Introduction: New Diffuse Large B-cell (DLBCL) treatments remain a clinical need despite the success of rituximab combined with CHOP chemotherapy (RCHOP), which results in durable responses in 60-70% of patients. Those refractory to, or who relapse following, first-line therapy have a very poor outcome, with only 20% surviving beyond 5 years. Rationally-targeted frontline strategies are needed, especially for those with high-risk disease. Significant advances have been made in the genomic classification of DLBCL, but none have impacted the design of phase III trials for untreated DLBCL patients. Building on new genetic profiling studies to personalize clinical treatment, an NCI initiative, could allow clinicians to add targeted therapies to the RCHOP backbone based on individual tumor signatures. The phase II ECOG-ACRIN1412 trial, which compared RCHOP combined with lenalidomide (R2CHOP) versus RCHOP showed significantly superior event free survival (EFS) and overall survival benefits for those treated with R2CHOP. Herein, we report the profile of a high-risk ABC/non-GCB subset of DLBCL driven by genomic alterations in inflammatory genes that are susceptible to front-line R2CHOP, but continue to experience poor outcome with RCHOP alone.
Methods: We studied a total of 196 DLBCL patients. 47 were treated with R2CHOP from an investigator-initiated, open-label, single-arm phase II study (NCT00670358). 149 were newly diagnosed DLBCL cases treated with RCHOP, or R-immunochemotherapy (herein called RCHOP), and followed prospectively through the Molecular Epidemiology Resource (MER) of the University of Iowa/Mayo Clinic SPORE that served as a contemporary cohort. Patients from each treatment group were divided based on their event free survival at 24 months (EFS24). DNA alterations within these populations were identified through whole exome sequencing (WES). Variants were analyzed for their presence in EFS24 achievement or failure groups in both RCHOP and R2CHOP. Both tumor and germline sequencing was performed for 47/47 R2CHOP cases and 49/149 RCHOP cases. Gene expression data from the PanCan panel of 730 B-cell-related genes was analyzed to determine gene expression profiles characteristic of the high-risk/R2CHOP-profile on 59 available non-GCB DLBCL cases (45 RCHOP; 14 R2CHOP). A two-sided comparative marker analysis T statistic test was applied to assess what genes displayed differential expression based on achieving EFS24 in both R2CHOP and RCHOP populations. Positive values represented associations with achieving EFS24 and negative values were associated with cases of EFS24 failure.
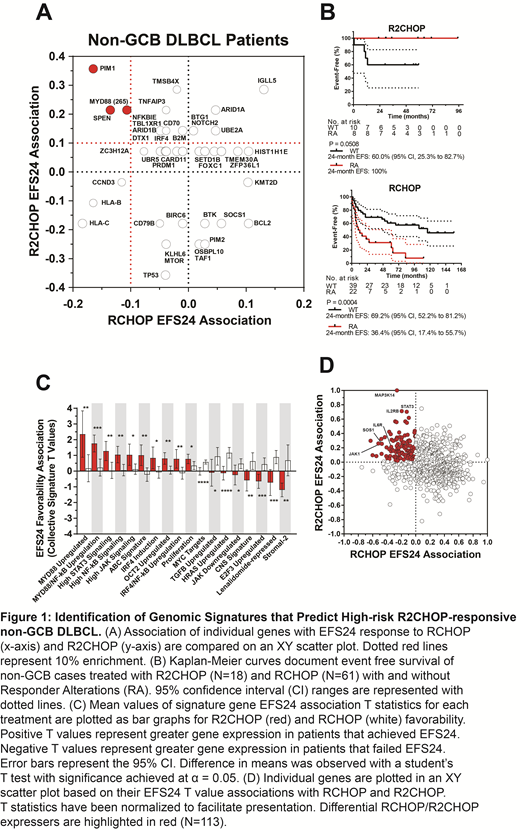
Results: Three genes were enriched in the RCHOP cases that failed EFS24 but achieved EFS24 with R2CHOP among non-GCB patients: PIM1, SPEN, and MYD88 (L265P), herein referred to as Responder Alterations. In R2CHOP cases, patients with a Responder Alteration had a better overall EFS (P = 0.051) compared to wild type patients. In contrast, RCHOP treated patients with a Responder Alteration in their tumor had a significantly worse overall EFS (P = 0.0004) compared to patients without a mutation. Together, PIM1, SPEN, or MYD88 (L265P) mutations were present in 38.0% (30/79) of all non-GCB cases. Collective R2CHOP and RCHOP EFS24 differential gene expression T values were significantly different for 18 previously-defined signatures. The R2CHOP cases that achieved EFS24 were enriched for genes involved in cell cycle, JAK-STAT, cytokine signaling, and NF-κB pathways based on these signatures and ontology analyses. Lastly, cases with WES and PanCan data were analyzed together to observe differential gene expression patterns between cases with and without signature mutations. These data suggest that R2CHOP disrupts tumors reliant on IRF4, NF-κB, and STAT transcription factors, leading to a loss of proliferative feedback systems.
Conclusions: Our combined analysis of DNA and RNA across R2CHOP and RCHOP treatment cohorts identifies a high-risk non-GCB phenotype that encompasses approximately 38% of non-GCB patients, is capable of sustaining JAK-STAT and NF-κB signaling, and is sensitive to R2CHOP. Our study lays the groundwork for a precision therapy approach in DLBCL in which DNA or RNA profiles can be used to identify patients early in treatment who may not benefit from the current standard of care, RCHOP, and who would benefit from the addition of lenalidomide or other targeted agents.
Gandhi:Celgene Corporation: Employment, Equity Ownership, Patents & Royalties. Ansell:LAM Therapeutics: Research Funding; Seattle Genetics: Research Funding; LAM Therapeutics: Research Funding; Seattle Genetics: Research Funding; Regeneron: Research Funding; Bristol-Myers Squibb: Research Funding; Affimed: Research Funding; Trillium: Research Funding; Trillium: Research Funding; Regeneron: Research Funding; Bristol-Myers Squibb: Research Funding; LAM Therapeutics: Research Funding; Trillium: Research Funding; Affimed: Research Funding; LAM Therapeutics: Research Funding; Bristol-Myers Squibb: Research Funding; Trillium: Research Funding; Affimed: Research Funding; Bristol-Myers Squibb: Research Funding; Trillium: Research Funding; LAM Therapeutics: Research Funding; Bristol-Myers Squibb: Research Funding; Regeneron: Research Funding; Affimed: Research Funding; Seattle Genetics: Research Funding; Mayo Clinic Rochester: Employment; Regeneron: Research Funding; Seattle Genetics: Research Funding; Mayo Clinic Rochester: Employment; Seattle Genetics: Research Funding; LAM Therapeutics: Research Funding; LAM Therapeutics: Research Funding; LAM Therapeutics: Research Funding; Regeneron: Research Funding; Regeneron: Research Funding; Seattle Genetics: Research Funding; Seattle Genetics: Research Funding; Trillium: Research Funding; Trillium: Research Funding; Affimed: Research Funding; Affimed: Research Funding; Mayo Clinic Rochester: Employment; Mayo Clinic Rochester: Employment; Mayo Clinic Rochester: Employment; Seattle Genetics: Research Funding; Mayo Clinic Rochester: Employment; Bristol-Myers Squibb: Research Funding; Bristol-Myers Squibb: Research Funding; Affimed: Research Funding; Regeneron: Research Funding; Regeneron: Research Funding; Bristol-Myers Squibb: Research Funding; Regeneron: Research Funding; Seattle Genetics: Research Funding; Trillium: Research Funding; Mayo Clinic Rochester: Employment; Mayo Clinic Rochester: Employment; Trillium: Research Funding; Affimed: Research Funding; Mayo Clinic Rochester: Employment; Bristol-Myers Squibb: Research Funding; Affimed: Research Funding; LAM Therapeutics: Research Funding. Cerhan:Celgene: Research Funding; NanoString: Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees. Nowakowski:Curis: Research Funding; Bayer: Consultancy, Research Funding; Celgene: Consultancy, Research Funding; F. Hoffmann-La Roche Ltd: Research Funding; Genentech, Inc.: Research Funding; MorphoSys: Consultancy, Research Funding; NanoString: Research Funding; Selvita: Membership on an entity's Board of Directors or advisory committees. Novak:Celgene Coorperation: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal