In this issue of Blood, Bazarbachi et al1 present results from whole genome sequencing (WGS) and transcriptome sequencing analysis of immunoglobulin M (IgM) multiple myeloma (MM) that support a pregerminal center origin and set it apart from non-IgM MM and Waldenström macroglobulinemia (WM) as a genomically distinct disease. They further show actionable findings that suggest tailored therapeutic options for patients with this rare disease, which accounts for <0.5% of all cases of myeloma.
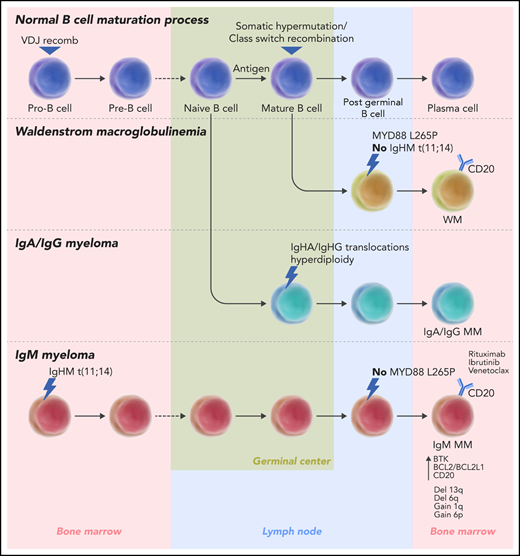
MM is a malignancy of plasma cells that is characterized by complex genetic alterations, such as hyperdiploidy and chromosomal translocations juxtaposing oncogenes such as CCND1 and MMSET with the immunoglobulin super enhancers.2 B-cell development is characterized by different molecular processes resulting in antibody diversity, including V(D)J recombination, which occurs early at the pro–B-cell stage in the bone marrow, and class switch recombination, which takes place in the germinal center.3 Because these 2 events involve different regions of the IGH locus, analysis of the immunoglobulin breakpoints in MM allows us to determine at what stage the disease-initiating events occur. Most MM translocation breakpoints are found in the IGH switch region, suggesting that the cell of origin of MM is a post–germinal center B cell. However, ∼20% of CCND1 translocations, t(11;14), are generated through V(D)J recombination, indicating that they occur at the pro–B-cell stage in the bone marrow.4 In their study, Bazarbachi et al performed comprehensive profiling of IgM MM, non-IgM MM, and WM using WGS and RNA-sequencing (RNA-Seq), which showed that all of the analyzed IgM MM cases harbor t(11;14) and that most of them have evidence of breakpoints in the V(D)J region, which implies an earlier onset of the disease during the early stages of B-cell maturation in the bone marrow. Therefore, they propose that IgM myeloma is a genomically distinct entity from the IgA and IgG types, as well as from WM, which is another post–germinal center-derived lymphoproliferative disease that is characterized by IgM monoclonal gammopathy (see figure). Our recent integrative analysis of WGS and RNA-Seq from >600 patients with MM5 showed significantly higher levels of IgM in the blood of patients with t(11;14), thus providing support for the association described in their study.
In normal B-cell development, immunoglobulin genes undergo V(D)J recombination in the bone marrow and somatic hypermutation and class switch recombination in the lymph nodes. All of the IgM MM cases analyzed by Bazarbachi et al harbor a t(11;14) translocation; 60% of them have evidence of the event occurring at the pro–B-cell stage during V(D)J recombination. In non-IgM MM, most translocations occur in the germinal center; only 25% of t(11;14) has evidence of earlier onset in the bone marrow. WM is further distinguished from IgM MM because of the lack of t(11;14) and the presence of the MYD88 L265P mutation in the majority of cases. Bazarbachi et al also identify upregulated markers in IgM MM, such as BTK, BCL2/BLC2L1, and CD20, suggesting potential therapeutic options specific for this disease. germ, germinal center; recomb, recombination.
In normal B-cell development, immunoglobulin genes undergo V(D)J recombination in the bone marrow and somatic hypermutation and class switch recombination in the lymph nodes. All of the IgM MM cases analyzed by Bazarbachi et al harbor a t(11;14) translocation; 60% of them have evidence of the event occurring at the pro–B-cell stage during V(D)J recombination. In non-IgM MM, most translocations occur in the germinal center; only 25% of t(11;14) has evidence of earlier onset in the bone marrow. WM is further distinguished from IgM MM because of the lack of t(11;14) and the presence of the MYD88 L265P mutation in the majority of cases. Bazarbachi et al also identify upregulated markers in IgM MM, such as BTK, BCL2/BLC2L1, and CD20, suggesting potential therapeutic options specific for this disease. germ, germinal center; recomb, recombination.
Although Bazarbachi et al provide evidence that the cell of origin of IgM MM is a pro-B cell, ∼25% of analyzed patients with IgM MM in their study do not show a V(D)J origin; conversely, ∼25% of t(11;14) cases in non-IgM MM have evidence of V(D)J breakpoints, raising the hypothesis that these may share the same origin as IgM MM. More studies on larger patient cohorts will be necessary to further elucidate the mechanisms of origin of IgM and non-IgM MM.
Being able to distinguish IgM myeloma from other IgM plasma cell disorders, such as WM, at presentation is crucial for proper therapeutic management.6 The analysis by Bazarbachi et al shows that the hallmark mutation of WM, MYD88 L265P, is not found in IgM MM and that t(11;14), which is a defining feature of IgM MM, is not present in any WM cases. Copy number lesions, such as deletion of 13q and 6q and gain of 6p, are more frequently associated with IgM MM compared with WM. Of note, the deletion of 6q is observed in a subset of WM cases and is associated with the progression from IgM monoclonal gammopathy of undetermined significance (MGUS) to WM.7 Future studies will be needed to identify the secondary genomic “hits” in IgM MGUS to distinguish patients who will progress to IgM MM vs WM vs non-Hodgkin lymphoma.
Lastly, the analysis of the transcriptome and surface markers provides clues for deploying therapeutics in a tailored manner in IgM MM. For example, immunotherapeutics, such as monoclonal antibodies, bispecifics, or chimeric antigen receptor T-cell treatment, may be useful in IgM MM, as well as in non-IgM MM, as a result of the shared expression of CD38, BCMA, and SLAMF7. Furthermore, the higher BCL2/BCL2L1 and BCL2/MCL1 ratios observed in IgM MM vs non-IgM MM suggest that there may be specific greater sensitivity to BCL2 inhibition in this subset of patients. Expression of BTK and CD20, which are typical findings of WM, is also seen in IgM MM, raising the possibility of targeted trials of BTK inhibitors or anti-CD20 monoclonal antibodies for these patients. These exciting findings will need to be validated in prospective clinical trials.
Conflict-of-interest disclosure: S.P. has acted as a consultant for Foundation Medicine, Inc and has received research support from Amgen Inc/Pfizer Inc Partnership, Celgene Corporation, and Karyopharm Inc. A.L. declares no competing financial interests.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal