Abstract

Background/Introduction:
Lower-risk (IPSS low risk and Int-1) myelodysplastic syndromes (MDS) are typically treated supportively to address cytopenias. DNA methyltransferase inhibitors (DNMTi) such as azacitidine and decitabine (DEC) are FDA-approved for higher risk MDS patients (pts), and while the DEC USPI includes IPSS Int-1 pts, it is not widely used in this population. Approved intravenous (IV) or subcutaneous (SC) regimens require 5-7 days of treatment every month burdening older cancer pts due to daily travel and treatment time and may increase potential risk from pandemic SARS-CoV-2 infection. Because DNMTis are rapidly degraded by cytidine deaminase (CDA) in the gut and liver, oral availability has only been recently possible. A randomized study with CC-486, an oral formulation of azacitidine, in the Int-1 population showed a median overall survival (mOS) of approximately 17 months for both placebo and treated patients (Garcia-Manero, 2021). Oral DEC 35 mg/cedazuridine 100 mg (ASTX727) or DEC-C, is an oral fixed dose combination (FDC) of DEC and the CDA inhibitor cedazuridine (CED) resulting in equivalent exposure (99%; 90% CI 93% to 106%) to standard IV DEC 20 mg/m 2 for 5 days in an intra-patient randomized cross-over study (Garcia-Manero et al, ASH 2019). Here, we present data on patients with lower risk MDS from that study.
Methods:
We used a randomized cross over design with pts randomized 1:1 in the first 2 cycles to either Sequence A: (DEC 35 mg/ CED 100 mg in Cycle 1 and IV DEC at 20 mg/m 2 in Cycle 2), or Sequence B (IV DEC in Cycle 1 and oral DEC/CED in Cycle 2). Cycles were repeated every 28 days unless delays were needed, and all patients received oral DEC-C in Cycles 3+ until disease progression or unacceptable toxicity. We conducted an intra-patient comparison of DEC PK (DEC AUC equivalence over 5 days of dosing). Pts were eligible as per the FDA-approved label of IV DEC (MDS pts by FAB classification including CMML, or MDS IPSS Intermediate-1, 2 or high-risk pts). Clinical endpoints were best response as assessed by an independent expert panel according to IWG 2006 response criteria, transfusion independence (TI), overall survival (OS), and safety.
Results:
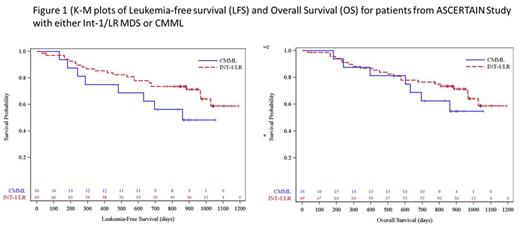
Of the 133 pts treated in ASCERTAIN, 69 had a diagnosis of lower-risk MDS (93% Int-1, 7% LR). Median age was 70.0 years (range 45-87), 65% were male, median weight was 84 kg (range 50-127), median baseline hematologic parameters were: hemoglobin 89 g/L (range 69.8-146.5), WBCs 1.50 X 10 9/L (range 0.11-7.1), platelets (plt) 86 x 10 9/L (range 5-703), bone marrow blasts 4% (range 0-18), cytogenetics: 7 (10.1%) poor-risk, 21 (30.4%) intermediate risk, 37 (53.6%) better-risk, 4 (5.7%) missing or not evaluable. 27(39%) of the pts were RBC transfusion dependent (TD) and 6 (9%) plt TD. 17 (25%) had received prior MDS treatment, 3% prior DNMTi. Pts received a median of 9 cycles of therapy (range 1-28). Treatment-emergent adverse events of CTCAE Gr 3 or higher in >10% of pts, independent of relationship to ASTX727, included cytopenias (neutropenia [59%], thrombocytopenia [58%], anemia [48%], leukopenia [26%]), febrile neutropenia (32%), and pneumonia (19%). Sixteen pts (23%) achieved Complete Response (CR), 18 (26%) had marrow CR (mCR), including 9 (13%) with hematologic improvement (HI). Overall Response rate (ORR; CR + PR+ mCR + HI) was 57%. Of those RBC or plt TD at baseline, 13 (48%) became RBC TI and 4 (67%) became plt TI. With approximately 32 months of median follow up, neither median leukemia-free survival (mLFS) nor mOS had been reached (Figure 1). Twelve pts (17%) went on to allogeneic stem cell transplant.
Conclusions:
Oral decitabine/cedazuridine given as a FDC in MDS pts produced equivalent PK exposure to 20 mg/m 2 IV DEC; in lower risk MDS pts with treatment indicated, the agent was generally well-tolerated with prolonged treatment and could result in mLFS and mOS which exceeds 32 months. This FDC and other dosing regimens of oral DEC-C should be further studied in this patient population.
References:
Garcia-Manero, et al, ASH 2019
Savona, et al, Int. MDS Symp. 2021
Garcia-Manero, et al, Phase III, Randomized, Placebo-Controlled Trial of CC-486 (Oral Azacitidine) in Patients with Lower-Risk Myelodysplastic Syndromes. J.Clin.Onc. 2021 39:13, 1426-1436
McCloskey: Pfizer: Consultancy; Jazz: Consultancy, Speakers Bureau; COTA: Other: Equity Ownership; Incyte: Speakers Bureau; Takeda: Consultancy, Speakers Bureau; Novartis: Consultancy; BMS: Honoraria, Speakers Bureau; Amgen: Speakers Bureau. Griffiths: Alexion Pharmaceuticals: Consultancy, Research Funding; Abbvie: Consultancy, Honoraria; Taiho Oncology: Consultancy, Honoraria; Genentech: Research Funding; Novartis: Honoraria; Takeda Oncology: Consultancy, Honoraria; Astex Pharmaceuticals: Honoraria, Research Funding; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria, Research Funding; Apellis Pharmaceuticals: Research Funding; Boston Biomedical: Consultancy. Yee: Paladin: Membership on an entity's Board of Directors or advisory committees; TaiHo: Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Genentech: Research Funding; Geron: Research Funding; Janssen: Research Funding; Jazz: Research Funding; MedImmune: Research Funding; Onconova: Research Funding; Tolero: Research Funding; AbbVie: Honoraria; Bristol-Myers Squibb/Celgene: Membership on an entity's Board of Directors or advisory committees; Otsuka: Membership on an entity's Board of Directors or advisory committees; Shattuck Labs: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; F. Hoffmann La Roche: Membership on an entity's Board of Directors or advisory committees, Research Funding; Forma Therapeutics: Research Funding; Astex: Membership on an entity's Board of Directors or advisory committees, Research Funding. Zeidan: Novartis: Consultancy, Other: Clinical Trial Committees, Travel support, Research Funding; Genentech: Consultancy; Ionis: Consultancy; Astellas: Consultancy; Epizyme: Consultancy; AbbVie: Consultancy, Other: Clinical Trial Committees, Research Funding; Jasper: Consultancy; Cardiff Oncology: Consultancy, Other: Travel support, Research Funding; BeyondSpring: Consultancy; Loxo Oncology: Consultancy, Other: Clinical Trial Committees; Janssen: Consultancy; Acceleron: Consultancy, Research Funding; AstraZeneca: Consultancy; Kura: Consultancy, Other: Clinical Trial Committees; Gilead: Consultancy, Other: Clinical Trial Committees; Agios: Consultancy; Daiichi Sankyo: Consultancy; Boehringer Ingelheim: Consultancy, Research Funding; Geron: Other: Clinical Trial Committees; BMS: Consultancy, Other: Clinical Trial Committees, Research Funding; BioCryst: Other: Clinical Trial Committees; Pfizer: Other: Travel support, Research Funding; Aprea: Consultancy, Research Funding; ADC Therapeutics: Research Funding; Jazz: Consultancy; Incyte: Consultancy, Research Funding; Amgen: Consultancy, Research Funding; Astex: Research Funding. Al-Kali: Astex: Other: Research support to institution; Novartis: Research Funding. Patel: Celgene-BMS: Membership on an entity's Board of Directors or advisory committees; PVI: Honoraria; Agios: Membership on an entity's Board of Directors or advisory committees. Sabloff: Pfizer: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Astellas: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; ROCHE: Membership on an entity's Board of Directors or advisory committees; TaiHo: Membership on an entity's Board of Directors or advisory committees; Jaxx: Membership on an entity's Board of Directors or advisory committees; Celgene: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees; Abbvie: Membership on an entity's Board of Directors or advisory committees. Dao: Astex Pharmaceuticals, Inc.: Current Employment. Fazal: Taiho Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Stemline Therapeutics: Consultancy, Honoraria, Speakers Bureau; Sanofi Genzyme: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Speakers Bureau; Karyopharm Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Jazz Pharmaceuticals: Consultancy, Honoraria, Speakers Bureau; Janssen Oncology: Consultancy, Honoraria, Speakers Bureau; Incyte: Consultancy, Honoraria, Speakers Bureau; Glaxo Smith Kline: Consultancy, Honoraria, Speakers Bureau; Gilead Sciences: Consultancy, Honoraria, Speakers Bureau; Bristol Myers Squibb: Consultancy, Honoraria, Speakers Bureau; AMGEN: Consultancy, Honoraria, Speakers Bureau; Agios: Consultancy, Honoraria, Speakers Bureau; Takeda: Consultancy, Honoraria, Speakers Bureau. Odenike: AbbVie, Celgene, Impact Biomedicines, Novartis, Taiho Oncology, Takeda: Consultancy; Celgene, Incyte, AstraZeneca, Astex, NS Pharma, AbbVie, Gilead, Janssen, Oncotherapy, Agios, CTI/Baxalta, Aprea: Research Funding. Kantarjian: AbbVie: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Ascentage: Research Funding; Pfizer: Honoraria, Research Funding; BMS: Research Funding; Daiichi-Sankyo: Research Funding; Amgen: Honoraria, Research Funding; Ipsen Pharmaceuticals: Honoraria; Jazz: Research Funding; Astellas Health: Honoraria; Immunogen: Research Funding; Astra Zeneca: Honoraria; Aptitude Health: Honoraria; KAHR Medical Ltd: Honoraria; NOVA Research: Honoraria; Precision Biosciences: Honoraria; Taiho Pharmaceutical Canada: Honoraria. DeZern: Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Taiho: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees. Roboz: Novartis: Consultancy; Mesoblast: Consultancy; Jasper Therapeutics: Consultancy; Jazz: Consultancy; MEI Pharma - IDMC Chair: Consultancy; Daiichi Sankyo: Consultancy; Otsuka: Consultancy; Bristol Myers Squibb: Consultancy; Blueprint Medicines: Consultancy; Bayer: Consultancy; AstraZeneca: Consultancy; Astellas: Consultancy; Astex: Consultancy; Amgen: Consultancy; Agios: Consultancy; Actinium: Consultancy; AbbVie: Consultancy; Janssen: Research Funding; Celgene: Consultancy; Glaxo SmithKline: Consultancy; Helsinn: Consultancy; Janssen: Consultancy; Pfizer: Consultancy; Roche/Genentech: Consultancy. Busque: Novartis: Consultancy. Leber: Astellas: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Otsuka: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; TaiHo: Honoraria, Membership on an entity's Board of Directors or advisory committees; AMGEN: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Abbvie: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Novartis: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Jazz: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Hao: Astex Pharmaceuticals, Inc.: Current Employment. Keer: Astex Pharmaceuticals, Inc.: Current Employment. Azab: Astex Pharmaceuticals, Inc.: Membership on an entity's Board of Directors or advisory committees. Savona: Geron: Consultancy, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Consultancy, Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees; CTI: Consultancy, Membership on an entity's Board of Directors or advisory committees; BMS-Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; NOVARTIS: Consultancy, Membership on an entity's Board of Directors or advisory committees; Ryvu: Consultancy, Membership on an entity's Board of Directors or advisory committees; Sierra Oncology: Consultancy, Membership on an entity's Board of Directors or advisory committees; Taiho: Consultancy, Membership on an entity's Board of Directors or advisory committees; TG Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; ALX Oncology: Research Funding; Astex: Research Funding; Incyte: Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal