Abstract
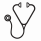
Introduction:
The interleukin-3 receptor alpha chain (CD123) is a cell-surface target aberrantly expressed on various myeloid neoplasms including blastic plasmacytoid dendritic cell neoplasm (BPDCN), acute myeloid leukemia (AML), chronic myelomonocytic leukemia (CMML), and myelofibrosis (MF). Tagraxofusp (TAG, SL-401), a first-in-class CD123-targeted therapy, is the only treatment approved by the FDA and EMA for patients (pts) with BPDCN. It has also been investigated in phase 1/2 clinical trials for AML (including pts with minimal residual disease [MRD]), CMML, and MF. We report the aggregated safety data of TAG monotherapy from trials in BPDCN, AML, CMML, and MF.
Methods:
An integrated safety analysis was performed for pts who received TAG monotherapy in three phase 1/2, multicenter clinical studies: Study 0114 (NCT02113982; BPDCN/AML), Study 0214 (NCT02270463; AML with/without MRD), and Study 0314 (NCT02268253; CMML/MF; Table 1). Pts received the recommended phase 2 dose of 12 mcg/kg/day (d) IV on d1-3 (MF and CMML) or d1-5 (BPDCN and AML) and were analyzed for incidence and timing of treatment-related adverse events (TRAEs), AEs of interest, and discontinuations. Safety data for TAG monotherapy, reported as individual case study reports (ICSRs) postapproval (US) and from an early access program (EAP; EU) are also presented.
Results:
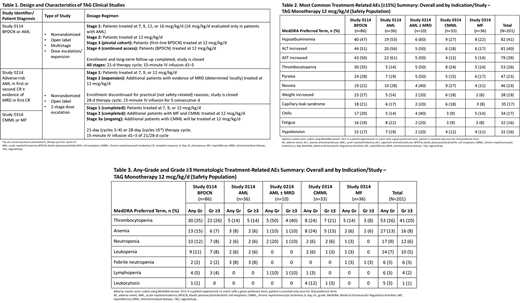
In total, 201 pts receiving TAG (12 mcg/kg/d) in a clinical trial setting were included: BPDCN, n=86; AML, n=36; AML with/without MRD, n=10; CMML, n=33; MF, n=36. As of Jul 2021, 11 (6%) pts discontinued TAG due to any-grade (Gr) TRAE. Most common TRAEs (any-Gr) are shown in Table 2 and included hypoalbuminemia (41%), increased alanine aminotransferase (ALT; 40%), increased aspartate aminotransferase (AST; 39%), and thrombocytopenia (26%). The most common Gr ≥3 TRAEs were thrombocytopenia (n=41; 20%), increased AST (n=40; 20%), and increased ALT (n=35; 17%). Tumor lysis syndrome occurred in 5% of pts and 1.5% had infusion-related reactions. Any-Gr and Gr ≥3 hematologic TRAEs are presented in Table 3. Most common was thrombocytopenia. All TRAEs were transient in nature; prolonged bone marrow suppression was not observed. Overall, 23% (47/201) of pts experienced ≥1 treatment-related serious AE. Onset of most TRAEs, irrespective of Gr, commonly occurred in cycle 1 and resolved by cycle 2. Although the number of pts in cycles ≥2 was lower than in cycle 1, the incidence rates of common TRAEs also substantially decreased in subsequent cycles.
Capillary leak syndrome (CLS) occurred in 17% of the overall 201 pts. The majority of all CLS events were nonsevere, Gr ≤2 (n=22; 63%); also reported were Gr 3, n=10 (29%); Gr 4, n=3 (9%), and 2 pts experienced a Gr 5 event. Onset of CLS was usually within the first week of cycle 1 (median time to onset [range], d: 5.5 [1-51]) and resolved quickly (median time to resolution [range], d: 5.0 [2-69]); no pts experienced the first onset after cycle 2. Of the 15 pts who resumed treatment after an initial CLS event, only 1 pt experienced a recurrence.
The most common AEs reported as ICSRs postapproval and from an EAP (December 2018 to June 2021) were CLS, blood albumin decreased, thrombocytopenia, pyrexia, and hepatic enzyme increased. No new safety signals that would alter the safety profile were observed.
Conclusions:
This integrated analysis represents the largest collection of safety data (N=201) following treatment with TAG monotherapy. TAG has an established and manageable safety profile, with the vast majority of AEs reported as transient and decreasing in frequency/severity with increasing cycles. CLS was reported in 17% of pts; onset was usually within cycle 1, resolved quickly (median time to resolution: 5.0 d) by early intervention measures including holding treatment, and no pts experienced first onset after cycle 2. Risk-minimization guidelines for CLS were developed and implemented during clinical trials and incorporated into prescribing information. No myelosuppression or cumulative toxicity was observed over multiple treatment cycles. The safety profile observed postapproval and in the EAP was consistent with the clinical trial data reported, reflecting successful adoption of risk-management strategies in real-world practice. These data confirm the favorable benefit-risk profile of TAG monotherapy has been maintained in the 3 years following US approval.
Pemmaraju: ASCO Leukemia Advisory Panel: Membership on an entity's Board of Directors or advisory committees; Sager Strong Foundation: Other; Blueprint Medicines: Consultancy; Incyte: Consultancy; Bristol-Myers Squibb Co.: Consultancy; Pacylex Pharmaceuticals: Consultancy; Cellectis S.A. ADR: Other, Research Funding; Aptitude Health: Consultancy; Celgene Corporation: Consultancy; CareDx, Inc.: Consultancy; Springer Science + Business Media: Other; Daiichi Sankyo, Inc.: Other, Research Funding; Samus: Other, Research Funding; Clearview Healthcare Partners: Consultancy; Dan's House of Hope: Membership on an entity's Board of Directors or advisory committees; MustangBio: Consultancy, Other; Abbvie Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; HemOnc Times/Oncology Times: Membership on an entity's Board of Directors or advisory committees; ASH Communications Committee: Membership on an entity's Board of Directors or advisory committees; LFB Biotechnologies: Consultancy; Novartis Pharmaceuticals: Consultancy, Other: Research Support, Research Funding; Protagonist Therapeutics, Inc.: Consultancy; Affymetrix: Consultancy, Research Funding; DAVA Oncology: Consultancy; Plexxicon: Other, Research Funding; Roche Diagnostics: Consultancy; Stemline Therapeutics, Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; ImmunoGen, Inc: Consultancy. Kantarjian: NOVA Research: Honoraria; Aptitude Health: Honoraria; Immunogen: Research Funding; BMS: Research Funding; Precision Biosciences: Honoraria; Novartis: Honoraria, Research Funding; Ascentage: Research Funding; Pfizer: Honoraria, Research Funding; Astra Zeneca: Honoraria; Astellas Health: Honoraria; Jazz: Research Funding; Ipsen Pharmaceuticals: Honoraria; Amgen: Honoraria, Research Funding; Daiichi-Sankyo: Research Funding; KAHR Medical Ltd: Honoraria; AbbVie: Honoraria, Research Funding; Taiho Pharmaceutical Canada: Honoraria. Sweet: Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; AROG: Membership on an entity's Board of Directors or advisory committees; Gilead: Membership on an entity's Board of Directors or advisory committees; Bristol Meyers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees. Wang: Kite Pharmaceuticals: Consultancy, Honoraria, Other: Advisory Board; Jazz Pharmaceuticals: Consultancy, Honoraria, Other: Advisory Board; DAVA Oncology: Consultancy, Speakers Bureau; BMS/Celgene: Membership on an entity's Board of Directors or advisory committees; Stemline Therapeutics: Consultancy, Honoraria, Other: Advisory board, Speakers Bureau; Genentech: Membership on an entity's Board of Directors or advisory committees; GlaxoSmithKline: Consultancy, Honoraria, Other: Advisory Board; Takeda: Consultancy, Honoraria, Other: Advisory board; Astellas: Consultancy, Membership on an entity's Board of Directors or advisory committees; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pfizer: Consultancy, Honoraria, Other: Advisory Board, Speakers Bureau; Kura Oncology: Consultancy, Honoraria, Other: Advisory board, steering committee, Speakers Bureau; Novartis: Consultancy, Honoraria, Other: Advisory Board; Mana Therapeutics: Consultancy, Honoraria; Rafael Pharmaceuticals: Other: Data safety monitoring committee; Gilead: Consultancy, Honoraria, Other: Advisory board; Daiichi Sankyo: Consultancy, Honoraria, Other: Advisory board; PTC Therapeutics: Consultancy, Honoraria, Other: Advisory board; Genentech: Consultancy; MacroGenics: Consultancy. Lane: N-of-One: Consultancy, Honoraria; Stemline Therapeutics: Research Funding; Qiagen: Consultancy, Honoraria; AbbVie: Research Funding. Ali: Incyte: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Speakers Bureau; CTI BioPharma: Membership on an entity's Board of Directors or advisory committees. Stein: Amgen: Consultancy, Speakers Bureau; Celgene: Speakers Bureau; Stemline: Speakers Bureau. Yacoub: Agios: Membership on an entity's Board of Directors or advisory committees; Acceleron Pharma: Membership on an entity's Board of Directors or advisory committees; CTI Biopharma: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Incyte: Speakers Bureau. Rizzieri: Celltron/Teva: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: presentation to FDA for biosimilar review ; AROG: Other; Amgen: Other: personal fee; Bayer: Consultancy, Honoraria; Kite: Other: personal fee; Mustang: Consultancy, Honoraria; Stemline Therapeutics: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Incyte: Other: personal fee; Gilead: Other: personal fee; Jazz: Other: personal fee; Pharmacyclics: Other. Vasu: Seattle Genetics: Other: travel support; Boehringer Ingelheim: Other: Travel support; Kiadis, Inc.: Research Funding; Omeros, Inc.: Membership on an entity's Board of Directors or advisory committees. Gupta: AbbVie: Consultancy, Honoraria; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Roche: Consultancy; Constellation Pharma: Consultancy, Honoraria; Pfizer: Consultancy; BMS-Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Sierra Oncology: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Incyte: Honoraria, Research Funding. Lee: BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees; Pin Therapeutics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Innate: Consultancy, Membership on an entity's Board of Directors or advisory committees; AstraZeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees. Schiller: Sangamo: Research Funding; Actinium Pharmaceuticals, Inc: Research Funding; FujiFilm: Research Funding; Pfizer: Current equity holder in publicly-traded company, Research Funding; Abbvie: Research Funding; Karyopharm: Research Funding; Onconova: Research Funding; Celator: Research Funding; Actuate: Research Funding; Incyte: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Trovagene: Research Funding; Deciphera: Research Funding; Gamida Cell Ltd.: Research Funding; Forma: Research Funding; Ono-UK: Consultancy, Research Funding; Novartis: Consultancy, Research Funding; Arog: Research Funding; Takeda: Research Funding; Geron: Research Funding; Jazz: Consultancy, Honoraria, Research Funding, Speakers Bureau; PrECOG: Research Funding; Kite/Gilead: Honoraria, Research Funding, Speakers Bureau; Mateon: Research Funding; Constellation Pharmaceuticals: Research Funding; Daiichi-Sankyo: Research Funding; Tolero: Research Funding; Stemline Therapeutics, Inc.: Honoraria, Research Funding, Speakers Bureau; Samus: Research Funding; Astellas: Honoraria, Research Funding, Speakers Bureau; Regimmune: Research Funding; BMS/Celgene: Consultancy, Current equity holder in publicly-traded company, Research Funding, Speakers Bureau; Agios: Consultancy, Research Funding, Speakers Bureau; Amgen: Consultancy, Current equity holder in publicly-traded company, Honoraria, Research Funding, Speakers Bureau; Bio: Research Funding; Elevate: Research Funding; Delta-Fly: Research Funding; Genentech-Roche: Research Funding; Ariad: Research Funding; Sanofi: Honoraria, Research Funding, Speakers Bureau; Pharma: Consultancy; Johnson & Johnson: Current equity holder in publicly-traded company; Biomed Valley Discoveries: Research Funding; Eli Lilly: Research Funding; ASH foundation: Other: Chair-unpaid; Sellas: Research Funding; Ono: Consultancy; Incyte: Consultancy; AstraZeneca: Consultancy; Kaiser Permanente: Consultancy; Cyclacel: Research Funding; MedImmune: Research Funding; Ambit: Research Funding; Leukemia & Lymphoma Society: Research Funding; Bluebird Bio: Research Funding; Boehringer-Ingleheim: Research Funding; Cellerant: Research Funding; CTI Biopharma: Research Funding; Janssen: Research Funding; Kura Oncology: Research Funding; Pharmacyclics: Honoraria, Speakers Bureau; Millennium: Research Funding; National Marrow Donor Program: Research Funding; NIH: Research Funding; Onyx: Research Funding; Pharmamar: Research Funding; UC Davis: Research Funding; UCSD: Research Funding; Evidera: Consultancy; NCI: Consultancy; Novartis: Speakers Bureau. Foran: trillium: Research Funding; actinium: Research Funding; aptose: Research Funding; novartis: Honoraria; OncLive: Honoraria; servier: Honoraria; gamida: Honoraria; takeda: Research Funding; boehringer ingelheim: Research Funding; abbvie: Research Funding; certara: Honoraria; bms: Honoraria; pfizer: Honoraria; sanofi aventis: Honoraria; revolution medicine: Honoraria; taiho: Honoraria; syros: Honoraria; kura: Research Funding; h3bioscience: Research Funding; aprea: Research Funding; sellas: Research Funding; stemline: Research Funding. Walter: Macrogenics: Consultancy, Research Funding; Jazz: Research Funding; Pfizer: Consultancy, Research Funding; Selvita: Research Funding; Amphivena: Consultancy, Other: ownership interests; Agios: Consultancy; Astellas: Consultancy; BMS: Consultancy; Genentech: Consultancy; Janssen: Consultancy; Kite: Consultancy; Immunogen: Research Funding; Celgene: Consultancy, Research Funding; Aptevo: Consultancy, Research Funding; Amgen: Research Funding. Sieminski: Stemline Therapeutics: Current Employment, Current holder of stock options in a privately-held company. Patnaik: Kura Oncology: Research Funding; Stemline Therapeutics: Membership on an entity's Board of Directors or advisory committees; Stemline Therapeutics: Membership on an entity's Board of Directors or advisory committees. Mughal: Oxford University Press, Informa: Other: financial benefit and/or patents ; Stemline: Current Employment, Current holder of stock options in a privately-held company. Konopleva: Calithera: Other: grant support, Research Funding; Reata Pharmaceuticals: Current holder of stock options in a privately-held company, Patents & Royalties: intellectual property rights; Genentech: Consultancy, Honoraria, Other: grant support, Research Funding; Agios: Other: grant support, Research Funding; Ascentage: Other: grant support, Research Funding; AstraZeneca: Other: grant support, Research Funding; Rafael Pharmaceuticals: Other: grant support, Research Funding; Forty Seven: Other: grant support, Research Funding; Ablynx: Other: grant support, Research Funding; Eli Lilly: Patents & Royalties: intellectual property rights, Research Funding; Novartis: Other: research funding pending, Patents & Royalties: intellectual property rights; Stemline Therapeutics: Research Funding; KisoJi: Research Funding; F. Hoffmann-La Roche: Consultancy, Honoraria, Other: grant support; Cellectis: Other: grant support; Sanofi: Other: grant support, Research Funding; AbbVie: Consultancy, Honoraria, Other: Grant Support, Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal