[Introduction]
AL amyloidosis is caused by the deposition of abnormally folded clonal immunoglobulin (IG) light chains (LCs, λ:κ = 3:1) made by malignant plasma cells in the bone marrow (BM), which leads to multi-organ dysfunction, often involving the heart, kidney, liver, skin, and nerves. However, little is known about what regulates organ tropism of amyloid deposition in this disease. In addition, no study has analyzed the repertoire of IG germlines of plasma cells in the BM in AL amyloidosis using next generation sequencing (NGS). In this study, we aimed to identify the clonal composition of IG λ light-chain variable region (IGLV) genes in BM cells in patients with AL amyloidosis using NGS.
[Material and method]
BM cells were obtained at diagnosis from 38 patients with AL amyloidosis and those with other plasma cell disorders: multiple myeloma (MM, n = 7), and monoclonal gammopathy of undetermined significance (MGUS, n = 11) with λ-type monoclonal paraprotein. Seven normal control (NC) patients had either immune thrombocytopenia or malignant lymphoma without BM invasion. Genomic DNA was extracted from the BM mononuclear cells preserved in LABO Banker1 or BM clots in O.C.T compound using QIAamp DNA Blood Mini kit. The IGLV1 and IGLV2 genes were amplified by polymerase chain reaction using a 5′ primer for the IGLV1/2 framework 3 (FR3) region and 3′ consensus primers for the IGLJ1/2/3 joining regions. Multiple samples were pooled, and paired-end 2 × 250 base pair sequencing reactions were performed using an Illumina MiSeq sequencer and then analyzed by an open-source program called Vidjil. All subjects provided written informed consent to participate in the study, in accordance with the Declaration of Helsinki. This study was approved by the ethics committee of the Chiba University Graduate School of Medicine and Japanese Red Cross Medical Center.
[Results]
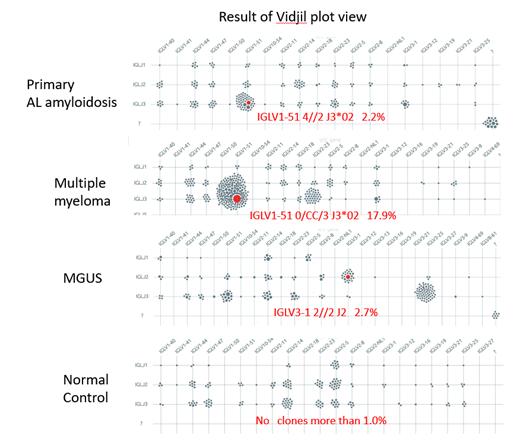
Clinical and laboratory features of 38 patients with AL amyloidosis were as follows: primary AL amyloidosis (n = 31); 15 and 20 patients had cardiac and renal dysfunctions, respectively, and secondary AL amyloidosis with MM (n = 7); 4 and 1 patient had cardiac and renal dysfunctions, respectively. In patients with AL amyloidosis, the median plasma cell count in BM aspirate smears was 3.3% (0.1%-50.4%), and the median difference in involved and uninvolved light chains (dFLC) was 104.5 mg/L (28.5mg/L -2673.3mg/L). Representative results of the Vidjil analysis in NC, MGUS, AL amyloidosis, and MM are shown in Figure 1. The most abundant IGLV gene accounted for not >1% of the reads, and there was no dominant germline in NC samples. Therefore, we defined the dominant clone as >1% of IG germlines in plasma cells. According to this definition, clonal IG germlines were found in 27 of 31 patients with primary AL amyloidosis (87%), 5 of 7 with secondary AL amyloidosis (71%), 7 of 7 with MM (100%), and 8 of 11 with MGUS (73%). However, the size of clones in AL amyloidosis (median 3.1%, 0.38%-14.3%) was significantly smaller than that in MM (median 17.8%, 2.2%-17.9%) (P<0.001), and similar to that in MGUS (median 3.8%, 0.4%-32.0%). Importantly, in patients with AL amyloidosis, the dFLC and involved/uninvolved FLC ratio was not correlated with the clonal size of plasma cells in our repertoire analysis using NGS, suggesting that small malignant clones of plasma cells may secret FLC and cause LC depositions in AL amyloidosis. Regarding IGLV germline usage, IGLV1-51 was the most frequent repertoire in AL amyloidosis with heart dysfunction (7 of 16 cases) and renal dysfunction (7 of 21 cases). No relationship between the IGLV germlines and organ tropisms was observed.
[Conclusion]
We successfully identified the clonal composition of IGLV genes in the BM of most patients with AL amyloidosis using NGS, according to the differences in the V and J region recombination and CDR3 sequences using the Vidjil program. In AL amyloidosis, the clonal size of plasma cells in the BM is small and small malignant clones of plasma cells may secret FLC and cause LC depositions in AL amyloidosis.
Suzuki:Ono: Research Funding; BMS: Honoraria, Research Funding; Takeda: Honoraria; Janssen: Honoraria; Celgene: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal