Abstract
Coronavirus disease-19 (COVID-19) includes a thromboinflammatory syndrome that may manifest with microvascular and macrovascular thrombosis. Patients with COVID-19 have a higher incidence of venous thromboembolism than other hospitalized patients. Three randomized control trials suggesting benefit of therapeutic heparin in hospitalized noncritically ill patients with COVID-19 have led to conditional guideline recommendations for this treatment. By contrast, prophylactic-dose heparin is recommended for critically ill patients. Unprecedented collaboration and rapidly funded research have improved care of hospitalized patients with COVID-19.
Introduction
The coronavirus disease-19 (COVID-19) pandemic has caused 568 million cases and 6.3 million deaths.1 In the United States, during the Omicron variant wave peak, the daily hospitalized patients with COVID-19 ranged from 84 000 to 152 000.2
It is well known that medical hospitalization increases the risk of venous thromboembolism (VTE), and the American Society of Hematology (ASH) guidelines for VTE prophylaxis discuss pharmacologic and mechanical prophylaxis.3 From early in the COVID-19 pandemic, frequent VTE was recognized.4 A procoagulant biomarker profile, including elevated D-dimer, predicted VTE and mortality.5
Estimates of the occurrence of VTE during hospitalization for COVID-19 depend on several factors, including critical illness and use of screening imaging, which is not generally recommended in medical inpatients. In a systematic literature search and meta-analysis of 66 studies involving 28 173 admitted patients, the overall VTE prevalence was 14.1%.6 Substantially higher rates were reported when screening imaging was used; 40.3% in those screened and 9.5% in those not screened. When patients required an intensive care unit (ICU) level of care, the prevalence was nearly 3 times greater than in ward patients (22.7% vs 7.9%). Importantly, the VTE rate decreased over time across studies. This could reflect reporting bias early in the pandemic, changes in VTE prophylaxis practices or testing frequency, or improved supportive care such as use of corticosteroids. Regardless, the preponderance of evidence suggests VTE is more common with COVID-19 hospitalization than the estimates of baseline VTE risk used in the ASH guideline on VTE prevention in medical patients (pulmonary embolism rate, 0.4%; proximal deep vein thrombosis rate, 0.5%; distal deep vein thrombosis rate, 1.4%).3 As such, clinical trials specific to patients with COVID-19 are needed to evaluate thrombosis prevention strategies.
Clinical deterioration in patients with COVID-19 involves endothelial injury, inflammation, and microvascular thrombosis, which lead to hypoxemic respiratory failure and other end organ damage.7,8 Autopsy studies have shown VTEs that were not recognized before death in addition to microthrombi in the capillaries of the lung, kidney, and brain.9,10 Therefore, antithrombotic therapy may prevent progression of COVID-19 coagulopathy and progression of disease severity.
Antithrombotic trials in COVID-19
Multiple randomized controlled trials evaluating the effectiveness and safety of anticoagulation to reduce disease progression in patients hospitalized for COVID-19 have been conducted over a short time. Methodologic differences in meta-analysis affected outcomes and subsequent clinical recommendations.
Noncritically ill patients
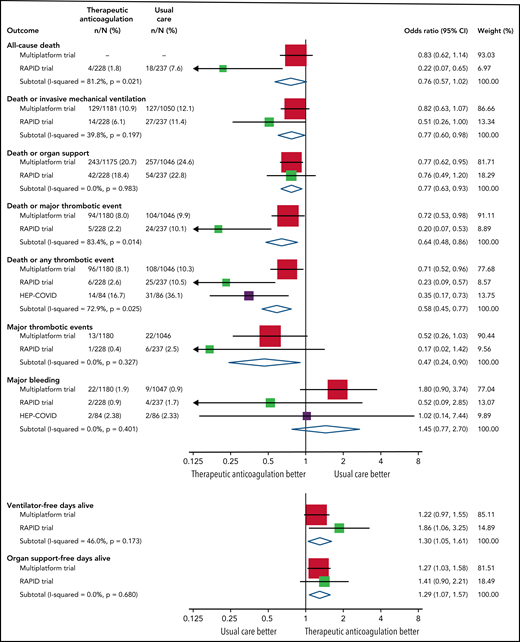
There are 5 published trials comparing therapeutic anticoagulation with heparin/low-molecular-weight heparin (LMWH; subsequently termed heparin) to usual care using lower doses in hospitalized patients with COVID-19. Three trials evaluated therapeutic-dose heparin in noncritically (moderately) ill ward patients: a multiplatform trial integrating the Antithrombotic Therapy to Ameliorate Complications of COVID-19 (ATTACC), Accelerating COVID-19 Therapeutic Interventions and Vaccines-4 Antithrombotics Inpatient Platform Trial (ACTIV-4a), and the Randomized, Embedded, Multifactorial Adaptive Platform Trial for Community-Acquired Pneumonia (REMAP-CAP); the Coagulopathy of COVID-1911; A Pragmatic Randomized Controlled Trial of Therapeutic Anticoagulation Versus Standard Care as a Rapid Response to the COVID-19 Pandemic (RAPID)12; and the Systemic Anticoagulation With Full Dose LMWH Versus Prophylactic or Intermediate Dose LMWH in High Risk COVID-19 Patients (HEP-COVID trial).13 The multiplatform trial showed that therapeutic heparin increased organ support-free days compared with usual care (odds ratio [OR], 1.29; 95% credible interval, 1.04-1.61), with 4.6% less patients who received therapeutic anticoagulation requiring organ support.11 Compared with prophylactic dose heparin, therapeutic heparin in the RAPID trial did not improve the primary outcome of ICU admission, noninvasive and invasive ventilation, and death at 28 days, but did reduce mortality (2% therapeutic vs 8% prophylactic; relative risk [RR], 0.22; 95% confidence interval [CI], 0.07-0.65).12 The effect size for organ support-free days was similar to the multiplatform trial (OR, 1.41; 95% CI, 0.90-2.21). The HEP-COVID trial recruited patients with D-dimer levels >4 times the upper limit of normal and showed that therapeutic heparin reduced VTE, arterial embolism, and death (17% therapeutic vs 36% usual care; RR, 0.46; 95% CI, 0.27-0.81).13 A meta-analysis of therapeutic heparin showed significant evidence of benefit in 5 important effectiveness outcomes (death or invasive mechanical ventilation, death or organ support, ventilator-free days alive, organ support–free days alive, and major thrombotic events; Figure 1).14 Critical care research often uses organ support–free days as an outcome.15,16 Not focusing on VTE in a trial of anticoagulation may have affected adoption of this treatment by clinicians.
Meta-analysis of randomized control trials of therapeutic heparin in hospitalized noncritically ill patients with COVID-19. Odds ratios of 3 trials11-13 were combined with Mantel-Haenszel fixed-effect meta-analyses. Treatment effects are represented by squares with area proportional to the trial weight with 95% confidence interval shown by horizontal lines. Diamonds show the meta-analyses treatment effect estimates. Reproduced with permission from Research and Practice in Thrombosis and Haemostasis.14
Meta-analysis of randomized control trials of therapeutic heparin in hospitalized noncritically ill patients with COVID-19. Odds ratios of 3 trials11-13 were combined with Mantel-Haenszel fixed-effect meta-analyses. Treatment effects are represented by squares with area proportional to the trial weight with 95% confidence interval shown by horizontal lines. Diamonds show the meta-analyses treatment effect estimates. Reproduced with permission from Research and Practice in Thrombosis and Haemostasis.14
Although randomized controlled trials (RCTs) show benefit of therapeutic heparin, differences in trial design affect interpretation of the results. The open-label trial design was pragmatic during the pandemic. Clinical suspicion of thrombosis might have differed between the treatment groups, but VTE was not the primary outcome. The trials were completed early in the pandemic, so it is possible that effectiveness of therapeutic heparin may be different because of vaccination, monoclonal antibody therapy, and viral variants. Additional limitations include use of screening imaging in the HEP-COVID trial and potential site differences such as availability of mechanical ventilation and criteria for ICU admission. Last, patient characteristics likely related to outcomes differed between the trials.
One trial, Therapeutic versus Prophylactic Anticoagulation for Patients Admitted to Hospital with COVID-19 and Elevated D-dimer Concentration (ACTION),17 evaluated therapeutic anticoagulation with rivaroxaban in noncritically ill patients and therapeutic enoxaparin in critically ill patients. Therapeutic rivaroxaban for 30 days did not improve the composite of mortality, duration of hospitalization, and duration of oxygen use (hierarchical analysis win ratio, 0.86; 95% CI, 0.59-1.22). Therapeutic rivaroxaban increased clinically relevant nonmajor bleeding but not major bleeding (3% rivaroxaban vs 1% usual care; RR, 2.45; 95% CI, 0.78-7.73).
Critically ill patients
In critically ill patients, 2 trials, Effect of Intermediate-Dose Versus Standard-Dose Prophylactic Anticoagulation on Thrombotic Events, Extracorporeal Membrane Oxygenation Treatment, or Mortality Among Patients With COVID-19 Admitted to the Intensive Care Unit (INSPIRATION)18 and Perepu et al19 used intermediate-dose heparin as their experimental arm. Three trials evaluated therapeutic-dose heparin in the critically ill: a multiplatform trial conducted by the same investigators in ICU patients,20 a phase 2 randomized trial of mechanically ventilated patients (Therapeutic Versus Prophylactic Anticoagulation for Severe COVID-19: A Randomized Phase II Clinical Trial [HESACOVID]),21 and HEP-COVID,13 which also included critically ill patients. In contrast to the findings in the noncritically ill, meta-analysis of trials of therapeutic heparin in critically ill patients showed no beneficial effects of therapeutic heparin, except a decrease in risk of major thrombotic events (OR, 0.59; 95% CI, 0.39-0.91).14
These consistent findings across trials evaluating therapeutic heparin suggest that treatment of hospitalized patients with less severe disease is important to change the course of thrombo-inflammatory–driven disease. Overall, the lack of benefit for mortality in both the noncritically and critically ill is in keeping with historical trials of therapeutic anticoagulation. Risk of major bleeding was low in all trials, whether conducted in the critically or noncritically ill, suggesting that the trials selected for patients at low risk of bleeding.
A meta-analysis that pooled anticoagulation trials of hospitalized patients regardless of critical illness, dose, or type of anticoagulation found no evidence for benefit. This led the authors to suggest prophylactic anticoagulation for all hospitalized patients with COVID-19, irrespective of disease severity.22 Given the contrast with the above results, this reminds us that anticoagulant drugs are not always interchangeable for a given indication or different illness severity. Completion of multiple ongoing studies will provide additional evidence about the balance of the risks and benefits of anticoagulation in hospitalized patients with COVID-19.
VTE after hospital discharge
After discharge for COVID-19, the rate of VTE is uncertain, with estimates in the low single digits over weeks.23 In a UK study, the rate was 4.8 per 1000 discharges over 6 weeks in 1877 patients.24 The authors compared this with medical discharges in 2019 before the pandemic, where the rate was 3.1 per 1000 discharges in 18 159 patients. They did not have power to conclude the rate with COVID-19 was higher and did not present analyses adjusted for illness severity. Several trials are ongoing assessing postdischarge VTE prophylaxis, with 1 completed. The open label Medically Ill hospitalized Patients for COVID-19 THrombosis Extended ProphyLaxis with rivaroxaban ThErapy (MICHELLE) trial selected patients at high predicted risk of postdischarge VTE based on the International Medical Prevention Registry on Venous Thromboembolism VTE score and D-dimer, who had low bleeding risk. They compared rivaroxaban 10 mg daily to placebo for 35 days after discharge. The composite primary outcome (symptomatic or asymptomatic screen-detected VTE, symptomatic arterial thromboembolism, or cardiovascular death at 35 days) was 3% with rivaroxaban and 9% with placebo (RR, 0.33; 95% CI, 0.12-0.90; P = .03 in favor of rivaroxaban).25 Most VTEs were asymptomatic; symptomatic and fatal VTEs occurred in 0.6% with rivaroxaban and 5.0% with placebo (RR, 0.13; 95% CI, 0.02-0.99; P = .049).
Guideline recommendations
Multiple national and international organizations published guidance statements and guideline recommendations early in the pandemic concerning anticoagulation in hospitalized patients with COVID-19. These were based on expert opinion, observational data, and clinical experience, which led to differences among these recommendations. Early recommendations by the International Society of Thrombosis and Haemostasis (ISTH) recommended routine thromboprophylaxis and consideration of intermediate-dose LMWH in high-risk patients.26 Guidance from the Anticoagulation Forum suggested increased intensity of heparin dosing in critically ill patients because of elevated risk of pulmonary embolism reported in early studies.27 Other panels, including the ASH,28 The American College of Chest Physicians (CHEST),29 National Institutes of Health (NIH) COVID-19 guideline panel, and World Health Organization,30 recommended prophylactic-dose anticoagulation in all hospitalized patients.
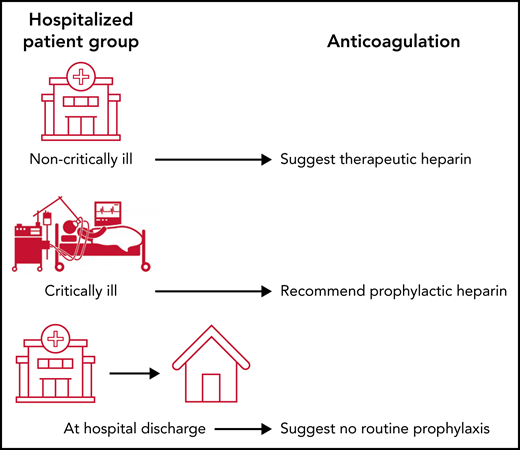
Guidelines changed after publication of randomized control trials. All guidelines now recommend prophylactic-dose anticoagulation for critically ill patients (Table 1). The results of the multiplatform,11 RAPID,12 and HEP-COVID13 trials changed the guidelines for hospitalized noncritically ill patients. The National Institutes of Health and Care Excellence (NICE) and NIH COVID-19 guidelines issued conditional recommendations to consider therapeutic-dose LMWH in hospitalized noncritically ill patients requiring low flow oxygen and without an increased bleeding risk (Table 1).31 The NIH also limited the recommendation to patients with elevated D-dimer level, because the inclusion criteria for the RAPID12 and HEP-COVID13 trials required this, and the treatment effects in the multiplatform trial11 were the largest in patients with elevated D-dimer levels. Both the ASH32 and CHEST33 guidelines used Grading of Recommendations Assessment, Development and Evaluation (GRADE) methodology and suggested therapeutic anticoagulation over prophylactic anticoagulation in hospitalized noncritically ill patients without narrowing the patient population based on oxygen needs or laboratory values. The conditional recommendations in the guidelines reflect the differences in inclusion criteria (all patients vs only those with elevated D-dimer level) and which outcomes were statistically significant in the randomized trials. The evidence to decision framework notes that the ASH recommendations were not unanimous reflecting debate within the community. The ISTH guidelines,34 currently available for public comment, gave a strong recommendation for therapeutic-dose anticoagulation in hospitalized noncritically ill patients at low risk of bleeding and with VTE risk factors or organ failure such as elevated D-dimer level or increased oxygen requirements. Updated meta-analyses and live integration into guidelines will be important as additional trials are completed.
Anticoagulation guidelines for anticoagulation in hospitalized patients with COVID-19
| . | Hospitalized noncritically ill patients . | Critically ill patients . | Postdischarge prophylaxis . |
|---|---|---|---|
| ASH32 | Suggest therapeutic-intensity over prophylactic-intensity anticoagulation (conditional recommendation, very low certainty) | Suggest prophylactic-intensity over intermediate-intensity anticoagulation (conditional recommendation, low certainty) | Suggests not using postdischarge prophylaxis (conditional recommendation, very low certainty) |
| CHEST29,33 | Suggest therapeutic dose heparin over standard dose anticoagulant thromboprophylaxis (conditional recommendation, ungraded consensus-based statement) | Suggest standard dose anticoagulant thromboprophylaxis over intermediate or therapeutic dose anticoagulation (conditional recommendation, ungraded consensus-based statement) | Recommend inpatient only over inpatient plus postdischarge prophylaxis |
| ISTH34 | Recommend therapeutic anticoagulation in select patients (I, A) | Intermediate or therapeutic dose anticoagulation not recommended over prophylactic-dose heparin (3, B-R) | In select patients, postdischarge prophylactic dose rivaroxaban may be considered (2b, B-R) |
| NICE31 | Consider treatment dose LMWH for young people and adults with COVID-19 who need low-flow oxygen and who do not have an increased bleeding risk (conditional recommendation) | Intermediate or treatment dose heparin offered only as part of a clinical trial (only in research settings) | In-hospital prophylaxis should continue for 7 d, including after discharge (recommendation) |
| NIH COVID-19 Guideline42 | Recommend therapeutic-dose heparin for patients who have a D-dimer above the upper limit of normal, require low-flow oxygen, and have no increased bleeding risk (CIIa) | Recommend prophylactic-dose heparin (AI). Recommends against the use of intermediate-dose (eg, enoxaparin 1 mg/kg daily) and therapeutic-dose anticoagulation for VTE prophylaxis, except in a clinical trial (BI) | Recommends against routinely continuing VTE prophylaxis after hospital discharge (AIII). For patients who are at high risk for VTE and at low risk of bleeding, extended VTE prophylaxis can be considered, as per the protocol for patients without COVID-19 (BI) |
| World Health Organization30 | Suggest standard thromboprophylaxis dosing* (conditional recommendation, very low certainty) | Suggest standard thromboprophylaxis dosing* (conditional recommendation, very low certainty) |
| . | Hospitalized noncritically ill patients . | Critically ill patients . | Postdischarge prophylaxis . |
|---|---|---|---|
| ASH32 | Suggest therapeutic-intensity over prophylactic-intensity anticoagulation (conditional recommendation, very low certainty) | Suggest prophylactic-intensity over intermediate-intensity anticoagulation (conditional recommendation, low certainty) | Suggests not using postdischarge prophylaxis (conditional recommendation, very low certainty) |
| CHEST29,33 | Suggest therapeutic dose heparin over standard dose anticoagulant thromboprophylaxis (conditional recommendation, ungraded consensus-based statement) | Suggest standard dose anticoagulant thromboprophylaxis over intermediate or therapeutic dose anticoagulation (conditional recommendation, ungraded consensus-based statement) | Recommend inpatient only over inpatient plus postdischarge prophylaxis |
| ISTH34 | Recommend therapeutic anticoagulation in select patients (I, A) | Intermediate or therapeutic dose anticoagulation not recommended over prophylactic-dose heparin (3, B-R) | In select patients, postdischarge prophylactic dose rivaroxaban may be considered (2b, B-R) |
| NICE31 | Consider treatment dose LMWH for young people and adults with COVID-19 who need low-flow oxygen and who do not have an increased bleeding risk (conditional recommendation) | Intermediate or treatment dose heparin offered only as part of a clinical trial (only in research settings) | In-hospital prophylaxis should continue for 7 d, including after discharge (recommendation) |
| NIH COVID-19 Guideline42 | Recommend therapeutic-dose heparin for patients who have a D-dimer above the upper limit of normal, require low-flow oxygen, and have no increased bleeding risk (CIIa) | Recommend prophylactic-dose heparin (AI). Recommends against the use of intermediate-dose (eg, enoxaparin 1 mg/kg daily) and therapeutic-dose anticoagulation for VTE prophylaxis, except in a clinical trial (BI) | Recommends against routinely continuing VTE prophylaxis after hospital discharge (AIII). For patients who are at high risk for VTE and at low risk of bleeding, extended VTE prophylaxis can be considered, as per the protocol for patients without COVID-19 (BI) |
| World Health Organization30 | Suggest standard thromboprophylaxis dosing* (conditional recommendation, very low certainty) | Suggest standard thromboprophylaxis dosing* (conditional recommendation, very low certainty) |
ISTH guideline used American Heart Association recommendations and levels of evidence: 1, strong; 2b, weak; 3, no benefit; A, high quality evidence; B-R, moderate quality evidence. NIH COVID-19 guideline strength of recommendation: A, strong recommendation for the statement; B, moderate recommendation for the statement; C, optional recommendation for the statement. Quality of evidence for recommendation: I, 1 or more randomized trials without major limitations; IIa, other randomized trials or subgroup analyses of randomized trials; IIb, nonrandomized trials or observational cohort studies; III, expert opinion.
Recommendation made before completion of randomized control trials.
Consistent with their recommendations for medical inpatients, the NICE guidelines recommend that inpatient prophylaxis be continued for a minimum of 7 days, which includes after hospital discharge.31 The other guidelines do not recommend routine prophylactic-dose anticoagulation after discharge (Table 1).
Potential mechanisms of differential effect of heparin and direct oral anticoagulants in patients with COVID-19
The anticoagulation guidelines differed in their consideration of anticoagulants: most considered them individual drugs based on their mechanism of action, whereas some bundled all anticoagulants, thus combining the results of studies using heparin only with the ACTION17 trial. Both heparin and rivaroxaban exert their anticoagulant effect against activated factor X, but they do so differently, 1 indirectly via the natural inhibitor antithrombin and 1 directly. Heparin inhibits thrombin and other coagulation factor serine proteases35 and has anti-inflammatory activity and possible antiviral activity.36 Increased heparinase activity has been associated with COVID severity, and noncritically ill patients who received LMWH had lower heparinase activity.37 Increased heparinase could degrade endogenous heparan sulfate and the endothelial barrier in the lung, leading to pulmonary edema.38 Blocking heparinase could also reduce release of high-molecular-weight kininogen from heparan and chondroitin sulfate, thus inhibiting bradykinin production.39 Heparin can directly bind chemokines and cytokines, such as interleukin-8, which affects neutrophil chemotaxis.40 Heparin’s antiviral effect occurs directly by inhibiting SARS-CoV-2 binding and cell invasion by inducing a conformational change in the spike protein.41 Overall, the proposed extended anticoagulant and nonanticoagulant properties of heparin may explain why heparin, unlike rivaroxaban, showed benefit in RCTs.14
Conclusions
The COVID-19 pandemic has illustrated the importance of hematologic complications of viral infectious disease. Collaboration between government agencies, the private sector, institutions, investigators, and research networks resulted in multiple completed and pending randomized clinical trials that have changed how we care for hospitalized patients with COVID-19.
Authorship
Contribution: All authors contributed to the writing, editing, and approval of the manuscript.
Conflict-of-interest disclosure: All authors were investigators in the RAPID trial. L.B.K. and M.C. received research funding from the NIH for ACTIV-4 trials.
Correspondence: Lisa M. Baumann Kreuziger, Blood Research Institute, Versiti, 8733 Watertown Plank Rd, Milwaukee, WI 53226; e-mail: lisakreuziger@versiti.org.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal