Key Points
A validated 3-miRNA classifier can effectively predict progression from early- to advanced-stage MF and survival at time of diagnosis.
This classifier outperforms existing clinical prognostic factors and paves the way for implementation of personalized treatment in MF.
Abstract
Mycosis fungoides (MF) is the most frequent form of cutaneous T-cell lymphoma. The disease often takes an indolent course, but in approximately one-third of the patients, the disease progresses to an aggressive malignancy with a poor prognosis. At the time of diagnosis, it is impossible to predict which patients develop severe disease and are in need of aggressive treatment. Accordingly, we investigated the prognostic potential of microRNAs (miRNAs) at the time of diagnosis in MF. Using a quantitative reverse transcription polymerase chain reaction platform, we analyzed miRNA expression in diagnostic skin biopsies from 154 Danish patients with early-stage MF. The patients were subdivided into a discovery cohort (n = 82) and an independent validation cohort (n = 72). The miRNA classifier was built using a LASSO (least absolute shrinkage and selection operator) Cox regression to predict progression-free survival (PFS). We developed a 3-miRNA classifier, based on miR-106b-5p, miR-148a-3p, and miR-338-3p, which successfully separated patients into high-risk and low-risk groups of disease progression. PFS was significantly different between these groups in both the discovery cohort and the validation cohort. The classifier was stronger than existing clinical prognostic factors and remained a strong independent prognostic tool after stratification and adjustment for these factors. Importantly, patients in the high-risk group had a significantly reduced overall survival. The 3-miRNA classifier is an effective tool to predict disease progression of early-stage MF at the time of diagnosis. The classifier adds significant prognostic value to existing clinical prognostic factors and may facilitate more individualized treatment of these patients.
Introduction
Mycosis fungoides (MF) is an extranodal non-Hodgkin lymphoma representing the most prevalent entity among cutaneous T-cell lymphomas (CTCLs).1 Its incidence rate is 4.1 per million person-years.2 MF is characterized by expansion of malignant T cells in a chronic inflammatory environment in the skin.3 MF most often presents as an early-stage disease with limited erythematous skin patches and/or plaques.4 Early disease may exhibit an indolent behavior and have a favorable prognosis.5 However, the skin lesions spread and evolve into overt tumors or generalized erythroderma in approximately one-third of the patients, developing advanced disease that may disseminate to the lymph nodes, blood, and internal organs.5,6 In the advanced stages, the disease becomes increasingly aggressive and associated with an increase in mortality.5,6 The mortality is highest during the first 5 years after diagnosis, indicating that a subset of patients have an aggressive form of MF.7 It remains challenging at the time of diagnosis to predict in which patients the disease will progress to the advanced stages. Current practice involves clinical monitoring and aggressive treatment at signs of disease progression; and existing treatment guidelines are based on the disease stage of MF in general rather than the individual patient’s risk of disease progression.8 Prognostic stratification of patients with early-stage MF therefore seems warranted. Genetic aberrations and epigenetic changes have been associated with disease progression and survival in patients with MF, but independently validated prognostic molecular classifiers have not yet been developed.9-11 Recently, a prognostic clinical index, cutaneous lymphoma international prognostic index (CLIPi), was proposed to classify early- and late-stage MF into low, intermediate, and high risk of disease progression and reduced survival.12 CLIPi is based on a combination of clinical variables that were associated with disease progression and/or reduced survival in a multivariable analysis in a large cohort of patients with MF and Sézary syndrome.5
In some solid cancers and hematological malignancies, prognostic microRNA (miRNA) classifiers have substantially improved the prognostic value of clinically based prognostic markers.13,14 miRNAs are small noncoding RNAs aberrantly expressed in cancer, including MF. It was recently proposed that miRNAs are key players in the pathogenesis of MF and could serve as a diagnostic tool in these patients.15,16 Although prognostic miRNA classifiers have not been previously reported, preliminary data on a limited number of patients have hinted that miRNAs may have a role in progressive MF.17,18 We therefore hypothesized that selected miRNAs can serve as a prognostic classifier predicting disease progression in patients at the time of diagnosis.
In the present nationwide study, we identified 154 patients diagnosed with early-stage MF. First, we identified the specific biopsy initially used to establish the histological diagnosis of MF in each patient. Then, we developed and validated a prognostic 3-miRNA classifier discriminating between patients at high or low risk of disease progression at the time of diagnosis. This prognostic miRNA classifier may substantially improve the existing clinical prognostic markers in patients with early-stage MF.
Materials and methods
Patient cohorts
We used the Danish Cancer Registry to identify patients diagnosed with MF in Denmark in the period from 1981 to 2013, and the Danish National Registry of Patients to geographically subdivide these patients into 2 equal, independent cohorts. By individual-level linkage to the medical records, we identified and collected the specific skin biopsy that established the diagnosis of early-stage MF for the first time. From the patient files, we extracted important clinical variables with complete and long-term follow-up. We excluded patients with advanced disease, defined as stage ≥ IIB at the time of diagnosis.
In total, we collected 174 formalin-fixed, paraffin-embedded (FFPE) skin biopsies from 154 patients with early-stage MF (stage < IIB) and 20 age- and sex-matched healthy controls. The biopsies were generously provided by departments of pathology in Denmark. The patients were subdivided into a discovery cohort of 82 patients diagnosed with early-stage MF in the western part of Denmark and an independent validation cohort consisting of 72 patients diagnosed with early-stage MF in the eastern part of Denmark. The early MF diagnosis (stage IA-IIA) was confirmed by clinical as well as immuno- and histopathological findings referenced in the patient files.19,20 The patients were staged according to the International Society for Cutaneous Lymphomas/European Organization of Research and Treatment of Cancer proposal19 and categorized according to whether their disease progressed to advanced MF (stage ≥ IIB) or remained in the early stages (stage < IIB) during follow-up. Patients categorized as having nonprogressive disease were followed for at least 5 years. Moreover, each patient’s clinical characteristics were collected from the patient files. These characteristics included age, sex, clinical stage, T stage, the CLIPi risk score for early MF,12 treatment upon time of diagnosis, date of death, and whether death was MF-related.
Disease progression was defined as progression from the early stages (stage IA-IIA) to the advanced stages of MF (stage IIB-IVB), because progression to advanced MF significantly impacts the prognosis.5 Progression-free survival (PFS) was defined as time from the diagnosis to progression and/or disease-specific death, and overall survival (OS) was defined as time from the diagnosis to death from any cause.
The study was approved by the local ethical committee (1-10-72-91-13) and the Data Protection Agency (Datatilsynet 1-16-02-478-15).
Procedures
First, we collected and processed the biopsies from patients with MF in the discovery cohort and the biopsies from the healthy controls. The data from these patients were examined and the classifier identified. Subsequently, we collected and processed biopsies from the independent validation cohort. The RNA extraction and quantitative reverse transcription polymerase chain reaction (qRT-PCR) analysis were performed with identical methods for the 2 cohorts.
RNA extraction
We isolated total RNA from the 154 FFPE biopsies from patients with MF and from the 20 FFPE biopsies from healthy controls. Tissue sections (10 μm) were used for RNA extraction using the RecoverAll Total Nucleic Acid Isolation Kit (ThermoFisher Scientific/Applied Biosystems) according to the manufacturer’s guidelines. Total RNA quantity and quality were checked using a NanoDrop-1000 spectrophotometer.
qRT-PCR miRNA profiling
The extracted RNA was used for qRT-PCR–based miRNA profiling covering 384 human miRNAs. Fifty nanograms of total RNA from each sample was reverse transcribed to complementary DNA (cDNA) using the Universal cDNA synthesis kit (Exiqon Vedbaek, Denmark). cDNA was diluted 100 times and ExiLENT SYRRGreen master mix was transferred to quantitative polymerase chain reaction panels preloaded with primers, using a pipetting robot. Amplification was performed in a LightCycler 480 Real-Time PCR System (Roche). Raw crossing point (Cp) values and melting points were detected using the Roche LC software and exported. Reactions with several melting points, melting points deviating from assay specifications and with an amplification efficacy below 1.6, were removed. We also removed reactions with Cp values >37 or Cp values within 5 Cp values of the negative control reaction. The data were normalized to the average of assays detected in all samples (global mean), which was identified as the best normalizer using NormFinder.
Statistical analysis
First, we aimed to identify disease-related miRNAs, which may include miRNAs derived from the entire MF milieu, including malignant and nonmalignant T cells as well as the surrounding inflammatory environment. Therefore, we compared the expression levels in the MF samples from the discovery cohort with the expression levels in the healthy control samples using a Student t test followed by correction for multiple testing (Benjamini-Hochberg correction). The significantly differently expressed miRNAs from this analysis were then used to develop a prognostic classifier. Raw data are shown in supplemental Table 2, available on the Blood Web site. To avoid problems with missing data, we first imputed missing values using the k-nearest-neighbors method.21 Next, we fitted a Cox’s proportional hazards model and used the least absolute shrinkage and selection operator (LASSO) method for variable selection and shrinkage.22 LASSO Cox regression is a well-established method for selection of the most predictive markers for time-to-event analysis with high-throughput data.22 The LASSO Cox regression model allows integration of multiple biomarkers into 1 tool providing more accurate prediction of disease progression than single biomarkers alone.22 The regularization parameter was chosen as the largest value where the error was within 1 standard error of the minimum as determined by leave-one-out cross-validation. The Cox regression model for PFS based on the discovery cohort returns a score for each individual that reflects the risk of disease progression. We divided the individuals into high- and low-risk groups by setting a cutoff on this risk score. The cutoff was set so that the number of high-risk individuals in the discovery cohort matched the number of individuals in the discovery cohort who progressed. Next, the discovered model was applied to the independent validation cohort. Raw data for miRNAs included in the model are shown in supplemental Table 3.
We drew time-dependent receiver operating characteristic (ROC) curves to assess the accuracy of the prediction of disease progression for the miRNA classifier and the clinical predictive variables associated with disease outcome.
For PFS and OS analysis, we used the Kaplan-Meier method to draw survival curves and Cox’s proportional hazards model to estimate the effect of various covariates. The survival curves were compared using the log-rank test.
R version 3.3.0 was used for the statistical analysis. The R packages include glmnet that was used for model building, impute for imputing missing value, survminer for plotting Kaplan-Meier curves, and survival for testing of Cox models.
Results
The clinical characteristics of the discovery cohort and the independent validation cohort are shown in Table 1. The median follow-up time for the entire cohort was 8.5 years (interquartile range [IQR]: 4.9-12.3 years); 9.1 years (IQR: 4.4-13.5 years) for the discovery cohort, and 8.0 years (IQR: 5.8-11.4 years) for the validation cohort. Fifty-one patients (33%) progressed from the early to the advanced stages of MF within a median 2-year period (IQR: 8.4 months to 4.2 years). Among those who progressed, 82% progressed within the first 5 years after diagnosis (supplemental Figure 1).
Patient characteristics of the discovery cohort and the independent validation cohort
| . | Discovery cohort . | Independent validation cohort . | ||||
|---|---|---|---|---|---|---|
| Patient characteristics . | Number of patients (%) (n = 82) . | Progression (%) (n = 31) . | Nonprogression (%) (n = 51) . | Number of patients (%) (n = 72) . | Progression (%) (n = 20) . | Nonprogression (%) (n = 52) . |
| Sex | ||||||
| Male | 54 (66) | 19 (61) | 35 (69) | 38 (53) | 10 (50) | 28 (54) |
| Female | 28 (34) | 12 (39) | 16 (31) | 34 (47) | 10 (50) | 24 (46) |
| Age | ||||||
| <60 | 26 (32) | 6 (19) | 13 (25) | 30 (42) | 9 (45) | 21 (40) |
| ≥60 | 56 (68) | 25 (81) | 38 (75) | 42 (58) | 11 (55) | 31 (60) |
| Clinical stage | ||||||
| IA | 29 (35) | 11 (35) | 18 (35) | 42 (58) | 4 (20) | 38 (73) |
| IB | 52 (63) | 20 (65) | 32 (63) | 29 (40) | 15 (75) | 14 (27) |
| IIA | 1 (2) | 0 | 1 (2) | 1 (1) | 1 (5) | 0 |
| T-stage | ||||||
| T1 | 29 (35) | 11 (35) | 18 (35) | 42 (58) | 4 (20) | 38 (73) |
| T1a | 5 | 0 | 5 | 15 | 0 | 15 |
| T1b | 24 | 11 | 13 | 27 | 4 | 23 |
| T2 | 53 (65) | 20 (65) | 33 (65) | 30 (42) | 16 (80) | 14 (27) |
| T2a | 4 | 1 | 3 | 1 | 1 | 0 |
| T2b | 49 | 19 | 30 | 29 | 15 | 14 |
| CLIPi* | ||||||
| 1. Low risk | 10 (12) | 3 (10) | 7 (14) | 19 (26) | 4 (20) | 15 (29) |
| 2. Intermediate risk | 32 (39) | 11 (35) | 21 (41) | 30 (42) | 8 (40) | 22 (42) |
| 3. High risk | 40 (49) | 17 (55) | 23 (45) | 23 (32) | 8 (40) | 15 (29) |
| Treatment† | ||||||
| No treatment | 57 (70) | 22 (71) | 35 (69) | 59 (82) | 14 (70) | 41 (79) |
| Topical treatment | 25 (30) | 9 (29) | 16 (31) | 13 (18) | 6 (30) | 11 (21) |
| . | Discovery cohort . | Independent validation cohort . | ||||
|---|---|---|---|---|---|---|
| Patient characteristics . | Number of patients (%) (n = 82) . | Progression (%) (n = 31) . | Nonprogression (%) (n = 51) . | Number of patients (%) (n = 72) . | Progression (%) (n = 20) . | Nonprogression (%) (n = 52) . |
| Sex | ||||||
| Male | 54 (66) | 19 (61) | 35 (69) | 38 (53) | 10 (50) | 28 (54) |
| Female | 28 (34) | 12 (39) | 16 (31) | 34 (47) | 10 (50) | 24 (46) |
| Age | ||||||
| <60 | 26 (32) | 6 (19) | 13 (25) | 30 (42) | 9 (45) | 21 (40) |
| ≥60 | 56 (68) | 25 (81) | 38 (75) | 42 (58) | 11 (55) | 31 (60) |
| Clinical stage | ||||||
| IA | 29 (35) | 11 (35) | 18 (35) | 42 (58) | 4 (20) | 38 (73) |
| IB | 52 (63) | 20 (65) | 32 (63) | 29 (40) | 15 (75) | 14 (27) |
| IIA | 1 (2) | 0 | 1 (2) | 1 (1) | 1 (5) | 0 |
| T-stage | ||||||
| T1 | 29 (35) | 11 (35) | 18 (35) | 42 (58) | 4 (20) | 38 (73) |
| T1a | 5 | 0 | 5 | 15 | 0 | 15 |
| T1b | 24 | 11 | 13 | 27 | 4 | 23 |
| T2 | 53 (65) | 20 (65) | 33 (65) | 30 (42) | 16 (80) | 14 (27) |
| T2a | 4 | 1 | 3 | 1 | 1 | 0 |
| T2b | 49 | 19 | 30 | 29 | 15 | 14 |
| CLIPi* | ||||||
| 1. Low risk | 10 (12) | 3 (10) | 7 (14) | 19 (26) | 4 (20) | 15 (29) |
| 2. Intermediate risk | 32 (39) | 11 (35) | 21 (41) | 30 (42) | 8 (40) | 22 (42) |
| 3. High risk | 40 (49) | 17 (55) | 23 (45) | 23 (32) | 8 (40) | 15 (29) |
| Treatment† | ||||||
| No treatment | 57 (70) | 22 (71) | 35 (69) | 59 (82) | 14 (70) | 41 (79) |
| Topical treatment | 25 (30) | 9 (29) | 16 (31) | 13 (18) | 6 (30) | 11 (21) |
CLIPi group 1 = low risk (0-1 risk factors); CLIPi group 2 = intermediate risk (2 risk factors); CLIPi group 3 = high risk (3-5 risk factors).12
Use of skin-directed therapies (topical corticosteroids, phototherapy, or nitrogen mustard) upon diagnosis. No patients received systemic therapies.
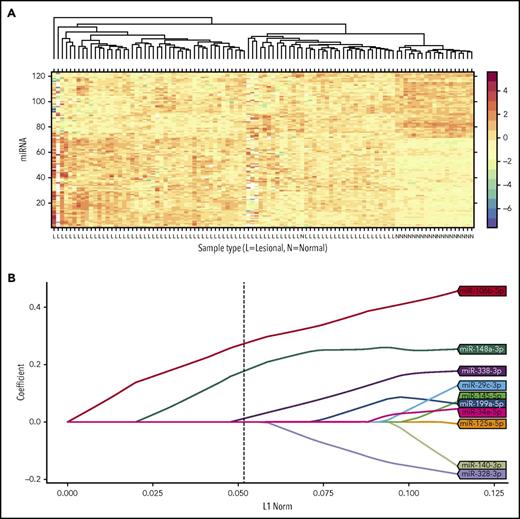
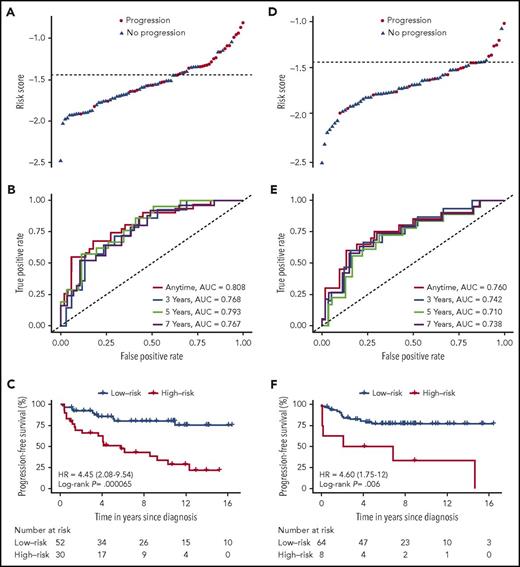
In order to develop a disease-related prognostic miRNA classifier, we assessed miRNA expression in the discovery cohort and compared this expression with the corresponding expression in age- and sex-matched healthy controls. We identified 123 differently expressed miRNAs; 72 miRNAs were upregulated and 51 miRNAs were downregulated (supplemental Table 1). More than two-thirds of the top 25 most upregulated miRNAs were previously identified as differentially upregulated in MF compared with inflammatory skin (supplemental Table 1). Hierarchical clustering based on these differently expressed miRNAs successfully separated the 82 MF samples from the 20 healthy control samples (Figure 1A). Univariate analysis of the association between the expression of the 123 miRNAs and the PFS is shown in supplemental Table 4. Based on the identified differences in how miRNAs were expressed in MF and healthy control samples, we used a LASSO Cox regression model and leave-one-out cross-validation to build a classifier to predict disease progression in patients with early-stage MF. A combination of 3 miRNAs was selected as the best predictor of disease progression in the discovery cohort: miR-106b-5p, miR-148a-3p, and miR-338-3p (Figure 1B; supplemental Figure 2). A risk score formula was derived to calculate a risk score of disease progression for each patient based on the expression level of the 3 miRNAs: miR-106b-5p × 0.273 + miR-148a-3p × 0.177 + miR-338-3p × 0.012. In order to subdivide the patients into a high- and low-risk group of disease progression, we set the risk score cutoff level to −1.44 so that the number of high-risk individuals in the discovery cohort matched the number of individuals who progressed. Thus, patients with a risk score above −1.44 were included in the high-risk group and risk scores below −1.44 categorized the patients into the low-risk group. When we assessed the distribution of risk scores according to disease progression, patients with a high-risk score generally had a higher risk of disease progression (Figure 2A). The prognostic accuracy of the 3-miRNA classifier was assessed by time-dependent ROC curves (Figure 2B). We observed a significant difference in PFS between the high-risk group and the low-risk group, hazard ratio (HR), 4.45; (95% confidence interval [CI], 2.08-9.54); P = .000065 (Figure 2C). Accordingly, the 5-year PFS was lower for the high-risk group, 51.4% (95% CI, 35.9% to 73.5%), than for the low-risk group, 85.3% (95% CI, 75.7% to 96.1%). Of note, the distribution of clinical patient characteristics was largely similar in patients with and without disease progression (Table 1).
Construction of the 3-miRNA classifier. (A) Hierarchical clustering of the 123 differently expressed miRNAs in the discovery cohort of 82 patients with MF (lesional sample) and the 20 age- and sex-matched healthy controls (normal control sample), using Euclidean distance with average linkage clustering. Each row represents a single miRNA, and each column represents an individual sample. Colors indicate the normalized expression level from low (blue colors) to high (red colors) expression on a log scale from 4 to −6. (B) LASSO coefficient profiles of the 10 miRNAs most predictive of disease progression. miR, microRNA.
Construction of the 3-miRNA classifier. (A) Hierarchical clustering of the 123 differently expressed miRNAs in the discovery cohort of 82 patients with MF (lesional sample) and the 20 age- and sex-matched healthy controls (normal control sample), using Euclidean distance with average linkage clustering. Each row represents a single miRNA, and each column represents an individual sample. Colors indicate the normalized expression level from low (blue colors) to high (red colors) expression on a log scale from 4 to −6. (B) LASSO coefficient profiles of the 10 miRNAs most predictive of disease progression. miR, microRNA.
Risk score by the 3-miRNA classifier, time-dependent ROC curves, and Kaplan-Meier PFS curves in the discovery cohort and the independent validation cohort. Risk score, time-dependent ROC curves, and PFS curves for the discovery cohort (A-C) and the validation cohort (D-F). The AUC was calculated for the ROC curves at 3, 5, or 7 years to assess prognostic accuracy (B,E). We calculated P values using the log-rank test (C,F). AUC, area under the curve.
Risk score by the 3-miRNA classifier, time-dependent ROC curves, and Kaplan-Meier PFS curves in the discovery cohort and the independent validation cohort. Risk score, time-dependent ROC curves, and PFS curves for the discovery cohort (A-C) and the validation cohort (D-F). The AUC was calculated for the ROC curves at 3, 5, or 7 years to assess prognostic accuracy (B,E). We calculated P values using the log-rank test (C,F). AUC, area under the curve.
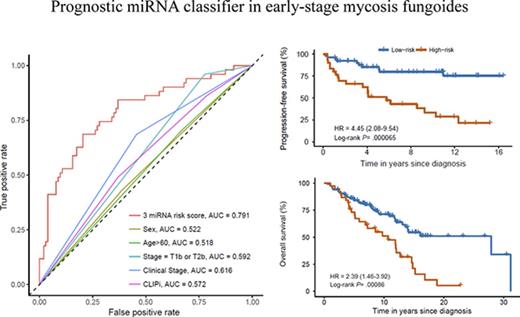
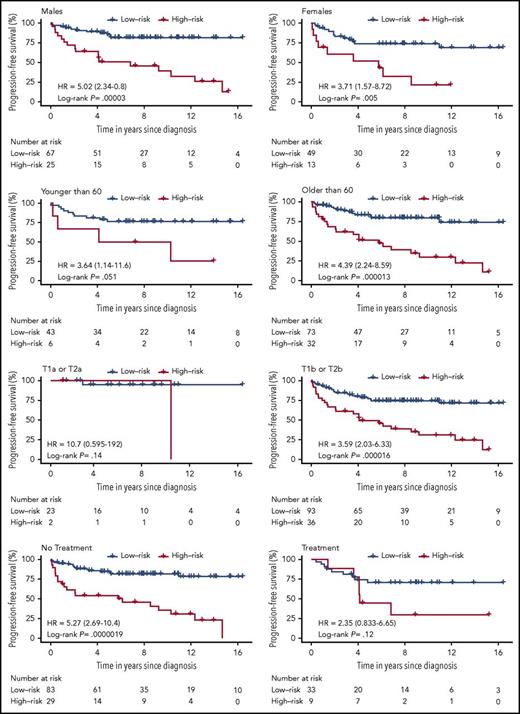
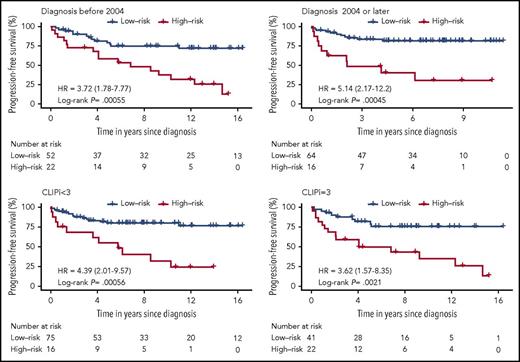
The prognostic value of the 3-miRNA classifier was validated in an independent analysis of the validation cohort of 71 patients with early-stage MF. We confirmed that patients from the validation cohort with a high-risk score generally had a higher risk of disease progression (Figure 2D). We assessed the accuracy of the classifier in the independent validation cohort by ROC curves at varying time points (Figure 2E). PFS curves were significantly different in the high-risk compared with the low-risk group, HR, 4.60; (95% CI, 1.75-12.0); P = .006 (Figure 2F). Performing univariate analysis, we found that the 3-miRNA classifier was a stronger predictor of disease progression than the miRNAs individually, and that it was significantly stronger than existing clinical prognostic factors, including sex, age, patch/plaque T stage (T1a/T2a vs T1b/T2b), and the CLIPi score (Table 2). Thirty-eight (25%) of all patients received symptomatic topical treatment at the time the diagnostic biopsy was obtained (Table 1): 33 patients were treated with topical corticosteroids, 3 patients were treated with UVB, and 2 patients were treated with nitrogen mustard. Systemic treatment was not used. Interestingly, use of topical treatment was not associated with disease progression (Table 2). Patch/plaque T stage (T1a/T2a vs T1b/T2b) was the only independent clinical factor that was significantly associated with disease progression (Table 2). Therefore, we adjusted the PFS analysis by patch/plaque T stage (T1a/T2a vs T1b/T2b) and found that the 3-miRNA classifier remained a powerful and independent predictor of disease progression; this was the case both in the entire cohort, HR, 3.79; (95% CI, 2.17-6.65); P = .000005, the discovery cohort, HR, 4.35 (95% CI, 2.03-9.33), P= .000087, and in the independent validation cohort, HR, 3.47; (95% CI, 1.30-9.24); P = .02. In addition, stratification by clinical prognostic factors revealed that the 3-miRNA classifier remained a significant prognostic marker of disease progression in patients with early-stage MF (Figure 3). Of note, use of topical treatment and the year of diagnosis had no impact on the 3-miRNA classifier. The prognostic accuracy was clearly higher of the 3-miRNA classifier than of the existing clinical prognostic factors as well as the individual miRNAs (Figure 4).
Univariate association of clinical characteristics, clinical prognostic factors, single miRNAs, and the 3-miRNA classifier with PFS
| . | Discovery cohort (n = 82) . | Independent validation cohort (n = 72) . | Entire cohort (n = 154) . | |||
|---|---|---|---|---|---|---|
| Risk factor . | HR (95% CI) . | P . | HR (95% CI) . | P . | HR (95% CI) . | P . |
| Sex (male vs female) | 0.84 (0.40-1.75) | .64 | 0.79 (0.33-1.89) | .59 | 0.84 (0.48-1.47) | .54 |
| Age (>60 vs ≤60 y) | 2.05 (0.78-5.38) | .12 | 0.93 (0.38-2.26) | .88 | 1.43 (0.77-2.65) | .25 |
| Stage (T1b+T2b vs T1a+T2a) | 2.94 (0.40-21.60) | .21 | 7.11 (0.95-53.4) | .01 | 5.01 (1.22-20.6) | .004 |
| Treatment (yes vs no)* | 1.04 (0.47-2.27) | .93 | 1.37 (0.52-3.6) | .54 | 1.16 (0.63-2.12) | .64 |
| CLIPi† | ||||||
| CLIPi group 3 vs 1-2 | 1.42 (0.69-2.93) | .34 | 1.41 (0.57-3.45) | .46 | 1.46 (0.84-2.55) | .18 |
| CLIPi group 2-3 vs 1 | 1.40 (0.42-4.63) | .57 | 1.58 (0.53-4.72) | .40 | 1.58 (0.71-3.52) | .24 |
| Single miRNAs | ||||||
| miR-106b-5p | 2.24 (1.49-3.37) | .00009 | 2.85 (1.49-5.46) | .0007 | 2.39 (1.72-3.33) | .0000002 |
| miR-148a-3p | 2.71 (1.56-4.73) | .0004 | 1.90 (0.92-3.93) | .07 | 2.41 (1.57-3.71) | .00005 |
| miR-338-3p | 1.64 (1.18-2.28) | .004 | 1.30 (0.84-2.00) | .24 | 1.49 (1.14-1.94) | .004 |
| miRNA classifier | ||||||
| miR classifier (low vs high risk) | 4.45 (2.08-9.54) | .000065 | 4.60 (1.75-12) | .006 | 4.28 (2.45-7.46) | .0000007 |
| miR classifier (risk score) | 13.9 (4.38-44.20) | .0000007 | 17 (3.45-83.60) | .0005 | 13.5 (5.6-32.7) | .0000000001 |
| . | Discovery cohort (n = 82) . | Independent validation cohort (n = 72) . | Entire cohort (n = 154) . | |||
|---|---|---|---|---|---|---|
| Risk factor . | HR (95% CI) . | P . | HR (95% CI) . | P . | HR (95% CI) . | P . |
| Sex (male vs female) | 0.84 (0.40-1.75) | .64 | 0.79 (0.33-1.89) | .59 | 0.84 (0.48-1.47) | .54 |
| Age (>60 vs ≤60 y) | 2.05 (0.78-5.38) | .12 | 0.93 (0.38-2.26) | .88 | 1.43 (0.77-2.65) | .25 |
| Stage (T1b+T2b vs T1a+T2a) | 2.94 (0.40-21.60) | .21 | 7.11 (0.95-53.4) | .01 | 5.01 (1.22-20.6) | .004 |
| Treatment (yes vs no)* | 1.04 (0.47-2.27) | .93 | 1.37 (0.52-3.6) | .54 | 1.16 (0.63-2.12) | .64 |
| CLIPi† | ||||||
| CLIPi group 3 vs 1-2 | 1.42 (0.69-2.93) | .34 | 1.41 (0.57-3.45) | .46 | 1.46 (0.84-2.55) | .18 |
| CLIPi group 2-3 vs 1 | 1.40 (0.42-4.63) | .57 | 1.58 (0.53-4.72) | .40 | 1.58 (0.71-3.52) | .24 |
| Single miRNAs | ||||||
| miR-106b-5p | 2.24 (1.49-3.37) | .00009 | 2.85 (1.49-5.46) | .0007 | 2.39 (1.72-3.33) | .0000002 |
| miR-148a-3p | 2.71 (1.56-4.73) | .0004 | 1.90 (0.92-3.93) | .07 | 2.41 (1.57-3.71) | .00005 |
| miR-338-3p | 1.64 (1.18-2.28) | .004 | 1.30 (0.84-2.00) | .24 | 1.49 (1.14-1.94) | .004 |
| miRNA classifier | ||||||
| miR classifier (low vs high risk) | 4.45 (2.08-9.54) | .000065 | 4.60 (1.75-12) | .006 | 4.28 (2.45-7.46) | .0000007 |
| miR classifier (risk score) | 13.9 (4.38-44.20) | .0000007 | 17 (3.45-83.60) | .0005 | 13.5 (5.6-32.7) | .0000000001 |
Use of skin-directed therapies (topical corticosteroids, phototherapy, or nitrogen mustard) upon diagnosis. No patients received systemic therapies.
CLIPi group 1 = low risk (0-1 risk factors); CLIPi group 2 = intermediate risk (2 risk factors); CLIPi group 3 = high risk (3-5 risk factors).12
PFS curves for all 154 patients with early-stage MF based on the 3-miRNA classifier stratified by clinical prognostic factors and clinical characteristics. Treatment is defined by use of skin-directed therapies (topical corticosteroids, phototherapy, or nitrogen mustard) upon diagnosis. No patients received systemic therapies. CLIPi was categorized as CLIPi < 3, including patients with CLIPi group 1 = low risk (0-1 risk factors) and CLIPi group 2 = intermediate risk (2 risk factors); and CLIPi = 3, including patients with CLIPi group 3 = high risk (3-5 risk factors).12 P values were calculated using the log-rank test.
PFS curves for all 154 patients with early-stage MF based on the 3-miRNA classifier stratified by clinical prognostic factors and clinical characteristics. Treatment is defined by use of skin-directed therapies (topical corticosteroids, phototherapy, or nitrogen mustard) upon diagnosis. No patients received systemic therapies. CLIPi was categorized as CLIPi < 3, including patients with CLIPi group 1 = low risk (0-1 risk factors) and CLIPi group 2 = intermediate risk (2 risk factors); and CLIPi = 3, including patients with CLIPi group 3 = high risk (3-5 risk factors).12 P values were calculated using the log-rank test.
Accuracy of the 3-miRNA classifier, clinical prognostic factors, and single miRNAs from the classifier assessed by time-dependent ROC curves of PFS. (A) Comparisons of the prognostic accuracy of the 3-miRNA classifier and existing clinical prognostic factors: sex, age, T1b or T2b, clinical stage, and CLIPi. (B) Comparisons of the prognostic accuracy of the 3-miRNA–based classifier and the single miRNAs: miR-106b-5p, miR-148a-3p, and miR-338-3p.
Accuracy of the 3-miRNA classifier, clinical prognostic factors, and single miRNAs from the classifier assessed by time-dependent ROC curves of PFS. (A) Comparisons of the prognostic accuracy of the 3-miRNA classifier and existing clinical prognostic factors: sex, age, T1b or T2b, clinical stage, and CLIPi. (B) Comparisons of the prognostic accuracy of the 3-miRNA–based classifier and the single miRNAs: miR-106b-5p, miR-148a-3p, and miR-338-3p.
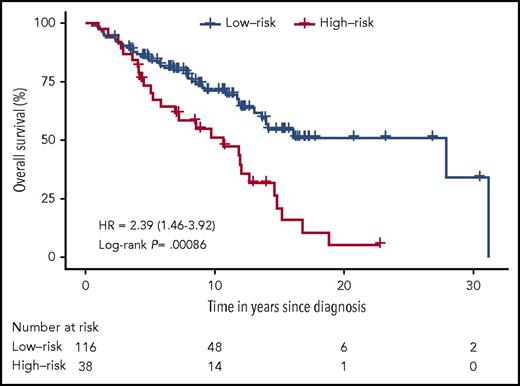
Notably, assessment of the proportion of OS in the entire patient cohort revealed that patients predicted to have a high risk of disease progression had a significantly decreased OS compared with patients predicted to have a low risk of disease progression, HR, 2.39; (95% CI, 1.46 to 3.92); P = .00086 (Figure 5). Thus, in early-stage MF, the 3-miRNA classifier identified patients with a high risk of disease progression, and it could be used to predict OS in patients who were diagnosed with early-stage MF.
Proportion of OS in the high- and low-risk group of disease progression in patients with early-stage MF predicted by the 3-miRNA classifier. We calculated P values using the log-rank test.
Proportion of OS in the high- and low-risk group of disease progression in patients with early-stage MF predicted by the 3-miRNA classifier. We calculated P values using the log-rank test.
Discussion
In this study, we used a large nationwide cohort of patients with early-stage MF to develop and validate a prognostic 3-miRNA classifier for patients diagnosed with early-stage MF. This 3-miRNA classifier successfully stratified patients into groups at high and low risk of disease progression at the time of diagnosis; clearly significant differences in PFS were seen in both the discovery cohort and the validation cohort. The proposed classifier outperformed existing clinical prognostic factors, for prediction of PFS. After stratification and adjustment for these factors, the 3-miRNA classifier remained a strong independent prognostic tool adding prognostic value to the existing clinical prognostic factors, including CLIPi. Importantly, patients whom the 3-miRNA classifier predicted would have a high risk of disease progression had a significantly reduced OS, suggesting that a more aggressive treatment strategy may be beneficial in these patients. Thus, the 3-miRNA classifier may add significant value to CLIPi for prediction of the prognosis for patients diagnosed with early-stage MF.
Current therapeutic management of patients diagnosed with early-stage MF includes a “watch-and-wait” approach or use of skin-directed therapies substantiated by an overall favorable life expectancy in these patients.8 Accordingly, systemic therapies are first initiated upon progression to the advanced stages of MF.8 Thus, the present miRNA classifier may allow for earlier initiation of more aggressive treatment in patients at high risk of disease progression and, conversely, longer periods with clinical monitoring without treatment in low-risk patients. The 3-miRNA classifier may therefore pave the way for a more personalized treatment of these patients.
The present study provides evidence that miRNAs are prognostic markers in MF, and they therefore may also play a pivotal role in the pathogenesis of the disease. Importantly, the majority of the most differentially expressed miRNAs identified here were previously linked to MF in 1 or more studies. For instance, more than two-thirds of the top 25 most upregulated miRNAs in the present investigation were previously identified as differentially upregulated in MF (supplemental Table 1). These findings are remarkable given the aberrant expression of several relevant miRNAs in benign inflammatory skin lesions, and they underline both the technical and the biological robustness of miRNA profiling in skin disease. Notably, these data strongly indicate that the present cohort of patients is representative of a typical cohort of MF patients, not only clinically but also with regard to global miRNA expression. Indeed, we identified multiple key miRNAs of pathological importance that were differently regulated in early-stage MF compared with healthy controls. In particular, we found deregulation of miR-155, miR-21, and miR-16, which are recognized as oncomiRs and linked to the pathogenesis of MF.15,23 The regulation and biological function of the 3-miRNA classifier, miR-106b-5p, miR-148a-3p, and miR-338-3p, are still sparsely investigated in MF. Previous studies, including a large miRNA profiling for MF/Sézary syndrome, did not highlight the 3 classifier miRNAs as differently expressed.16,24,25 This difference may rely on use of different study designs and control groups. Previous studies used benign inflammatory diseases as controls for identifying diagnostic or disease-unique miRNAs, whereas the present classifier was based on disease-related miRNAs, thus miRNAs derived from malignant T cells and the surrounding inflammatory environment, using healthy individuals as controls. Thus, as the classifier miRNAs are not differently expressed from benign inflammatory diseases, they have not been previously highlighted.18 Recently, a number of mutational genome profiling and gene expression profiling studies have highlighted heterogeneity of CTCL, which may also contribute to minor differences in reported miRNA expression.26-31 However, miR-106b* has previously been associated with advanced MF.18 Moreover, in the aggressive leukemic variant of CTCL, Sézary syndrome, miR-106b is associated with a poor disease outcome and suggested to have an oncogenic function by targeting PTEN.32,33 This indicates that miR-106b could be involved in MF disease progression. miR-106b is a part of the miR-106b-25 cluster, which also includes miR-93 and miR-25. We found that both these miRNAs are deregulated in early-stage MF compared with healthy controls. Moreover, miR-93 and miR-25 are associated with advanced MF,18,25 which indicates that the miR-106b-25 cluster is an oncogenic driver in MF as also previously reported in other hematological and solid cancers.34
miR-148a and miR-338 are also deregulated in a number of other cancer types, and they have been linked to both classification and prognosis of breast and colorectal cancer35,36 as well as to tumor cell proliferation, migration, and metastasis.37,38 Of note, miR-148a is suggested to be involved in tumor cell proliferation through the PI3K/AKT/mTOR signaling pathway and apoptosis through BCL-2 and BIM,39 whereas miR-338-3p impacts tumor cell apoptosis through the PTEN-AKT pathway,40 all important pathways in the pathogenesis of MF.41,42 Therefore, the miRNAs included in the 3-miRNA classifier may act as both a “marker” and a “maker” in the carcinogenesis of MF.
This study is limited by the retrospective study design, and an even larger sample size would have been preferred. An international multicenter setting would have provided a larger sample size and strengthened the study, but it was not currently possible. However, taking the rarity of the disease into account, the present sample size was relatively large. The Danish nationwide registries and precise individual-level linkage to the medical records provided a unique opportunity to identify a cohort of well-characterized MF patients with complete and long-term follow-up. Indeed, the follow-up time was certainly longer than previously presented in large-scale clinical prognostic studies of patients with early-stage MF, explaining a slightly higher percentage of disease progression in the present study compared with what has previously been reported.5,6,12 In addition, the Danish registry system enabled collection of the specific FFPE biopsy used for the initial diagnosis of early-stage MF in each individual patient and subsequent development of a prognostic tool specifically designed to predict disease progression at the time of diagnosis. FFPE preserved tissue yields high-quality miRNA with an expression profile that correlates with matched fresh frozen tissue.43,44 Moreover, the robustness of miRNA in FFPE biopsies is largely independent of formalin fixation time and duration of storage, which was confirmed in this study (Figure 3).43,45,46 Using the clinical variables, we further demonstrated that the 3-miRNA classifier is robust independently of clinical factors and use of topical treatments at the time the patients are biopsied.
In conclusion, we have developed and validated a 3-miRNA classifier that at the time of diagnosis effectively predicts progression from early to advanced stage MF, and it could be used to predict OS in these patients. Thus, the 3-miRNA classifier adds significant prognostic value to existing clinical prognostic factors (CLIPi) and paves the way for implementation of personalized treatment of patients diagnosed with early-stage MF.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank the departments of pathology in Denmark for generously providing biopsies to the study.
This study was supported by grants from LINAK A/S Nordborg (L.I.), the Danish Cancer Society (Kræftens Bekæmpelse), the Fight Cancer Program (Knæk Cancer) (N.O.), the Novo Nordisk Research Foundation (N.O and L.I.), Aage Bangs Foundation (L.M.L.), and Kræftfonden (L.I.).
Authorship
Contribution: L.I., L.M.L., S.B., N.O., and A.W.-O. designed the study; L.M.L., L.I., and P.C. obtained and assembled the data; L.M.L. and L.I. collected the samples and the clinical information; S.B., L.M.L., L.I., A.H.R., N.O., C.J., A.W.-O., L.M.R.G., T.L., A.W., and T.K. analyzed and interpreted the data; L.M.L., L.I., N.O., and S.B. wrote the report; and all authors revised the report and approved the final version.
Conflict-of-interest disclosure: T.L. is employed by LEO Pharma A/S. Some of the information presented is a part of a patent application filed by Aarhus University and University of Copenhagen. The remaining authors declare no competing financial interests.
Correspondence: Lise M. Lindahl, Department of Dermatology, Aarhus University Hospital, P.P. Orumsgade 11, DK-8000 Aarhus C, Denmark; e-mail: lise.lindahl@clin.au.dk; and Lars Iversen, Department of Dermatology, Aarhus University Hospital, P.P. Orumsgade 11, DK-8000 Aarhus C, Denmark; e-mail: lars.iversen@clin.au.dk.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal