Key Points
Expression level, viability, and functional capacity of CD70scFv-CAR constructs are strongly influenced by their structural composition.
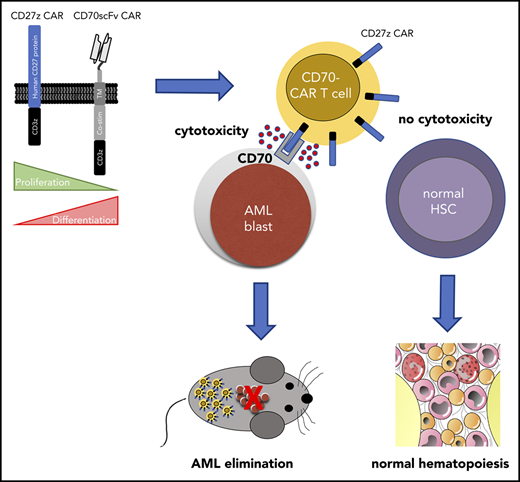
CD27z-CAR T cells can effectively eliminate AML cells in vitro and in vivo, but spare normal hematopoiesis.
Abstract
The prognosis of patients with acute myeloid leukemia (AML) remains dismal, highlighting the need for novel innovative treatment strategies. The application of chimeric antigen receptor (CAR) T-cell therapy to patients with AML has been limited, in particular by the lack of a tumor-specific target antigen. CD70 is a promising antigen to target AML, as it is expressed on most leukemic blasts, whereas little or no expression is detectable in normal bone marrow samples. To target CD70 on AML cells, we generated a panel of CD70-CAR T cells that contained a common single-chain variable fragment (scFv) for antigen detection, but differed in size and flexibility of the extracellular spacer and in the transmembrane and the costimulatory domains. These CD70scFv CAR T cells were compared with a CAR construct that contained human CD27, the ligand of CD70 fused to the CD3ζ chain (CD27z). The structural composition of the CAR strongly influenced expression levels, viability, expansion, and cytotoxic capacities of CD70scFv-based CAR T cells, but CD27z-CAR T cells demonstrated superior proliferation and antitumor activity in vitro and in vivo, compared with all CD70scFv-CAR T cells. Although CD70-CAR T cells recognized activated virus-specific T cells (VSTs) that expressed CD70, they did not prevent colony formation by normal hematopoietic stem cells. Thus, CD70-targeted immunotherapy is a promising new treatment strategy for patients with CD70-positive AML that does not affect normal hematopoiesis but will require monitoring of virus-specific T-cell responses.
Introduction
Treatment of patients with acute myeloid leukemia (AML), the most common acute leukemia among adults, remains challenging. Changes in standard treatment have been moderate during the past 3 decades, and the outcome remains poor for most patients, in particular for those aged >60.1,2 Disease remission can be achieved in >80% of patients with AML with intensive chemotherapy regimens. However, more than 50% of patients eventually relapse. This high rate of treatment failure has been attributed in part to leukemic stem cells (LSCs), a minor fraction of leukemic cells that maintain and reinitiate the disease. LSCs are resistant to conventional chemotherapy because of their quiescent cellular state,3,4 but their elimination is paramount to maintaining long-term remissions.
The adoptive transfer of chimeric antigen receptor (CAR)-modified T cells has evolved as a promising treatment of patients with CD19-expressing malignancies, such as acute lymphoblastic leukemia and B-cell lymphoma. So far, patients with AML have not benefitted from this innovative treatment strategy. A major obstacle is the identification of target antigens that are expressed on the surface of AML bulk cells and LSCs but are absent or expressed at low levels on normal hematopoietic stem cells (HSCs) and other healthy tissues. CAR T cells specific for Lewis Y,5 CD33,6,7 CD123,7 CD44-v6,8 CLL-1,9 FLT-3,10 and folate receptor-β11 have proved to be effective in preclinical AML models, and CD33 and CD123 are currently being evaluated in clinical trials as targets for CAR T-cell therapy for AML.12 Despite their potency in eliminating AML bulk cells and LSCs, CD33- and CD123-CAR T cells can be used only as a bridge to transplant therapy, because both target antigens are also expressed on normal HSCs.
CD70 is a type 2 transmembrane glycoprotein and a member of the tumor necrosis factor (TNF) ligand family. It is a promising target for CAR T-cell therapy for AML, because it is expressed on both leukemic blasts and LSCs in patients with AML.13,14 In contrast to CD33 and CD123, CD70 is not detectable on normal HSCs,13,14 suggesting that CD70-specific CAR T cells could provide a therapy for AML without adversely affecting hematopoiesis. A promising antitumor activity of a CD70-specific monoclonal antibody, in combination with hypomethylating agents for the treatment of patients with AML, has been reported.15
In the present study, we evaluated CD70 as a novel therapeutic target for CAR T-cell therapy for AML. We designed a panel of CD70-CAR T cells containing different antigen binding, extracellular spacer, and intracellular signaling domains. Two of the CAR constructs exhibited significant antitumor efficacy in vitro and in vivo, with major effects imposed by antigen binding, hinge and costimulatory domains. Although AML cells were effectively eliminated, CD70-CAR T cells spared normal HSCs, thereby avoiding potentially dangerous on-target/off-tumor toxicity. Our data support CD70 as a promising target for immunotherapy, as well as early phase clinical testing of CD70-CAR T cells in patients with CD70-positive AML.
Material and methods
CD70-CAR construction
To generate a panel of CD70scFv CAR constructs, cDNA containing the VH and VL chains derived from the single-chain variable regions (scFvs) of a published antibody sequence16 was commercially synthesized (IDT, Coralville, IA) and fused to different spacers (intermediate [IM] and long-flexible [LF]), transmembrane domains (CD28 and CD27 derived), 1 of 3 costimulatory domains (CD27, CD28, and 4-1BB), and the CD3ζ chain, using In-Fusion Cloning (Takara, Mountain View, CA). The IM spacer contained an IgG1-derived CH3 and an IgG4 hinge domain, and the LF spacer consisted of a modified CH2 and CH3 domain17 that are connected through 2 IgG4 hinge domains for additional flexibility. CAR constructs that included the CD28 and the 4-1BB costimulatory domains contained the CD28 transmembrane domain, and the transmembrane domain of CAR T cells with the CD27 costimulatory domain was also derived from the CD27 molecule (supplemental Figure 2A-B, available on the Blood Web site). For the CD70ligand-CAR (CD27z-CAR), we followed our previously published design18 and fused the cDNA of the human CD27 protein to CD3ζ (supplemental Figure 2C). Using the XhoI and NotI sites, we cloned the fragments into a linearized SFG vector that contained an internal ribosome entry site sequence and a truncated NGFR or CD19 downstream of the CAR insertion site for plasmid detection (supplemental Figure 2D).
Retroviral vector production and T-cell transduction
Retroviral vector production and T-cell transduction were performed as previously described.19,20 In brief, 293T cells were transfected with packaging plasmids (PeqPam, RD114) and the SFG vector containing the CAR construct, using GeneJuice (Merck Millipore, Billerica, MA). The viral supernatant was harvested after 48 hours. Peripheral blood mononuclear cells were isolated from the peripheral blood by density gradient centrifugation, and the T cells were activated by CD3 and CD28 antibodies. The T cells were transduced in 24-well plates coated with RetroNectin (Takara) after 48 hours of expansion in complete medium (45% RPMI-1640; Hyclone, Logan, UT), 45% Click’s medium (FujiFilm; Irvine Scientific, Santa Ana, CA), 2 mM l-glutamine (Invitrogen, Carlsbad, CA), and 10% fetal bovine serum (Hyclone) supplemented with 10 ng/mL interleukin-7 (IL-7) and 5 ng/mL IL-15 (Peprotech, Rocky Hill, NJ).21
Cell lines
We obtained 293T cells and the AML cell lines THP-1 and KG-1a from ATCC. The AML cell line Molm-13 was purchased from DSMZ. The cells were maintained in Iscove’s modified Dulbecco’s medium, for Molm-13, KG-1a, and 293T, and RPMI 1640 for THP-1, both supplemented with 2 mM l-glutamine and 10% or 20% fetal bovine serum, according to the manufacturer’s recommendations, and 1% penicillin-streptomycin in a humidified atmosphere containing 5% CO2 at 37°C. IMS-M2 was kindly provided by Margaret Goodell (Baylor College of Medicine, Houston, TX). All cell lines were mycoplasma free, according to the Mycoalert Detection Kit (Lonza, Basel, Switzerland).
Flow cytometry
Fluorochrome-conjugated isotype controls and antihuman CD70, CD45, CD4, CD8, CD3, CD19, CD45RO, CD34, NGFR, CCR7, LAG3, TIM-3, PD-1, interferon-γ (IFN-γ), and TNF-⍺ antibodies were purchased from BD Biosciences (San Jose, CA), Beckman Coulter (Brea, CA), Thermo Fisher Scientific (Waltham, MA), or Biolegend (San Diego, CA). Biotin-labeled protein L and fluorochrome-labeled streptavidin for CAR detection were purchased from Thermo Fisher Scientific and BD Biosciences. Flow cytometric data were acquired using the FACSCanto II (BD Biosciences) and analyzed with FlowJo, version 10 (Tree Star, Ashland, OR).
TMA construction, immunohistochemistry, and evaluation
A tissue microarray was established using pretherapeutic bone marrow biopsy specimens from patients diagnosed with AML who had received intensive treatment in the Department of Internal Medicine A at the University Hospital of Muenster between 2006 and 2016. Informed consent was obtained from all patients, in accordance with the Declaration of Helsinki. The local ethics committee approved the study. Bone marrow specimens were fixed in formaldehyde, decalcified, and embedded in paraffin. IHCPlus Polyclonal CD27L/CD70 (clone LS-A8809; LSBio, Seattle, WA) was used as the primary antibody. Staining was performed with the UltraVision LP Detection System (Thermo Fisher Scientific). Two experienced hematologists without knowledge of the patients’ clinical data evaluated all tissue microarray samples. The staining of all bone marrow samples was evaluated using an H-score.22
Chromium-51 release assay
Short-term cytotoxicity was determined with the chromium-51 release assay. In brief, target cells were incubated with 51Cr sodium chromate for 1 hour and plated in 96-well plates. CAR or nontransduced (NT) T cells were added at multiple effector-to-target (E:T) ratios. After 5 hours of incubation, the supernatant was harvested, and chromium release was detected with a gamma counter. The mean percentage of specific lysis of triplicate wells was calculated as previously reported.23
Generation of multivirus-specific T cells
Generation of multivirus-specific T cells (MVSTs) for use in coculture assays was performed as previously described.24,25
Coculture assay
A schematic of the experimental design is shown in supplemental Figure 5. In brief, CAR or NT T cells were cocultured with tumor cells in 96-well plates in the absence of exogenous cytokines. We harvested 1 well per condition every 5 days and calculated the total number of T cells and tumor cells by flow cytometry with CountBright beads (Invitrogen). The dead cell population was excluded by 7AAD-staining. Tumor cells were identified by ZsGreen expression, and T cells were detected with an CD3 antibody. If tumor cells were eliminated, T cells were rechallenged with the same number of fresh tumor cells that was initially used. For coculture with MVSTs, we labeled the MVSTs with CellTrace Violet (Thermo Fisher Scientific) before setup of the coculture, to distinguish CAR or NT T cells from MVSTs. One well per condition was harvested after 3 days; the remaining MVSTs were stimulated with Epstein-Barr virus (EBV), adenovirus, and cytomegalovirus (CMV) pepmixes overnight, and intracellular cytokine staining for IFN-γ and TNF-⍺ was performed by flow cytometry.
CRISPR/Cas9-mediated T-cell receptor knockout
CRIPSR/Cas9-mediated gene editing on T cells has been extensively described elsewhere.26 In brief, CD27z-CAR T cellsTs and NT T cells were electroporated with precomplexed TrueCut Cas9 protein (Thermo Fisher Scientific) and guide RNA, which targets the ⍺-chain of the human T-cell receptor (TCR).27 After electroporation and verification of successful TCR knockout, fluorescence-activated cell sorting was used to obtain pure TCR-negative and TCR-positive CAR T-cell populations.
Cytokine release and enzyme-linked immunosorbent assays
Supernatants were harvested at selected time points after the first and second cocultures and analyzed for multiple cytokines and chemokines (GM-CSF, IFN-γ, TNF-⍺ and IL-1β, -2, 4, -5, -6, -7, -8 -10, -12 [p70], and -13), using the Multiplex Biomarker Immunoassay System (Merck Millipore, Billerica, MA), according to the manufacturer’s recommendation. Levels of soluble CD27 (sCD27) were determined with the Human CD27/TNFRSF7 DuoSet ELISA Kit (R&D Systems, Minneapolis, MN), according to the manufacturer’s recommendation.
Colony formation assay with normal HSCs
Normal HSCs were selected from the cord blood of healthy individuals by using CD34-specific microbeads (Miltenyi, Bergisch-Gladbach, Germany). After separation, CD34-positive HSCs were incubated with CAR or NT T cells, or medium only in CTL medium at an E:T ratio of 10:1 for 6 hours. After incubation, cell suspensions were plated in duplicate in MethoCult H4434 medium supplemented with recombinant cytokines (Stemcell Technologies, Vancouver, BC, Canada). After 14 days of incubation, the median total number of colonies per condition was counted using an inverted microscope. After they were counted, the cells were harvested and seeded for the second plating.
Xenograft model of AML and bioluminescent imaging
NOD.Cg-Prkdcscid IL2rgtm1Wjl/SzJ (NSG) mice (6-10 weeks of age) were purchased from The Jackson Laboratory (Bar Harbor, ME) and maintained at the Baylor College of Medicine Animal Facility. If required for tumor establishment, animals were sublethally irradiated (200 cGy). Animals received injection of tumor and T cells via the tail vein, as described in “Results.” Leukemia burden or T-cell expansion and trafficking were monitored by bioluminescent imaging (BLI; photons per second per cm2 spectral radiance), using the Xenogen In Vivo Imaging System (IVIS; Caliper Life Sciences, Hopkinton, MA). All procedures complied with the requirements of the Institutional Animal Care and Usage Committee of Baylor College of Medicine.
Statistical analysis
Prism 5 (GraphPad Software, San Diego, CA) was used for statistical analysis. For comparisons between 2 groups, we used the 2-tailed Student t test. For 3 or more groups, 2-way analysis of variance (ANOVA) with Tukey’s multiple-comparison test was used. For animal experiments, we analyzed survival from the time of T-cell injection by constructing Kaplan-Meier curves and using log-rank (Mantel-Cox) tests.
Results
Expression of CD70 on AML cell lines and AML bone marrow samples
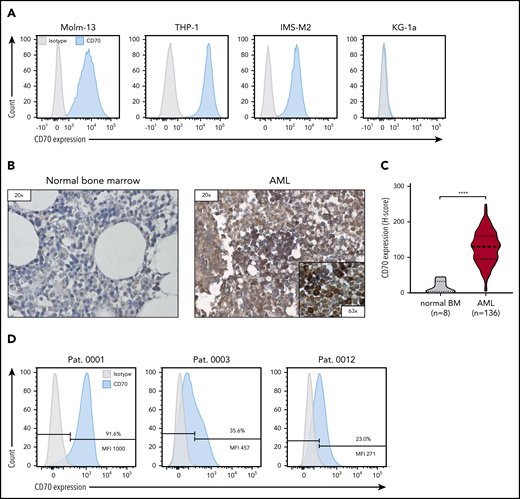
To validate CD70 as a target antigen for CAR T-cell therapy for AML, we evaluated CD70 expression on AML cell lines, bone marrow samples obtained from patients with AML, and normal bone marrow samples from patients without AML. CD70 expression was detectable in 3 of 4 AML cell lines that we tested (Molm-13, THP-1, and IMS-M2), whereas KG-1a was negative (Figure 1A). CD70 expression was significantly higher in primary bone marrow samples from 136 patients with newly diagnosed AML than in normal bone marrow (n = 8) in which CD70 was not detected (Figure 1B-C; P < .0001). By flow cytometry, CD70 expression varied in primary AML samples, both in the frequency of CD70-positive cells and expression level (Figure 1D; see supplemental Figure 1 for the gating strategy).
CD70 expression of AML cell lines and primary bone marrow samples from patients with AML. (A) Evaluation of CD70 expression in several primary AML cell lines by flow cytometry. (B) Representative images of immunohistochemical staining for CD70 expression of bone marrow samples derived from patients without AML (normal bone marrow) and patients with newly diagnosed AML. Leukemic blasts in patients with AML were identified by their morphology. (C) Comparison of CD70 expression in bone marrow samples of 136 patients with AML and 8 individuals with normal bone marrow findings by immunohistochemical staining. The H-score was used to determine the staining intensity. ****P < .0001, by unpaired Student t test. (D) Analysis of CD70 expression in bone marrow samples of patients with newly diagnosed AML by flow cytometry. Percentage of CD70-expressing cells and median fluorescence intensity (MFI) of the CD70-positive cell population are depicted.
CD70 expression of AML cell lines and primary bone marrow samples from patients with AML. (A) Evaluation of CD70 expression in several primary AML cell lines by flow cytometry. (B) Representative images of immunohistochemical staining for CD70 expression of bone marrow samples derived from patients without AML (normal bone marrow) and patients with newly diagnosed AML. Leukemic blasts in patients with AML were identified by their morphology. (C) Comparison of CD70 expression in bone marrow samples of 136 patients with AML and 8 individuals with normal bone marrow findings by immunohistochemical staining. The H-score was used to determine the staining intensity. ****P < .0001, by unpaired Student t test. (D) Analysis of CD70 expression in bone marrow samples of patients with newly diagnosed AML by flow cytometry. Percentage of CD70-expressing cells and median fluorescence intensity (MFI) of the CD70-positive cell population are depicted.
Generation and characterization of CD70-CAR T cells
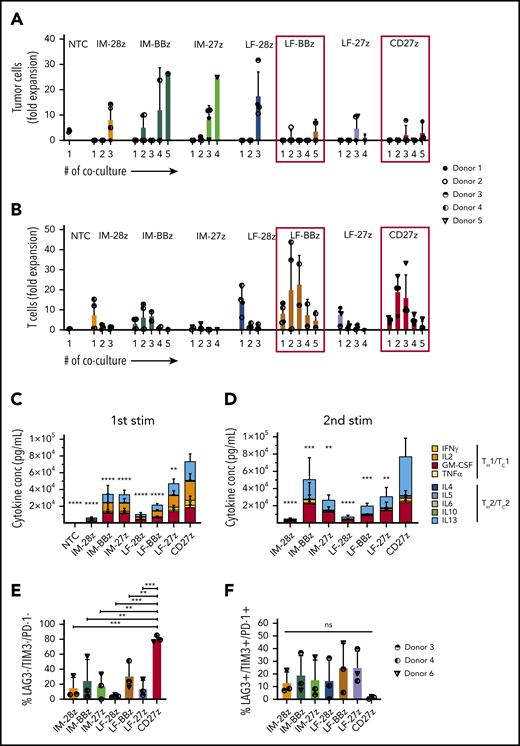
We generated a panel of 6 CD70scFv-based CAR constructs with different spacers (IM and LF) and signaling domains (CD27, CD28, or 4-1BB) (IM-28z, IM-BBz, IM-27z, LF-28z, LF-BBz, and LF-27z; supplemental Figure 2A-B). In addition, we generated 1 CAR that consisted of CD27, the CD70 receptor, linked to the CD3ζ signaling domain (CD27z; supplemental Figure 2C). CAR T cells were generated by retroviral transduction, with the median percentage of CAR-positive T cells ranging between 44.4% and 95.2% (Figure 2A-B; supplemental Figure 3A). In downstream experiments, we adjusted the number of T cells according to the percentage of CAR-positive cells. All CAR T-cell populations, except for LF-27z- and CD27z-CAR T cells, had a significantly lower viability than the NT T cells (Figure 2C); in particular, the IM spacer and the 4-1BB costimulatory domain contributed to decreased viability (Figure 2C), which translated into reduced T-cell expansion after transduction (Figure 2D). Even though CD70 expression is known to be upregulated on activated T cells, CD70-CAR T cells in contrast to NT T cells expressed minimal levels of CD70 (supplemental Figure 3B). Further phenotypic analysis revealed that all CD70scFv-based CAR T cells, except for LF-27z, induced T-cell differentiation, as judged by an increased frequency of effector memory–like (CD45RO+, CCR7−) and terminally differentiated–like (CD45RO− CCR7−) T cells in comparison with NT T cells and CD27z-CAR T cells (Figure 2E; supplemental Figure 3C). CD4/CD8 composition was not significantly different between T cells expressing the different CAR constructs (supplemental Figure 4A-B).
Generation and characterization of CD70-CAR T cells. (A) Representative histograms of CD70-CAR expression levels after transduction of activated T cells. CD70scFv-CAR T cells were detected by staining with biotin-labeled protein L, followed by staining with APC-conjugated streptavidin. CD27z-CAR expression was confirmed by detection of a truncated CD19 using an APC-conjugated CD19 specific antibody. (B) The frequency of CD70-CAR expressing T cells of 4 different donors. (C) The frequency of viable cells for CD70-CAR and NT T cells of 4 different donors, as determined by forward vs side scatter gating. (D) Fold change in the number of CD70-CAR T cells in culture during the manufacturing process without antigen stimulation. CAR T cells from 4 different donors were counted in weekly intervals. (E) Phenotypical characterization of CD70-CAR T cells by flow cytometry. The frequency of CD70-CAR T cells with a less (naive/CM T cells; left) and a more (effector memory/terminally differentiated effector memory cells reexpressing CD45RA [EM/EMRA] T cells; right) differentiated phenotype, as determined by the expression of CD45RO and CCR7 in 4 different donors. The colored bars represent the mean of results from 4 different donors, and the error bars indicate the standard deviation (SD). *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test. (F-G) A chromium-51 release assay was used to determine antigen-dependent lysis of CD70-CAR T cells against CD70-positive AML cell lines (Molm-13, THP-1) (F) and KG-1a (CD70−) cells (G) as the control. The graph shows the mean results of 3 technical replicates from 4 different donors, and the error bars indicate the SD. P values were calculated by 2-way ANOVA and are shown in the table below the graphs. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant.
Generation and characterization of CD70-CAR T cells. (A) Representative histograms of CD70-CAR expression levels after transduction of activated T cells. CD70scFv-CAR T cells were detected by staining with biotin-labeled protein L, followed by staining with APC-conjugated streptavidin. CD27z-CAR expression was confirmed by detection of a truncated CD19 using an APC-conjugated CD19 specific antibody. (B) The frequency of CD70-CAR expressing T cells of 4 different donors. (C) The frequency of viable cells for CD70-CAR and NT T cells of 4 different donors, as determined by forward vs side scatter gating. (D) Fold change in the number of CD70-CAR T cells in culture during the manufacturing process without antigen stimulation. CAR T cells from 4 different donors were counted in weekly intervals. (E) Phenotypical characterization of CD70-CAR T cells by flow cytometry. The frequency of CD70-CAR T cells with a less (naive/CM T cells; left) and a more (effector memory/terminally differentiated effector memory cells reexpressing CD45RA [EM/EMRA] T cells; right) differentiated phenotype, as determined by the expression of CD45RO and CCR7 in 4 different donors. The colored bars represent the mean of results from 4 different donors, and the error bars indicate the standard deviation (SD). *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test. (F-G) A chromium-51 release assay was used to determine antigen-dependent lysis of CD70-CAR T cells against CD70-positive AML cell lines (Molm-13, THP-1) (F) and KG-1a (CD70−) cells (G) as the control. The graph shows the mean results of 3 technical replicates from 4 different donors, and the error bars indicate the SD. P values were calculated by 2-way ANOVA and are shown in the table below the graphs. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant.
LF-BBz- and CD27z-CAR T cells have superior antitumor activity in vitro
To investigate the cytolytic capacity of our CD70-CAR T cells, we used a standardized chromium-51 release assay.28 All CAR T-cell populations mediated cytotoxicity against the CD70-positive tumor cell lines (Figure 2F; Molm-13, THP-1) but not against CD70-negative cells (Figure 2G; KG-1a). T cells expressing IM spacer–based CAR T cells with the CD28 costimulatory domain had a significantly greater cytolytic activity than all other CAR constructs. Because CAR T-cell efficacy is determined not only by its cytolytic activity, but also by the capacity to proliferate upon tumor challenge, we performed serial coculture assays, in which CD70-CAR T cells were repeatedly challenged with Molm-13 cells. Cocultures were terminated when CAR T cells failed to eliminate tumor cells or to persist after tumor cell elimination (supplemental Figure 5). T cells expressing all of the CAR constructs eliminated tumor cells and proliferated during the first coculture, but differentially lost their capacity to kill and proliferate during subsequent cocultures. LF-BBz- and the CD27z-CAR T cells eliminated tumor cells for 5 consecutive cocultures in at least 2 of the 4 donors in contrast to other CAR T-cell populations for which tumor outgrowth occurred invariably with the third coculture (Figure 3A). Increased antitumor activity of LF-BBz- and CD27z-CAR T cells was mirrored by an increased ability to expand after tumor cell challenge (Figure 3B). As it has been reported that T-cell activation leads to the release of soluble CD27 (sCD27) mainly by proteolytic events,29 we sought to determine whether this process interferes with the functionality of the CD27z-CAR. Membrane-bound CD27 (mCD27) expression before antigen stimulation was significantly higher in CD70-CAR T cells than in NT T cells. As expected, due to detection of endogenous and CAR-associated CD27, the highest levels of mCD27 were detectable in CD27z-CAR T cells, which also resulted in a significantly higher secretion of sCD27 (supplemental Figure 6A-B). Expression of mCD27 on and secretion of sCD27 by CD27z-CAR T cells remained at high levels at different time points after antigen stimulation, suggesting that CD27z-CAR T cells are not functionally impaired by interaction with the CD70 antigen (supplemental Figure 6C-D).
Functional characterization of CD70-CAR T cells after repeated antigen stimulation in vitro. CD70-CAR/NT T cells and AML cells (Molm-13) were cocultured for 5 days, and fresh Molm-13 cells were repeatedly added every 5 days thereafter. The absolute cell count of tumor and T cells at the end of each coculture was determined by flow cytometry with CountBright counting beads. (A) The absolute cell count of Molm-13 cells at the end of each coculture. (B) The absolute cell count of CD70-CAR/NT T cells at the end of each coculture. (C-D) Cytokine production by CD70-CAR and NT T cells after antigen stimulation was determined with the multiplex detection assay. Data show the quantitative determination of TH1/TC1– and TH2/TC2–associated cytokines 24 hours after the first (C) and second (D) antigen stimulation. Data are the mean of results from 4 different donors. P values indicating the statistical significance for secreted cytokines of CD27z-CAR-T cells in comparison with all other constructs and NT T cells were calculated by 2-way ANOVA. (E-F) Expression of LAG-3, TIM-3 and PD-1, markers associated with T-cell exhaustion on CD70-CAR T cells after antigen stimulation. Data are the mean frequency ± standard deviation (SD) of triple-negative (E) and triple-positive (F) T cells from 3 different donors 72 hours after the third antigen stimulation. The error bars indicate the SD. **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test.
Functional characterization of CD70-CAR T cells after repeated antigen stimulation in vitro. CD70-CAR/NT T cells and AML cells (Molm-13) were cocultured for 5 days, and fresh Molm-13 cells were repeatedly added every 5 days thereafter. The absolute cell count of tumor and T cells at the end of each coculture was determined by flow cytometry with CountBright counting beads. (A) The absolute cell count of Molm-13 cells at the end of each coculture. (B) The absolute cell count of CD70-CAR/NT T cells at the end of each coculture. (C-D) Cytokine production by CD70-CAR and NT T cells after antigen stimulation was determined with the multiplex detection assay. Data show the quantitative determination of TH1/TC1– and TH2/TC2–associated cytokines 24 hours after the first (C) and second (D) antigen stimulation. Data are the mean of results from 4 different donors. P values indicating the statistical significance for secreted cytokines of CD27z-CAR-T cells in comparison with all other constructs and NT T cells were calculated by 2-way ANOVA. (E-F) Expression of LAG-3, TIM-3 and PD-1, markers associated with T-cell exhaustion on CD70-CAR T cells after antigen stimulation. Data are the mean frequency ± standard deviation (SD) of triple-negative (E) and triple-positive (F) T cells from 3 different donors 72 hours after the third antigen stimulation. The error bars indicate the SD. **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test.
We also measured cytokine production by CD70-CAR T cells after the first 2 stimulations. CD27z- and LF-27z-CAR T cells produced the highest levels of TH1/TC1 cytokines, such as IFN-γ and TNF-α, after both stimulations (Figure 3C-D; supplemental Figures 7 and 8). To further investigate the functional differences between T cells expressing the different CD70-CAR constructs in the coculture assays, we examined the expression of exhaustion markers LAG-3, TIM-3, and PD-1, 5 days after each of the first 3 stimulations. At the end of cocultures 1 and 2, LAG-3, TIM-3, and PD-1 expression patterns did not differ significantly among the CAR T-cell populations (supplemental Figure 9A). However, after the third stimulation, the frequency of triple-negative cells (LAG-3−/TIM-3−/PD-1−) in the CD27z-CAR T-cell product was significantly higher, and the percentage of triple-positive cells was lower than that of the other CD70-CAR T cells (Figure 3E-F). CD4/CD8 composition did not differ between the CAR constructs after antigen stimulation (supplemental Figure 9B). Even though target and effector cells were not HLA matched, alloreactivity did not contribute to cytotoxicity of CD70-CAR T cells, as CD27z-CAR T cells, with and without CRISPR/Cas9-mediated disruption of the endogenous T-cell receptor, exhibited similar antitumor efficacy and T-cell expansion capacity (supplemental Figure 10).
CD27z- and LF-28z-CAR T cells have potent antitumor efficacy in vivo and against primary AML
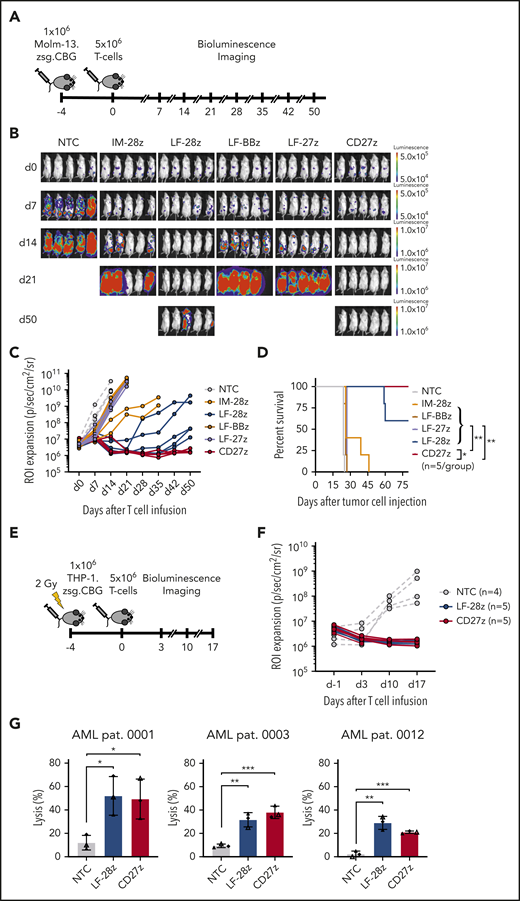
To evaluate antileukemic activity of our CD70-CAR T cells in vivo, we used a murine xenograft model, in which NSG mice received IV injections of Molm-13 cells that were genetically modified to express click beetle green luciferase and ZsGreen. On day 4, animals received a single IV dose of 5 × 106 CD70-CAR or NT T cells. Tumor growth was observed by weekly BLI (Figure 4A). Only LF-28z- and the CD27z-CAR T cells efficiently controlled leukemic growth, leading to complete leukemia remission in all mice by day 21 (Figure 4B-C), resulting in a significant survival advantage compared with mice treated with NT or other CD70-CAR T cells (Figure 4D). In contrast, IM-28z-, LF-BBz-, and LF-27z-CAR T cells had only transient antitumor activity. In vivo testing of IM-BBz- and IM-27z-CAR T cells could not be performed because of limited in vitro expansion after CAR transduction (Figure 2D).
Antileukemic activity of CD70-CAR T cells in 2 murine AML xenograft models and against primary AML blast cells. (A) The experimental setup of the Molm-13 xenograft model. CD70-CAR T cells (5 × 106) were injected 4 days after engraftment of luciferase-labeled Molm-13 cells (1 × 106). BLI was performed before T-cell injection on day 0 and weekly thereafter (n = 5 for each treatment group). (B) BLI on the indicated days (after T-cell injection) of mice with Molm-13 cell engraftment and treatment with CD70-CAR or NT T cells. (C) Quantitative analysis of bioluminescence signals for individual mice from each treatment group. (D) Kaplan-Meier survival plot of mice treated with CD70-CAR or NT T cells. The log-rank (Mantel-Cox) test was used to perform statistical analyses of survival between the treatment groups. (E) The experimental setup of the THP-1 xenograft model. After sublethal irradiation, NSG mice underwent injection of 1 × 106 luciferase-labeled THP-1 tumor cells. 4 days later, after engraftment, the mice then received an injection of 5 × 106 CD70-CAR or NT T cells and were subsequently monitored by BLI. (F) Quantitative analysis of bioluminescence signals for individual mice with THP-1 tumor cell engraftment and treatment with LF-28z-, CD27z-CAR, or NT T cells. (G) Cytotoxicity of LF-28z- and CD27z-CAR T cells Ts against AML blasts from 3 patients with AML and various levels of CD70 expression was determined with a chromium-51 release assay. Data are the mean percentage of lysis ± standard deviation at an E:T target ratio of 40:1 from 3 different donors, with 3 technical replicates. *P < .05; **P < .01; ***P < .001, by unpaired Student t test.
Antileukemic activity of CD70-CAR T cells in 2 murine AML xenograft models and against primary AML blast cells. (A) The experimental setup of the Molm-13 xenograft model. CD70-CAR T cells (5 × 106) were injected 4 days after engraftment of luciferase-labeled Molm-13 cells (1 × 106). BLI was performed before T-cell injection on day 0 and weekly thereafter (n = 5 for each treatment group). (B) BLI on the indicated days (after T-cell injection) of mice with Molm-13 cell engraftment and treatment with CD70-CAR or NT T cells. (C) Quantitative analysis of bioluminescence signals for individual mice from each treatment group. (D) Kaplan-Meier survival plot of mice treated with CD70-CAR or NT T cells. The log-rank (Mantel-Cox) test was used to perform statistical analyses of survival between the treatment groups. (E) The experimental setup of the THP-1 xenograft model. After sublethal irradiation, NSG mice underwent injection of 1 × 106 luciferase-labeled THP-1 tumor cells. 4 days later, after engraftment, the mice then received an injection of 5 × 106 CD70-CAR or NT T cells and were subsequently monitored by BLI. (F) Quantitative analysis of bioluminescence signals for individual mice with THP-1 tumor cell engraftment and treatment with LF-28z-, CD27z-CAR, or NT T cells. (G) Cytotoxicity of LF-28z- and CD27z-CAR T cells Ts against AML blasts from 3 patients with AML and various levels of CD70 expression was determined with a chromium-51 release assay. Data are the mean percentage of lysis ± standard deviation at an E:T target ratio of 40:1 from 3 different donors, with 3 technical replicates. *P < .05; **P < .01; ***P < .001, by unpaired Student t test.
We used a second xenograft model to confirm the potent antitumor activity of LF-28z- and CD27z-CAR T cells in vivo. After sublethal irradiation with 2 Gy, NSG mice received an injection of THP-1 cells, which had been genetically modified to express click beetle green luciferase and ZsGreen. Five days after tumor cell injection, the animals received LF-28z-, CD27z-, or NT T cells (Figure 4E). In contrast to NT T cells, LF-28z- and CD27z-CAR T cells rapidly eliminated THP-1 cells (Figure 4F). After having established the most potent antitumor efficacy of LF-28z- and CD27z-CAR T cells against AML cell lines, we sought to determine their cytotoxic capacity against patient-derived AML blasts. We challenged CAR T cells with blasts expressing different levels of CD70 (Figure 1D) and found that, in contrast to NT T cells, LF-28z- and CD27z-CAR T cells eliminated leukemic blasts from patients with AML, even with low CD70 expression (Figure 4G).
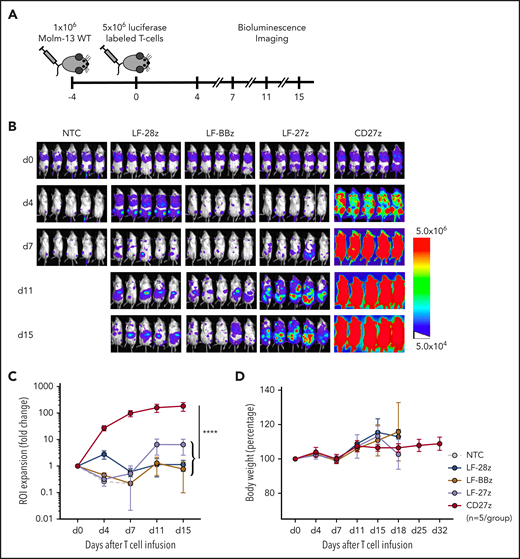
CD27z-CAR T cells induce significant T-cell expansion in vivo
To determine whether the improved antitumor activity of LF-28z- and CD27z-CAR T cells is associated with greater T-cell expansion and persistence, we used our Molm-13 xenograft model with unmodified tumor cells and CD70-CAR T cells that were genetically modified to express a GFP firefly luciferase fusion protein (Figure 5A; supplemental Figure 11A-B). Expansion of CD27z-CAR T cells was significantly greater than that of CD70scFv-based CAR and NT T cells (Figure 5B-C; P < .0001), and infusions were well tolerated, given that none of the mice had significant weight loss (Figure 5D). On day 21 after T-cell infusion, we measured the frequency of human T cells and tumor cells in the peripheral blood of mice infused with LF-28z-, LF-BBz-, and CD27z-CAR T cells. More than 95% of circulating human cells in mice infused with CD27z- and LF-28z-CAR T cells were human T cells; in contrast, most of the human cells in mice infused with LF-BBz-CAR T cells were AML blasts (supplemental Figure 11C-D).
Expansion and trafficking of CD70-CAR T cells in vivo. (A) The experimental setup. NSG mice underwent AML engraftment with 1 × 106 Molm-13 cells and injection of 5 × 106 CD70-CAR or NT T cells genetically modified to express a luciferase-GFP fusion protein 4 days later. T-cell expansion and trafficking were monitored by BLI on days 4, 7, 11, and 15 (n = 5 for each treatment group). (B) BLI of mice on the indicated days after infusion of CD70-CAR or NT T cells. (C) Quantitative analysis of BLI for each treatment group. Data are the mean ± standard deviation (SD) of 5 animals per treatment group. The area under the curve was calculated for each treatment group and compared by unpaired Student t test; ****P < .0001. (D) The body weight of treated mice was measured as an indicator of their overall condition. The change in body weight compared with the day of T-cell injection for each treatment group is depicted. Data represent the mean of each group ± SD. Results represent pooled data from 5 animals.
Expansion and trafficking of CD70-CAR T cells in vivo. (A) The experimental setup. NSG mice underwent AML engraftment with 1 × 106 Molm-13 cells and injection of 5 × 106 CD70-CAR or NT T cells genetically modified to express a luciferase-GFP fusion protein 4 days later. T-cell expansion and trafficking were monitored by BLI on days 4, 7, 11, and 15 (n = 5 for each treatment group). (B) BLI of mice on the indicated days after infusion of CD70-CAR or NT T cells. (C) Quantitative analysis of BLI for each treatment group. Data are the mean ± standard deviation (SD) of 5 animals per treatment group. The area under the curve was calculated for each treatment group and compared by unpaired Student t test; ****P < .0001. (D) The body weight of treated mice was measured as an indicator of their overall condition. The change in body weight compared with the day of T-cell injection for each treatment group is depicted. Data represent the mean of each group ± SD. Results represent pooled data from 5 animals.
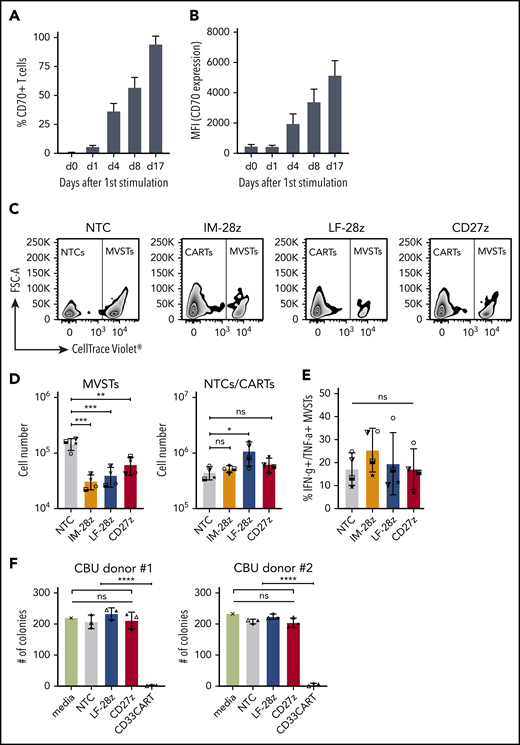
CD70-CAR T cells recognize activated virus-specific T cells, but do not recognize normal HSCs
CD70 expression on activated T cells has been reported,30 and, as expected, we observed decreased viability and poor expansion of several of our CD70-CAR T-cell populations. To determine whether CD70-CAR T cells kill activated T cells, we focused on MVSTs that recognize EBV, adenovirus, and CMV. We first measured CD70 expression by MVSTs after activation by incubating peripheral blood mononuclear cells with EBV, adenovirus, and CMV pepmixes. By 4 days after stimulation, CD70 expression was detectable on a median of 38.9% (range, 27.1% to 41.0%) of T cells (Figure 6A-B). Expression increased (percentage and intensity) over time and by day 17 after first stimulation, 97.4% (range, 89.3% to 98.3%) of cells were CD70-positive (Figure 6A-B). To investigate whether CD70-CAR T cells kill MVSTs, we cocultured IM-28z-, LF-28z-, and CD27z-CAR T cells with autologous, CellTrace Violet–labeled MVSTs for 3 days and evaluated their survival by flow cytometry. Compared with NT T cells, all CD70-CAR T-cell populations reduced the number of MVSTs significantly (Figure 6C-D). To further investigate this finding, we determined the ability of MVSTs to produce IFN-γ and TNF-⍺ in response to viral pepmixes after coculture with CD70-CAR T cells. CD70-specific CAR T cells did not lead to a significant reduction of responsive MVSTs in comparison with NT T cells (Figure 6E), suggesting that CD70-CAR T cells may impair but not completely abrogate viral immunity.
CD70-CAR T cells eliminate multivirus-specific T cells, but spare normal HSCs. (A-B) CD70 expression on MVSTs was determined by flow cytometry at different time points after stimulation. Bar graphs representing the percentage of CD70-positive cells (A) and the mean fluorescence intensity (MFI) of CD70 expression of MVSTs (B) at the indicated time points. CellTrace Violet–labeled MVSTs were cocultured with autologous CD70-CAR or NT T cells and harvested after 72 hours. The absolute cell count of both T-cell populations was determined by flow cytometry with CountBright counting beads. (C) Representative dot plots for the different T-cell groups are shown, in which autologous MVSTs can be distinguished from CAR or NT T cells by their CellTrace Violet labeling. (D) The total number of MVTSs (left graph) and CAR/NT T cells (right graph) after 3 days of coculture. (E) The percentage of MVSTs with intracellular IFN-γ and TNF-α expression after 3 days of coculture with CD70-CAR or NT T cells, followed by stimulation with EBV-, adenovirus-, and CMV-specific pepmixes. Data are the mean ± standard deviation (SD) of results from 4 different donors. (F) CD70-CAR, CD33-CAR, and NT T cells were cocultured with normal CD34-positive HSCs for 6 hours, and the cells were seeded in a standardized medium for CFU assays. The total number of colonies after the first plating were determined after 2 weeks of incubation. Data are the mean number of colonies formed by HSCs from 2 different donors after incubation with CAR or NT T cells from 3 different donors. The error bars indicate the SD. Two independent investigators counted colonies from 2 technical replicates for each condition. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test.
CD70-CAR T cells eliminate multivirus-specific T cells, but spare normal HSCs. (A-B) CD70 expression on MVSTs was determined by flow cytometry at different time points after stimulation. Bar graphs representing the percentage of CD70-positive cells (A) and the mean fluorescence intensity (MFI) of CD70 expression of MVSTs (B) at the indicated time points. CellTrace Violet–labeled MVSTs were cocultured with autologous CD70-CAR or NT T cells and harvested after 72 hours. The absolute cell count of both T-cell populations was determined by flow cytometry with CountBright counting beads. (C) Representative dot plots for the different T-cell groups are shown, in which autologous MVSTs can be distinguished from CAR or NT T cells by their CellTrace Violet labeling. (D) The total number of MVTSs (left graph) and CAR/NT T cells (right graph) after 3 days of coculture. (E) The percentage of MVSTs with intracellular IFN-γ and TNF-α expression after 3 days of coculture with CD70-CAR or NT T cells, followed by stimulation with EBV-, adenovirus-, and CMV-specific pepmixes. Data are the mean ± standard deviation (SD) of results from 4 different donors. (F) CD70-CAR, CD33-CAR, and NT T cells were cocultured with normal CD34-positive HSCs for 6 hours, and the cells were seeded in a standardized medium for CFU assays. The total number of colonies after the first plating were determined after 2 weeks of incubation. Data are the mean number of colonies formed by HSCs from 2 different donors after incubation with CAR or NT T cells from 3 different donors. The error bars indicate the SD. Two independent investigators counted colonies from 2 technical replicates for each condition. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test.
In contrast to CD33 and CD123, which are frequently used as target antigens for AML, CD70 expression is not detectable on normal human HSCs.13 Thus, to validate the safety of our CD70-CAR T cells, we examined their cytotoxicity towards normal HSCs. For this purpose, CD34-positive HSCs, isolated from cord blood units (CBU; n = 2), were cultured with T cells expressing LF-28z- or CD27z-CAR T cells, the 2 CAR T cells with the most promising antitumor activity. A third-generation, CD33-directed CAR (CD33CART; M.S., unpublished data), which is known to cause toxicity in normal hematopoiesis, NT T cells, and T-cell culture medium served as the control. After 6 hours of incubation, a serial colony-forming unit (CFU) assay was performed to determine the deleterious effect of CD70- and CD33-CAR T cells on human hematopoietic stem and progenitor cell self-renewal potential in vitro. With respect to the number of colonies counted 14 days after primary plating, we did not observe significant differences between NT and CD70-CAR T cells, whereas CD33-CAR T cells led to a significant reduction in the number of colonies (Figure 6F; P < .0001; supplemental Figure 12A), suggesting that CD70-CAR T cells do not impair normal hematopoiesis. The count after the second plating revealed a lower number of colonies only in the CD27z-CAR condition for CBU donor 1, whereas no significant differences were detectable in CBU donor 2 and any of the other conditions (supplemental Figure 12B-C).
Discussion
In this study, we designed a panel of CD70-CAR T cells and evaluated the anti-AML activity of T cells expressing these CAR T cells in vitro and in vivo. We demonstrated that CAR design influences CD70-CAR T-cell function and that a CAR based on the CD70 receptor CD27 has superior antitumor activity compared with standard, scFv-based CAR T cells.
The identification of optimal target antigens for CAR T-cell therapy for AML has been challenging, as the majority of targetable surface proteins are expressed not only on leukemic blasts, but also on healthy HSCs or on normal myeloid progenitor cells.13 CD70 has emerged as a promising target for AML-directed therapies, because, under physiological conditions, it is not expressed on healthy HSCs.13 Expression on normal tissue is limited to a subset of immune cells such as activated T and B lymphocytes and various types of antigen-presenting cells (eg, epithelial and dendritic cells in the thymic medulla). CD70 provides costimulation to T cells through binding to its ligand CD27.31,32 Both, CD70 and CD27 are frequently coexpressed in myeloid leukemia blasts, and their interaction induces a stem cell–like phenotype14 that is associated with impaired survival of patients with AML.33 In our study, we demonstrated unequivocally, in a large series of primary AML bone marrow samples, that CD70 was consistently overexpressed, extending studies conducted by other investigators.13,14
Most CD70-targeted immunotherapies so far have focused on monoclonal antibodies, which have been evaluated as monotherapy34,35 or are being evaluated in combination with other therapeutic agents (studies registered at www.clinicaltrials.gov as #NCT04023526, #NCT04150887, and #NCT04241549). Recently, Riether et al15 reported very promising results from an early-phase clinical trial that investigated the combination of cusatuzumab, a humanized monoclonal CD70-specific antibody in combination with azacytidine for the treatment of patients with AML. However, immunotherapy with monoclonal antibodies has several limitations. The injection of large amounts of recombinant antibody is frequently necessary to achieve sufficient drug levels in the serum and is associated with significant costs. In addition, because of the limited persistence, continuous or repeated infusions of the monoclonal antibodies are necessary for the duration of the treatment. The adoptive transfer of CAR-modified T cells has the potential to overcome these limitations. In this study, we preclinically evaluated a CAR T-cell–based approach for the treatment of CD70-positive AML. We designed a panel of scFv-based CAR T cells with different hinges (IM, LF) and costimulatory molecules (CD27, CD28, and 4-1BB) and 1 CAR based on the CD70 receptor CD27.18 The different CAR constructs produced striking differences in the baseline characteristics of the transduced cells with regard to viability, phenotype, and ability to proliferate. Specifically, expression of IM-based CAR T cells resulted in decreased T-cell viability and terminal differentiation, in particular when paired with the 4-1BB signaling domain. These findings suggest the presence of some baseline signaling in our designed CAR T cells, also known as tonic signaling, that depends on the structural composition of our designed CAR T cells. In this regard, others have reported that the 4-1BB costimulatory domain and structural components of the CAR, such as spacers, can modulate baseline signaling of CAR T cells.36,37 The IM-based CAR T cells were also expressed on the T-cell surface at higher levels than LF-based CAR T cells, which most likely contributed to baseline signaling as reported by others.36,38
LF-BBz- and CD27z-CAR T cells had superior effector function in a repeat stimulation assays as judged by their sustained cytolytic activity and ability to proliferate. Phenotypic analysis revealed that CD27z-CAR T cells expressed proteins associated with T-cell exhaustion (LAG-3, TIM-3, and PD-1) at significantly lower levels after the third stimulation than T cells expressing all other CAR T cells, most likely because the CD27z-CAR T cells induced less differentiation than scFv-based CAR T cells with CD28 and 4-1BB costimulatory domains. Of interest, LF-27z-CAR T cells, which, before antigen stimulation, had an indistinguishable phenotype to CD27z-CAR T cells, also expressed higher levels of LAG-3, TIM-3, and PD-1 after the third stimulation. Thus, whereas CD27 costimulation has been shown to improve CAR T-cell function,39 in our system, CD27 costimulation by itself was insufficient to prevent T-cell exhaustion.
In vivo, only LF-28z- and CD27z-CAR T cells had significant antitumor activity in 2 different AML xenograft models. Although this finding was expected for CD27z-CAR T cells based on our coculture assay, which mimics chronic antigen exposure, it was unexpected for CD70scFv-based CAR T cells because LF-BBz-CAR T cells had a greater ability to sequentially kill tumor cells than LF-28z-CAR T cells in vitro. However, LF-28z-CAR T cells showed robust expansion after the first stimulation in vitro in comparison with LF-BBz-CAR T cells, suggesting that initial CAR T-cell expansion is important for tumor control in our AML xenograft models. Although LF-28z- and CD27z-CAR T cells had similar anti-AML activity, the CD27z-CAR T cells induced greater T-cell expansion and persistence in vivo than the LF-28z-CAR T cells. This result is consistent with our in vitro repeat stimulation assay and findings by others that CD28-based CAR T cells have limited persistence in preclinical xenograft models.40 Several groups have generated CD70-CAR T cells, using CD27 as the antigen recognition domain to target CD70-positive solid tumors, and have shown significant antitumor activity in preclinical models.18,41-43 To our knowledge, our study is the first to systematically compare the effector function of CD70scFv- and CD27-based CAR T cells. In addition, it highlights that it is critical to perform a comprehensive in vitro and in vivo analysis.
CD70-CAR T cells did not recognize and eliminate normal HSCs in CFU assays, suggesting a clear advantage over CD33- and CD123-CAR T cells, which produce significant on-target/off-tumor toxicity to HSCs and myeloid progenitor cells.44,45 However, we observed that CD70-CAR T cells recognized activated CD70 expressing MVSTs in vitro. Even though it is unclear at this time whether this finding indicates that targeting CD70 would put patients at higher risk for infections caused by T-cell–specific immune defects, it has been shown that mice infused with murine CD70-CAR T cells could still mount antigen-specific T-cell responses,43 and treatment with the CD70 monoclonal antibody ARGX-110 was well tolerated in a cohort of 26 heavily pretreated cancer patients, without infectious complications.34 Of note, another T-cell activation marker, CD30, is also expressed on activated VSTs; however, patients receiving CD30-CAR T cells for the treatment of CD30-positive lymphomas are not at higher risk for viral infections.46 A clinical study (registered on www.clinicaltrials.gov as #NCT02830724) with CD70-CAR T cells for solid tumors is already accruing patients, but safety data from this trial has not yet been published. Nevertheless, based on our findings, we believe that monitoring T-cell–specific immune responses in early-phase clinical studies with CD70-CAR T cells is advisable. In addition, CD70 expression on activated T cells may cause fratricide of CD70-CAR T cells, thereby potentially interfering with the production of CAR T cells ex vivo and/or their in-patient expansion after antigen stimulation by tumor cells. However, as we were able to detect robust proliferation of CD27z-CAR T cells in the murine xenograft model, our data suggest that decreased viability and expansion capacity of some CD70-CAR T-cell products were attributable rather to differences in the structural composition of the CAR than to fratricide induced by CD70 expression of CAR T cells.
In summary, CD70 is a promising target antigen for CAR T-cell therapy for AML. We designed a panel of CD70 CAR T cells, and T cells expressing CAR T cells based on the CD70 receptor (CD27z-CAR T cells) emerged as the most beneficial CAR T-cell product, as shown by their anti-AML activity and ability to expand and persist. Thus, our results warrant future early-phase clinical testing of CD27z-CAR T cells in patients with CD70-positive AML.
Original data are available by e-mail request to the corresponding author.
Presented in abstract form at the 61st annual meeting of the American Society of Hematology, Orlando, FL, 7 December 2019.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This work was supported by National Institutes of Health, National Cancer Institute grants P50 CA126752 and P01 CA094237 (C.M.R.), Leukemia and Lymphoma Society grant 7001-19 (C.M.R.), and Cancer Prevention and Research Institute of Texas grant RP160283 (Baylor College of Medicine Comprehensive Cancer Training Program) (S.S.).
Authorship
Contribution: T.S., K.P., S.S., D.S., Q.C., L.A., and C.S. performed the experiments; T.S., S.G., and C.M.R. analyzed and interpreted the data; T.S., S.G., and C.M.R. designed the research and wrote the paper; and B.O., M.S., and C.M.-T. contributed to the interpretation of the results.
Conflict-of-interest disclosure: T.S. has received support of educational activities from Pfizer and AbbVie and has served on the Data and Safety Monitoring Board and the Science Advisory Board (DSMB/SAB) of Matterhorn Biosciences AG and Takeda. C.M.R. holds patents and patent applications in the fields of T-cell and gene therapy for cancer; has received research support from TESSA Therapeutics; and has served on the DSMB/SAB of TESSA Therapeutics, CellGenix, and Marker Therapeutics. S.G. holds patents and patent applications in the fields of T-cell and gene therapy for cancer; has received research support from TESSA Therapeutics; has served on the DSMB/SAB of Immatics and Tidal; and has been a consultant within the past 2 years for Merck and ViraCyte. C.M.-T. has received grants and/or provisions of investigational medicinal products (IMPs) from Pfizer, Daiichi Sankyo, and BiolineRx; has received research funding from Deutsche Forschungsgemeinschaft (DFG), Deutsche Krebshilfe, German Ministry of Education and Science (BMBF), Wilhelm-Sander-Stiftung, Jose-Carreras-Stiftung, and Bayer; and has served on the DSMB/SAB of Pfizer and Janssen-Cilag. M.S. has received research funding from Apogenix, Hexal, and Novartis; has served on the DSMB/SAB of MSD; is a cofounder of and shareholder in Tolerogenixx; and has received financial support of educational activities and conference attendance from Kite. C.S. has received reimbursement for travel, accommodations, and expenses from Celgene, PharmaMar, and Pfizer and has served on the DSMB/SAB of AbbVie, Jazz Pharmaceuticals, Pfizer, Novartis, and Takeda. The remaining authors declare no competing financial interests.
Correspondence: Tim Sauer, Department of Hematology and Oncology, University Hospital of Heidelberg, Im Neuenheimer Feld 410, 69120 Heidelberg, Germany; e-mail: tim.sauer@med.uni-heidelberg.de.

![Generation and characterization of CD70-CAR T cells. (A) Representative histograms of CD70-CAR expression levels after transduction of activated T cells. CD70scFv-CAR T cells were detected by staining with biotin-labeled protein L, followed by staining with APC-conjugated streptavidin. CD27z-CAR expression was confirmed by detection of a truncated CD19 using an APC-conjugated CD19 specific antibody. (B) The frequency of CD70-CAR expressing T cells of 4 different donors. (C) The frequency of viable cells for CD70-CAR and NT T cells of 4 different donors, as determined by forward vs side scatter gating. (D) Fold change in the number of CD70-CAR T cells in culture during the manufacturing process without antigen stimulation. CAR T cells from 4 different donors were counted in weekly intervals. (E) Phenotypical characterization of CD70-CAR T cells by flow cytometry. The frequency of CD70-CAR T cells with a less (naive/CM T cells; left) and a more (effector memory/terminally differentiated effector memory cells reexpressing CD45RA [EM/EMRA] T cells; right) differentiated phenotype, as determined by the expression of CD45RO and CCR7 in 4 different donors. The colored bars represent the mean of results from 4 different donors, and the error bars indicate the standard deviation (SD). *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant, by unpaired Student t test. (F-G) A chromium-51 release assay was used to determine antigen-dependent lysis of CD70-CAR T cells against CD70-positive AML cell lines (Molm-13, THP-1) (F) and KG-1a (CD70−) cells (G) as the control. The graph shows the mean results of 3 technical replicates from 4 different donors, and the error bars indicate the SD. P values were calculated by 2-way ANOVA and are shown in the table below the graphs. *P < .05; **P < .01; ***P < .001; ****P < .0001; ns, not significant.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/138/4/10.1182_blood.2020008221/6/m_bloodbld2020008221f2.png?Expires=1769144031&Signature=LI3mC1-3-B2LSOzFCF5nVF1kNm9RBBr2srJnfvDlt6n1RsoG9zL4HjrfUn~nRId2kGy0BwSQAiToUs5-VR-z9uCRbO7w0T1xrbCfx2YBkTsNmOmj2Evr2cKuNx2OoE7rqrHEoW~238lW9INOw3zljTa4BS4OOTe7gWQ5tjF-kMaP3gz-u4pna4GzMbXAeOcYc05l1V6uZNYPfWmT3kH5hSKMxQGRT~FwkeuuK4eGaHyHK0sEfeeu1AGjoHGnNKSY6PqE7Vv1qIVHGvRnwoZjw5A4MTq~-9O-lMa3t3KvRc9TJY92ZJOGRyHR0UEEQQ-s4yLW6b4OwKqLLDTAP4k6Dg__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal