Abstract
In multiple myeloma (MM), among the anomalies associated with shorter survival, it should be noted recurrent copy number variations (CNV), such as del(17p13), del(1p32), or 1q gains. These abnormalities are considered secondary, thus acquired during the course of the disease. Although they can be observed from diagnosis, they are only detected at the time of relapses in some patients. It is now clearly demonstrated that MM is a molecular subclonal disease, with the coexistence of different subclones of varying size. As in other cancers, it has also been shown that these different subclones can vary in size according to the different relapses, suggesting that an unnoticed minor subclone at diagnosis could become the major one at the time of relapse. Although subclonality has been largely reported at the mutational level, little is known about this phenomenon at the CNV level. Since a few CNV are associated with a poor outcome, and assuming they will impact the disease course, it would be clinically highly valuable to track these abnormalities at the time of diagnosis in minor subclones. Here, we report the first analysis at the single cell level of CNV of tumor cells from 81 patients with MM analyzed at the time of diagnosis, relapse or pre-symptomatic stages.
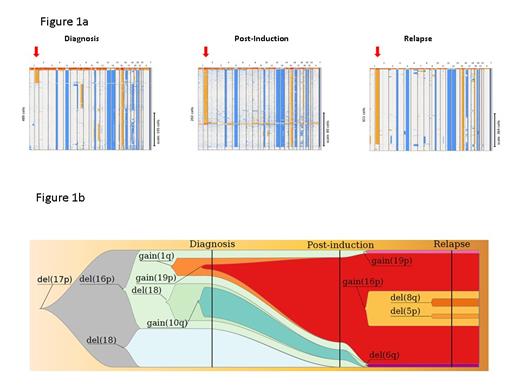
A total of 52,176 single MM cells were analyzed. The number of analyzable tumor cells was variable from patients to patients, due to heterogeneity in the sample cellularity, but also variability in cell capture rate for the single cell analysis. The mean number of analyzable cells was 656 MM cells per patient (range=83-5192). To avoid possible false positive cases, we arbitrarily defined a subclone by at least 10 cells presenting the same CNV. In these 81 patients, at least one subclone was detected in 74 of them (91%). In this cohort, a subclonal high-risk CNV (not detected on the routine assessment) was observed in 19 patients (23.5%). We had the opportunity to also analyze at single cell level residual MM cells from one patient at the end of induction therapy, and early relapse. This patient displayed a 1q gain subclone at diagnosis (16 % of the analyzed cells), not detected in the bulk analysis. At the post-induction time (thus before high-dose melphalan and autologous stem cell transplantation), 70 % of the 202 analyzable tumor cells presented this high-risk feature. This patient did relapse only 18 months after diagnosis and at this time, 92% of the cells harbored the 1q gain (Figure 1).
To address the question whether these high-risk subclonal features may impact outcome as soon as diagnosis, we explored our database. We found that 1q gains are observed in 34.1% of patients (1891/5539) at the time of diagnosis. When analyzing patients at relapse, 46.3% of them displayed a 1q gain (622/1341), thus a 12% increase. This increasing frequency reflects clonal selection of minor subclones already present at diagnosis but not detectable by conventional technique (found in comparable proportions), rather than actual acquisition after diagnosis during evolution of the disease. We then looked at the prognostic impact of 1q gain in patients displaying this CNV at diagnosis versus those presenting it only at the time of first relapse. Interestingly, the PFS and OS) curves were totally superimposable, suggesting that this subclonal 1q gain present from diagnosis in these latter patients impacts survival, possibly through early expansion. In order to go further in the molecular consequences of these high-risk subclones, we did perform single cell RNA-sequencing in 63 of the 81 patients. 1q gains defined specific subclones in the MM cells expression analyses from affected patients. Based on the expression of CDC28 protein kinase regulatory subunit 1B (CKS1B), located at 1q21, we found that high expression was restricted to specific subclones.
In conclusion, our study highly suggests that high-risk CNVs are not acquired after, but are present from the diagnosis, and selected by the treatment. These findings may have implications in high-risk definition and patient's management, by reinforcing the therapeutic strategy in these patients as soon as the first line therapy to prevent the clonal selection of these aggressive subclones. Whether this finding could be extended to cancer in general but also in some pre-malignant stages is possible, but needs to be explored.
Perrot: GSK: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; BMS Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Honoraria; Sanofi: Honoraria, Research Funding; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Leleu: Carsgen Therapeutics Ltd: Honoraria; Celgene: Honoraria; Gilead Sciences: Honoraria; Janssen-Cilag: Honoraria; Karyopharm Therapeutics: Honoraria; Merck: Honoraria; Mundipharma: Honoraria; Novartis: Honoraria; Oncopeptides: Honoraria; Pierre Fabre: Honoraria; Roche: Honoraria; Sanofi: Honoraria; Bristol-Myers Squibb: Honoraria; Amgen: Honoraria; Takeda: Honoraria, Other: Non-financial support; AbbVie: Honoraria. Manier: Oncopeptides: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; GSK: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene - Bristol Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Adaptive Biotechnologies: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Regeneron: Consultancy, Research Funding; Novartis: Consultancy, Research Funding. Macro: Sanofi: Honoraria; GSK: Honoraria; Takeda: Honoraria, Other: Travel accomodation, Research Funding; Janssen: Honoraria, Other: Travel accomodation, Research Funding; Celgen/BMS: Honoraria. Mohty: Novartis: Honoraria; Celgene: Honoraria, Research Funding; Janssen: Honoraria, Research Funding; Amgen: Honoraria; Adaptive Biotechnologies: Honoraria; Bristol Myers Squibb: Honoraria; Astellas: Honoraria; Sanofi: Honoraria, Research Funding; Takeda: Honoraria; Gilead: Honoraria; Pfizer: Honoraria; Jazz: Honoraria, Research Funding. Munshi: Novartis: Consultancy; Takeda: Consultancy; Bristol-Myers Squibb: Consultancy; Janssen: Consultancy; Oncopep: Consultancy, Current equity holder in publicly-traded company, Other: scientific founder, Patents & Royalties; Abbvie: Consultancy; Amgen: Consultancy; Karyopharm: Consultancy; Celgene: Consultancy; Adaptive Biotechnology: Consultancy; Legend: Consultancy; Pfizer: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal