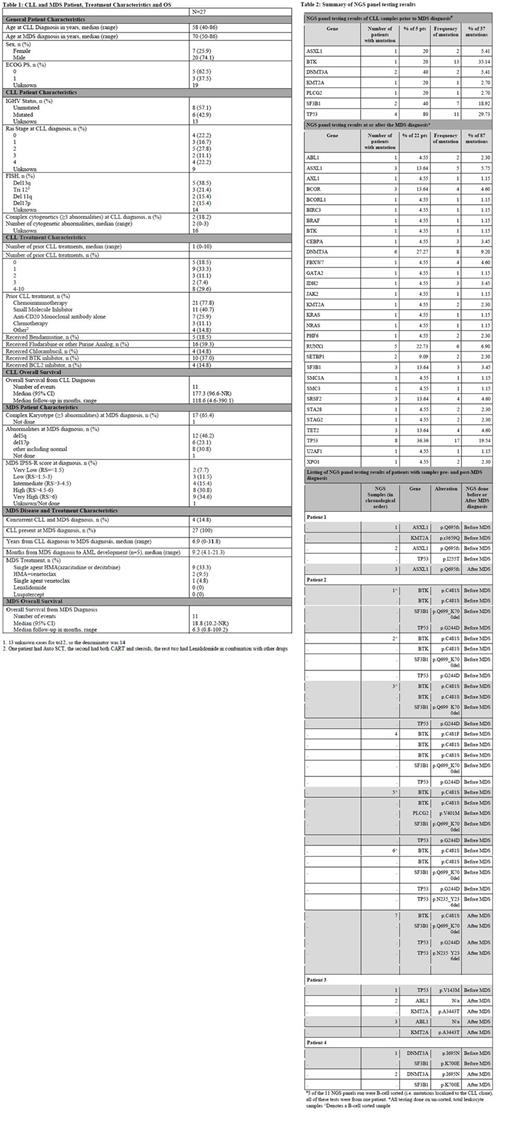
We identified 27 sMDS pts with antecedent/concurrent CLL. At CLL diagnosis, median age was 58 years (range 40-86). 5 pts (18.5%) had a TP53 mutation and/or del(17p) noted during their CLL disease course prior to sMDS diagnosis. Median number of therapies for CLL prior to sMDS was 1 (range 0-10), with 4 pts diagnosed with CLL at the same time as MDS. CLL treatments, as any line of therapy, included CT/CIT in 21 pts (77.8%) and ibrutinib in 10 pts (37.0%). Of the pts that received CLL therapy, only 1 pt did not receive CT/CIT. 5 pts (18.5%) were not treated for CLL. A median of 6.9 years (range 0-31.8) elapsed between the diagnosis of CLL and sMDS. At sMDS diagnosis median age was 70 years (range 50-86), 17 pts (65.4%) were MDS IPSS-R category high/very high. 5 pts transformed to AML after a median of 9.2 months (mos) from MDS diagnosis (range 4.1-21.3). After a median follow-up of 6.3 mos (range 0.8-109.2), the median OS from MDS diagnosis was 18.8 mos (95% CI, 10.2-not reached). (Table 1)
In pts who received CT/CIT for CLL (n=21) and those who did not (n=6), a TP53 mutation developed following CLL or MDS diagnosis in 10 pts (47.6%) and 1 pt, respectively. All but 1 pt who developed TP53 mutations (n=11) received prior CT. 15 pts (71.4%) who received CT/CIT for CLL therapy had MDS with complex karyotype; this was seen in 2 pts who never received CT/CIT.
Twenty-six (96.3%) pts had NGS testing. 5 pts (18.5%) had 1 or more tests completed for CLL prior to sMDS: 4 pts had TP53 mutations, 2 pts each had DNMT3A and SF3B1 mutations, and 1 pt each had an ASXL1, KMT2A, BTK, or PLCG2 mutation. Of these 5 pts, 1 pt had 5 NGS tests performed on serial B-cell selected samples (as opposed to total leukocytes): mutations included 2 TP53, 1 SF3B1 and 1 BTK mutation. 25 (92.6%) pts had 1 or more tests completed at or after sMDS diagnosis (all assessed total leukocytes): 8 pts (36.4%) had TP53, 6 pts (27.3%) had DNMT3A, 5 pts (22.7%) had RUNX1, and 3 pts (13.6%) each had ASXL1, SF3B1, and TET2 mutations. Finally, 4 pts (14.8%) had NGS testing done both pre and post sMDS diagnosis (all assessed total leukocytes). Two pts had the same mutation present pre and post sMDS diagnosis: 1 ASXL1, 1 DNMT3A. A third pt had 3 identical mutations pre and post sMDS diagnosis: 1 TP53 p.G244D, 1 TP53 p.N235_Y236del, and 1 SF3B1. (Table 2)
Conclusions
Here we have characterized a cohort of CLL pts who developed sMDS in the modern era, where small molecule inhibitors are commonly used for CLL, and NGS is widely available for both CLL and MDS. While the majority of pts with CLL received CT, 5 pts were treatment-naive, and 1 pt only received a BTK inhibitor and an anti-CD20 antibody. The OS of this cohort following MDS diagnosis is similar to historical data of pts with comparable IPSS-R scores, despite the fact that this is a sMDS population; this may be due to lead time bias as pts likely received frequent blood work for CLL. Prior to sMDS diagnosis, complex karyotype and TP53, SF3B1 and CH-associated mutations DNMT3A and ASXL1 were prevalent. TP53, SF3B1, and CH-associated mutations DNMT3A, ASXL1, and TET2 mutations were prevalent at the time of or following a sMDS diagnosis. Further investigation into the interplay between CLL and CH in MDS development in the modern therapy era is needed.
Disclosures
Koenig:Hairy Cell Leukemia Foundation: Research Funding. Huang:AstraZeneca: Other: Statistical support. Rogers:Innate Pharma: Consultancy; Beigene: Consultancy; Pharmacyclics: Consultancy; AstraZeneca: Consultancy, Other: Travel Funding; Novartis: Research Funding; Janssen: Research Funding; AbbVie: Consultancy, Research Funding; Genentech: Consultancy, Research Funding. Woyach:Pharmacyclics: Consultancy; Genentech: Consultancy; Schrodinger: Research Funding; Loxo@Lilly: Research Funding; AbbVie: Consultancy, Research Funding; MorphoSys: Consultancy, Research Funding; Newave: Consultancy; Janssen: Consultancy; ArQule: Consultancy; AstraZeneca: Consultancy; BeiGene: Consultancy; Karyopharm Therapeutics: Research Funding. Grever:Hairy Cell Leukemia Foundation: Membership on an entity's Board of Directors or advisory committees, Research Funding; Innate Pharma: Consultancy; Serono: Consultancy; Axio: Consultancy; Ascerta: Consultancy; AstraZeneca: Consultancy; Pharmacyclics: Consultancy. Borate:AbbVie/Genentech: Membership on an entity's Board of Directors or advisory committees; Daiichi Sankyo: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees. Mims:Genentech: Membership on an entity's Board of Directors or advisory committees; AbbVie: Membership on an entity's Board of Directors or advisory committees; Astellas: Membership on an entity's Board of Directors or advisory committees; Syndax: Membership on an entity's Board of Directors or advisory committees; Servier: Membership on an entity's Board of Directors or advisory committees; Ryvu: Membership on an entity's Board of Directors or advisory committees; Zentalis: Membership on an entity's Board of Directors or advisory committees; Jazz Pharmaceuticals: Other: Data Safety and Monitoring Board; Daiichi Sankyo: Other: Data Safety and Monitoring Board; BMS: Membership on an entity's Board of Directors or advisory committees. Blachly:MingSight Pharmaceuticals: Research Funding; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees; INNATE Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; KITE Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees. Kittai:Abbvie: Consultancy; Astrazeneca: Consultancy, Research Funding; Beigene: Consultancy; Janssen: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal