Key Points
Prophylactic administration of NAC was effective in preventing severe TTP signs in mice by reducing the VWF multimer size.
In both mice and baboons, NAC was not effective in resolving preexisting TTP signs, as thrombus resolution could not be achieved.
Abstract
Thrombotic thrombocytopenic purpura (TTP) is a microangiopathic disorder diagnosed by thrombocytopenia and hemolytic anemia, associated with a deficiency in von Willebrand factor (VWF)–cleaving protease ADAMTS13. Current treatment is based on plasma infusion for congenital TTP, or plasma exchange, often in combination with immunosuppressive agents, for acquired TTP. These treatment methods are not always effective; therefore, new treatment methods are highly necessary. N-acetylcysteine (NAC), an FDA-approved anti-mucolytic agent, is a possible new treatment strategy for TTP, as it was demonstrated to reduce disulfide bonds in VWF, thereby decreasing VWF multimers size and hence their prothrombotic potential. We investigated whether NAC, without concurrent plasma exchange and immunosuppressive therapy, is effective in preventing and resolving TTP signs, using well-established murine and baboon models for TTP. In mice, prophylactic administration of NAC was effective in preventing severe TTP signs. This in vivo finding was supported by in vitro data demonstrating the VWF multimer–reducing properties of NAC in solution. Nonetheless, in both mice and baboons, administration of NAC was not effective in resolving preexisting TTP signs; thrombocytopenia, hemolytic anemia, and organ damage could not be reversed, as thrombus resolution was not achieved. Failure to improve clinical outcome occurred even though a reduction in VWF multimers was observed, demonstrating that NAC was efficient in reducing disulfide bonds in circulating VWF multimers. In conclusion, prophylactic administration of NAC, without concurrent plasma exchange, was effective in preventing severe TTP signs in mice, but NAC was not effective in resolving preexistent acute TTP signs in mice and baboons.
Introduction
Thrombotic thrombocytopenic purpura (TTP) is a microangiopathic disorder in which patients experience acute episodes of microvascular thrombosis resulting in neurological complications and organ failure. Acute TTP is diagnosed by thrombocytopenia and microangiopathic hemolytic anemia1 and associated with a deficiency in the von Willebrand factor (VWF)–cleaving protease ADAMTS13 (a disintegrin and metalloprotease with thrombospondin type-1 repeats, member 13).2,3 Under physiological conditions, ADAMTS13 cleaves prothrombotic ultra-large (UL) VWF multimers, secreted from activated endothelial cells, in the VWF A2 domain at the Y1605-M1606 cleavage site.2 In TTP, however, ADAMTS13 activity is absent or reduced, either due to congenital mutations or to the presence of anti-ADAMTS13 autoantibodies.4 This impedes cleavage of UL VWF multimers, resulting in spontaneous platelet adhesion and subsequent microthrombi formation.
Patients can experience a single acute episode of TTP. However, in one-third of patients, the disease returns with one or more relapses after a period of remission.5 Congenital TTP patients are currently treated with fresh frozen plasma infusion. Acquired TTP patients receive plasma exchange, often in combination with immunosuppressive agents to tone down the immune response and suppress the formation of autoantibodies.4 However, these treatment methods are not always effective in achieving remission and are associated with a mortality rate of 10% to 20%.6 Therefore, new treatment methods are highly necessary. As previously suggested, N-acetylcysteine (NAC) might be a good alternative for patients with acute TTP.7 NAC, an anti-mucolytic agent approved by the US Food and Drug Administration (FDA) and used for treatment of congestive and obstructive lung diseases as well as for acetaminophen overdose, contains a free thiol and is therefore able to reduce disulfide bonds.8 As dimerization of VWF monomers is achieved by C-terminal disulfide bonds and further multimerization through N-terminal disulfide bond formation, NAC was tested for its VWF-reducing capabilities.7 A reduction in UL VWF multimers was observed, as well as diminished calcium ionophore–induced thrombus formation in mesenteric venules of Adamts13−/− mice. As microthrombus formation during TTP pathology is dependent on UL VWF multimers, NAC could therefore potentially prevent the formation of occlusive VWF-rich microthrombi. Following the publication of the study by Chen et al,7 several case reports demonstrated that treatment of acute acquired TTP patients with NAC combined with plasma exchange therapy resulted in an improved recovery of the platelet count in 5 patients,9-11 whereas it was ineffective in 3 other patients.12-15 Due to the low number of patients and the variable outcome, as well as the use of NAC as an adjuvant to standard plasma exchange therapy, it is unclear if NAC is indeed an effective therapy to treat TTP. Proof of the concept that NAC is effective as a treatment for TTP, independent of plasma exchange, has not been demonstrated thus far. Therefore, preclinical experiments in animal models are required to gain more insight into the potential benefits of NAC.
In the current study, we investigated the effect of NAC on TTP signs, without concurrent plasma infusion or exchange and immunosuppressive therapy, using well-established preclinical murine and baboon models for TTP. This study was performed to examine whether the UL VWF–reducing properties of NAC are sufficient to prevent and/or resolve the clinical signs of TTP.
Materials and methods
Methods used for analyses of VWF multimers and VWF ristocetin and botrocetin cofactor activity are provided in the supplemental Methods (available on the Blood Web site).
Platelet agglutination
Pooled citrated normal human plasma (NHP) was incubated with 0 or 32 mM NAC for 30 minutes at 37°C (9/10 volume plasma, 1/10 volume NAC or Tyrode buffer containing N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid (HEPES). The reaction mixture (1/5 volume) was added to 4/5 volume of a final concentration of 200 × 103 lyophilized platelets per microliter (Bio/Data Corporation, Horsham, PA), and agglutination was induced by using 0.9 mg/mL ristocetin (American Biochemical and Pharmaceuticals, Ltd., Marlton, NJ). As a control, our in-house–developed anti-GP1b antibody 6B416 was used at 20 nM to block platelet-VWF interaction. Agglutination was followed for 12 minutes on a platelet aggregometer (Chrono-log 490; Stago, Leiden, The Netherlands).
To break down preexisting agglutinates, plasma was mixed with a final concentration of 200 × 103 lyophilized platelets per microliter, and agglutinations were induced by using 0.9 mg/mL ristocetin. When agglutinates were formed, 32 mM NAC was added and measurements were continued for 30 minutes to observe possible agglutinate breakdown.
Congenital TTP mouse model
Experimental procedure
Mouse studies were approved by the Institutional Animal Care and Use Committee of KU Leuven (Belgium; project number: P152/2014). Adamts13−/− mice (CASA/Rk-C57BL/6J-129X1/SvJ background) were injected intravenously with 2250 U/kg recombinant human VWF (rVWF; Shire, Vienna, Austria) to trigger TTP signs, as previously described.17 NAC was intravenously injected either 15 minutes before rVWF administration to test its prophylactic effect (for preventing TTP), or 1 hour and 12 hours after rVWF administration to test its treatment effect (for resolving TTP), at a dose of 800 mg/kg under isoflurane-O2 anesthesia. Control Adamts13−/− mice were injected with an equal volume of saline (vehicle). Blood collection was performed at baseline (7 days before the start of the experiment), 6 hours after rVWF injection for VWF multimer analysis, and 24 hours after rVWF injections for total blood cell counts and lactate dehydrogenase (LDH) measurements. Blood was collected via retro-orbital venipuncture, using trisodium citrate (5/1 (vol/vol) of blood/3.8% sodium citrate) or EDTA (10/1 (vol/vol) of blood/0.5 M EDTA) for anticoagulation. Total blood cell counts were determined in EDTA-anticoagulated blood by using a veterinary multispecies hematology system (Hemavet 950; Drew Scientific, Dallas, TX). Plasma was obtained from blood collected on sodium citrate, subjected to centrifugation at 2500g for 6 minutes, and stored at −80°C.18,19 Analyses of VWF antigen and LDH activity levels and hematoxylin and eosin (H&E) staining were performed as described in the supplemental Methods.
Acquired TTP baboon model
Experimental procedure
Housing, treatment, blood withdrawals, NAC injections, and care of the Cape baboons (Papio ursinus), as well as the final protocol, were approved by the Control Committee on Animal Experimentation of the University of the Free State (Bloemfontein, South Africa) in accordance with the National Code for Animal Use in Research, Education, Diagnosis and Testing of Drugs and Related Substances in South Africa, and the Institutional Animal Care and Use Committee of KU Leuven (Belgium), before the experiments were begun. Animals, both male and female, weighed between 7.5 and 14 kg and were disease-free for at least 6 months. All animals were anesthetized by an intramuscular injection of 10 mg/kg ketamine hydrochloride. While under anesthesia, blood withdrawals and antibody injections were performed by venipuncture of the femoral vein. Blood was obtained daily on EDTA (BD Vacutainer; BD, Woodmead, South Africa) for total blood cell counts and every other day on citrate (3.2%; BD Vacutainer) for the determination of hematological parameters and plasma collection. In-house–developed anti-ADAMTS13 antibody 3H9 was injected every 48 hours at a concentration of 600 μg/kg to inhibit ADAMTS13 activity and induce TTP signs, as previously described.20,21 After the onset of TTP signs, NAC was injected via the cephalic vein every 12 hours at a dose of 400 mg/kg over 20 minutes. The control group was injected with a similar volume of saline. Blood measurements were performed as described in the supplemental Methods.
Statistics
All data are presented as the mean and standard deviation. Statistical comparisons between mice at different time points were done by a two-tailed paired Student t test, and comparisons between two independent groups were performed by a two-tailed unpaired Student t test using Prism 6 software (GraphPad Software, Inc., La Jolla, CA).
Results
NAC reduces the size and activity of VWF in plasma
First, we assessed the ability of NAC to reduce high molecular weight (HMW) VWF multimers in human plasma, as was previously reported by Chen et al.7 To this end, pooled citrated NHP was incubated with 0, 4, 8, 16, or 32 mM NAC for 30 minutes at 37°C. Addition of NAC to NHP resulted in a concentration-dependent reduction of HMW VWF multimers (Figure 1A, quantification in Figure 1B) and disappearance of HMW multimers when ≥ 8 mM NAC was used. The reduction in VWF multimers could be blocked by the addition of 64 mM N-ethylmaleimide (NEM), which alkylates NAC (supplemental Figure 1). The decrease in VWF multimer size corresponded to a dose-dependent reduction in platelet GP1b binding, as demonstrated by a reduction in ristocetin cofactor activity (supplemental Figure 2A). As we aimed to study the effect of NAC treatment in preclinical mouse and baboon models, we next investigated whether NAC also reduces VWF in murine and baboon plasma. NAC was able to reduce HMW VWF in murine plasma obtained from Adamts13−/− mice (Figure 1C-D). The reduction in murine HMW VWF was also reflected in a decrease in botrocetin cofactor activity (supplemental Figure 2B). In baboon plasma, a concentration-dependent decrease in HMW VWF multimers was observed (Figure 1E-F), correlating with a reduced ristocetin cofactor activity (supplemental Figure 2C). Higher concentrations of NAC were needed to reduce the HMW VWF multimers (containing UL VWF multimers) in plasma of the Adamts13−/− mice (Figure 1C-D) compared with those in human (Figure 1A-B), baboon (Figure 1E-F), and Adamts13+/+ mouse plasma (data not shown) (all devoid of UL VWF multimers). In conclusion, these results confirm that NAC is able to reduce soluble VWF multimers, leading to decreased GP1b binding in multiple species.
NAC reduces the size and activity of soluble VWF multimers in plasma. NHP, normal mouse plasma (NMP), and normal baboon plasma (NBP) was incubated with NAC, and VWF multimer composition was analyzed in NHP (A), NMP (C), and NBP (E). The percentage of VWF multimers was calculated using densitometric analysis of NHP (B), NMP (D), and NBP (F). The detection limit of the VWF multimer analysis was limited to multimers, and therefore the monomers obtained when incubating NMP with NAC are not visible on the gel. Each bar on the graphs represents the mean and standard deviation from 3 measurements. HMW, high molecular weight; LMW, low molecular weight; MMW, medium molecular weight.
NAC reduces the size and activity of soluble VWF multimers in plasma. NHP, normal mouse plasma (NMP), and normal baboon plasma (NBP) was incubated with NAC, and VWF multimer composition was analyzed in NHP (A), NMP (C), and NBP (E). The percentage of VWF multimers was calculated using densitometric analysis of NHP (B), NMP (D), and NBP (F). The detection limit of the VWF multimer analysis was limited to multimers, and therefore the monomers obtained when incubating NMP with NAC are not visible on the gel. Each bar on the graphs represents the mean and standard deviation from 3 measurements. HMW, high molecular weight; LMW, low molecular weight; MMW, medium molecular weight.
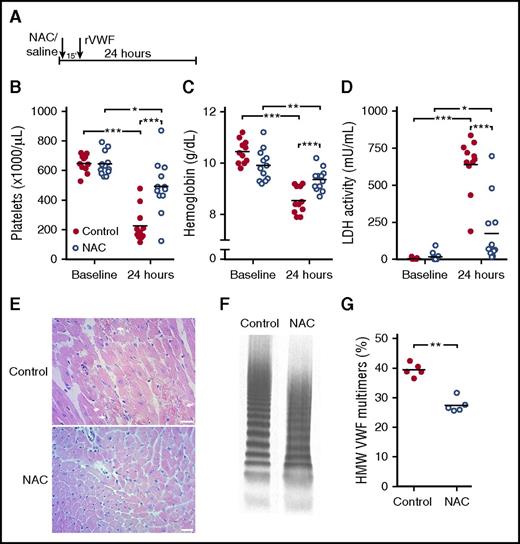
Prophylactic administration of NAC prevents severe TTP signs in mice
As NAC has the capacity to reduce VWF multimer size in both murine and baboon plasma, we continued to investigate the effect of NAC on TTP signs in preclinical animal models, as targeting VWF was previously demonstrated to be successful in the treatment of TTP.21,22 We first investigated whether prophylactic administration of NAC would be able to prevent TTP signs in mice. For this, we used a mouse model for congenital TTP in which administration of rVWF containing UL VWF multimers to Adamts13−/− mice triggers TTP signs.17 To test the effect of prophylactic administration of NAC, mice were either injected with 800 mg/kg7 NAC (NAC treatment group) or vehicle (control group) 15 minutes prior to the rVWF trigger (Figure 2A). Twenty-four hours after rVWF injection, blood was drawn and analyzed for the TTP signs of active disease. In the control group, TTP signs were observed, as platelet counts were severely decreased (Figure 2B), hemoglobin levels as a measurement for hemolytic anemia were severely decreased (Figure 2C), and LDH activity levels indicating tissue damage and hemolysis were highly increased (Figure 2D). Myocardial necrosis was observed in heart sections obtained from these mice (Figure 2E, upper panel). In the group of mice that received prophylactic administration of NAC, only a minor decrease in platelet counts (Figure 2B) and hemoglobin levels (Figure 2C) was observed, as well as a moderate increase in LDH levels (Figure 2D). Furthermore, these mice were protected from myocardial necrosis (Figure 2E, lower panel). HMW VWF multimers were reduced in all mice that received NAC, but not in control mice (Figure 2F-G). These results demonstrate that when NAC is administered before the onset of TTP signs, the manifestation of severe TTP signs is averted, an effect that most probably can be attributed to the reduction of HMW VWF multimers.
Prophylactic administration of NAC prevents severe TTP signs in mice. (A) Adamts13−/− mice (control n = 11; NAC treatment n = 12) were injected with 800 mg/kg NAC (open blue circles) or equal volumes of saline for control (solid red circles) 15 minutes prior to rVWF injection. (B-D) At baseline and 24 hours after rVWF injection, platelet count (B), hemoglobin level (C), and LDH activity level (D) were determined. (E) H&E staining was performed on myocardial tissue sections from control mice (top panel) and mice with prophylactic NAC administration (bottom panel). Arrows indicate sites of myocardial necrosis. Scale bars indicate 50 μm. (F) VWF multimer composition was analyzed, and representative images of VWF multimeric profiles from a control mouse and an NAC-treated mouse are shown. (G) Percentage of HMW VWF multimers was quantified using densitometry. Statistical significance is indicated with asterisks: *P < .05, **P < .01, ***P < .001.
Prophylactic administration of NAC prevents severe TTP signs in mice. (A) Adamts13−/− mice (control n = 11; NAC treatment n = 12) were injected with 800 mg/kg NAC (open blue circles) or equal volumes of saline for control (solid red circles) 15 minutes prior to rVWF injection. (B-D) At baseline and 24 hours after rVWF injection, platelet count (B), hemoglobin level (C), and LDH activity level (D) were determined. (E) H&E staining was performed on myocardial tissue sections from control mice (top panel) and mice with prophylactic NAC administration (bottom panel). Arrows indicate sites of myocardial necrosis. Scale bars indicate 50 μm. (F) VWF multimer composition was analyzed, and representative images of VWF multimeric profiles from a control mouse and an NAC-treated mouse are shown. (G) Percentage of HMW VWF multimers was quantified using densitometry. Statistical significance is indicated with asterisks: *P < .05, **P < .01, ***P < .001.
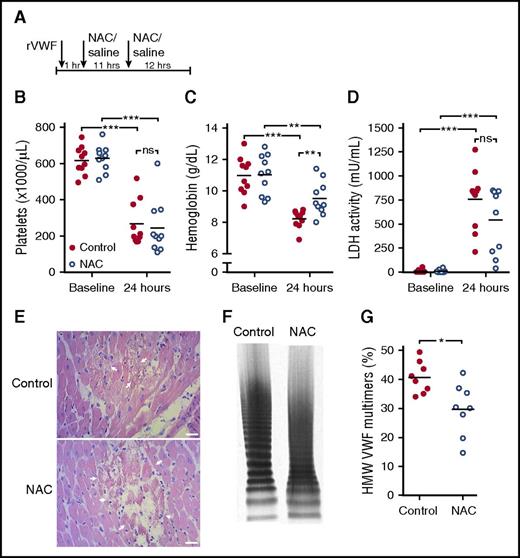
NAC is not effective in reversing TTP signs in Adamts13−/− mice despite a reduction in HMW VWF multimers
During acute TTP episodes, patients require immediate treatment to recover from TTP signs. Therefore, we next investigated the therapeutic potential of NAC for recovery from acute TTP signs in mice. TTP signs were triggered in Adamts13−/− mice with rVWF containing UL VWF multimers, as described above. Signs were manifested within 15 minutes after injection of rVWF.17,23 Treatment with 800 mg/kg NAC (NAC treatment group) or vehicle (control group) was initiated 1 hour and 12 hours after rVWF injection (Figure 3A). Control mice developed TTP signs, as a severe decrease in mean platelet count was observed after 24 hours (Figure 3B). Concomitantly, hemoglobin levels strongly decreased (Figure 3C) and LDH activity was highly elevated (Figure 3D). Organ damage could be detected, as myocardial necrosis was present in heart sections of these mice (Figure 3E, upper panel). A severe decrease in platelet count and a strong increase in LDH levels were also measured in NAC-treated mice, and these values did not significantly differ from the ones in the control group (Figure 3B,D). Myocardial necrosis in NAC-treated mice was observed to a similar extent as in control mice (Figure 3E, lower panel). Hemoglobin levels were also decreased in NAC-treated mice, but they were significantly higher than in control mice (Figure 3C). Although NAC-treated mice did not recover from TTP signs, NAC acted to reduce the size of circulating HMW VWF multimers in mice injected with NAC, compared with HMW VWF multimers in control mice (Figure 3F-G). Together, these findings indicate that although NAC is able to reduce soluble HMW VWF multimers, the reduction is not sufficient to reverse TTP signs.
NAC is not effective in reversing TTP signs in Adamts13−/− mice despite a reduction in HMW VWF multimers. (A) Adamts13−/− mice (n = 10 per group) were injected with rVWF to trigger TTP signs. One hour and 12 hours later, mice were injected with 800 mg/kg NAC (open blue circles) or equal volumes of saline for control (solid red circles). (B-C) At baseline and 24 hours after rVWF injection, platelet count (B), hemoglobin level (C), and LDH activity level (D) were determined. (E) H&E staining was performed on myocardial tissue sections from control mice (upper panel) and mice with prophylactic NAC administration (lower panel). Arrows indicate sites of myocardial necrosis. Scale bars indicate 50 μm. (F) VWF multimer composition was analyzed and representative images of VWF multimeric profiles from a control mouse and NAC mouse are shown. (G) Percentage of HMW VWF multimers was quantified using densitometry. Statistical significance is indicated with asterisks: *P < .05, **P < .01, ***P < .001; ns, not significant.
NAC is not effective in reversing TTP signs in Adamts13−/− mice despite a reduction in HMW VWF multimers. (A) Adamts13−/− mice (n = 10 per group) were injected with rVWF to trigger TTP signs. One hour and 12 hours later, mice were injected with 800 mg/kg NAC (open blue circles) or equal volumes of saline for control (solid red circles). (B-C) At baseline and 24 hours after rVWF injection, platelet count (B), hemoglobin level (C), and LDH activity level (D) were determined. (E) H&E staining was performed on myocardial tissue sections from control mice (upper panel) and mice with prophylactic NAC administration (lower panel). Arrows indicate sites of myocardial necrosis. Scale bars indicate 50 μm. (F) VWF multimer composition was analyzed and representative images of VWF multimeric profiles from a control mouse and NAC mouse are shown. (G) Percentage of HMW VWF multimers was quantified using densitometry. Statistical significance is indicated with asterisks: *P < .05, **P < .01, ***P < .001; ns, not significant.
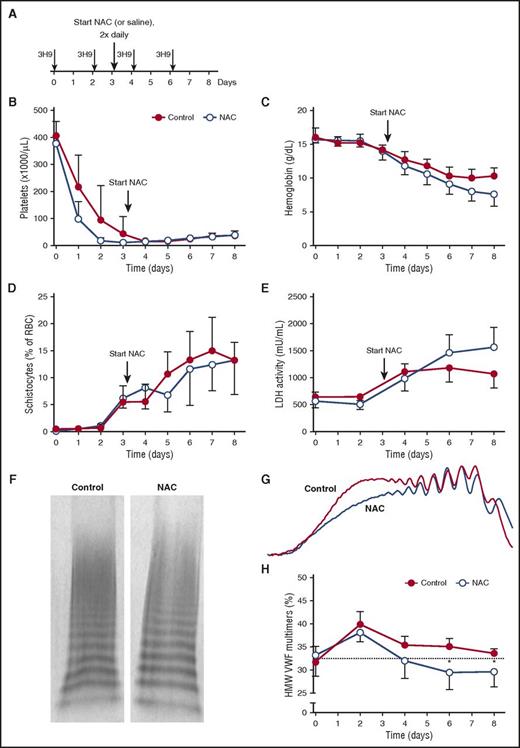
NAC treatment does not resolve TTP signs in a preclinical baboon model
We next studied the effect of NAC on TTP signs in our preclinical baboon model for acquired TTP, as baboons are more closely related to humans than are mice. Acquired TTP was induced by injecting our inhibitory anti-ADAMTS13 monoclonal antibody (mAb) 3H9,20,21 which results in typical TTP signs including severe thrombocytopenia, hemolytic anemia, and tissue damage, without the need for an additional trigger. At day 0, 3H9 injections were initiated, and they were repeated every 48 hours thereafter (Figure 4A), resulting in profound thrombocytopenia, decreased hemoglobin levels, increased schistocytes, and elevated LDH activity at day 3, confirming the presence of TTP as published previously (Figure 4B-E).20,21 After the onset of TTP signs, 4 baboons were treated with NAC and 4 other baboons were treated with saline as a control. NAC was administered by intravenous infusion at a dose of 400 mg/kg over a 30-minute period, with infusions repeated every 12 hours up to day 8 (Figure 4A). Platelet count was determined daily, and the 4 control baboons remained thrombocytopenic during the complete study period (Figure 4B). In addition, their hemoglobin levels decreased (Figure 4C) and the percentage of schistocytes increased (Figure 4D). Accordingly, LDH activity levels increased, demonstrating tissue damage and hemolysis as a result of TTP (Figure 4E). In the 4 baboons treated with NAC, no positive effect was observed, as TTP signs did not resolve. NAC-treated baboons remained thrombocytopenic during the study period (Figure 4B), their hemoglobin levels did not normalize (Figure 4C), the percentage of schistocytes did not decrease (Figure 4D), and LDH activity levels remained elevated (Figure 4E).
NAC treatment does not resolve TTP signs in the preclinical baboon model. (A) Anti-ADAMTS13 antibody 3H9 was injected on days 0, 2, 4, and 6, directly after blood withdrawal. NAC (400 mg/kg) treatment was initiated (large arrow) after blood withdrawal on day 3 and continued twice daily at 12-hour intervals up to day 8 (n = 4; open blue circles). Control baboons were injected with equal volumes of saline (n = 4; solid red circles). (B-C) Platelet count (B) and hemoglobin level (C) were determined daily. (D) Blood smears were made, and the number of schistocytes as a percentage of total red blood cells was counted manually. (E) LDH activity levels were determined. (F) Representative image of the VWF multimers in blood taken from control baboons and NAC-treated baboons on day 7. (G) Densitometric analysis was performed, and the two traces from the VWF multimers in panel F are shown. (H) Using densitometric analysis, the percentage of HMW VWF multimers was quantified for control baboons and NAC-treated baboons. Graphs represent the mean and standard deviation. Statistical significance is indicated with an asterisk: *P < .05.
NAC treatment does not resolve TTP signs in the preclinical baboon model. (A) Anti-ADAMTS13 antibody 3H9 was injected on days 0, 2, 4, and 6, directly after blood withdrawal. NAC (400 mg/kg) treatment was initiated (large arrow) after blood withdrawal on day 3 and continued twice daily at 12-hour intervals up to day 8 (n = 4; open blue circles). Control baboons were injected with equal volumes of saline (n = 4; solid red circles). (B-C) Platelet count (B) and hemoglobin level (C) were determined daily. (D) Blood smears were made, and the number of schistocytes as a percentage of total red blood cells was counted manually. (E) LDH activity levels were determined. (F) Representative image of the VWF multimers in blood taken from control baboons and NAC-treated baboons on day 7. (G) Densitometric analysis was performed, and the two traces from the VWF multimers in panel F are shown. (H) Using densitometric analysis, the percentage of HMW VWF multimers was quantified for control baboons and NAC-treated baboons. Graphs represent the mean and standard deviation. Statistical significance is indicated with an asterisk: *P < .05.
We next analyzed the VWF multimer pattern to demonstrate an effect of the disulfide bond–reducing properties of NAC in vivo (Figure 4F-G). An increase in HMW VWF multimers, demonstrating the presence of UL VWF, was observed at day 2 in both groups, in line with findings in our previous studies.21 UL VWF multimers remained present in the plasma of control baboons during the remainder of the study.21 In the treatment group, however, HMW VWF multimers decreased, demonstrating the reduction of UL VWF as a result of NAC treatment (Figure 4H). Additionally, no differences in prothrombin time (PT) and activated partial thromboplastin time (aPTT) between the two groups were observed (data not shown). As it had done in the murine TTP treatment model, NAC reduced the size of circulating HMW VWF multimers in baboons.
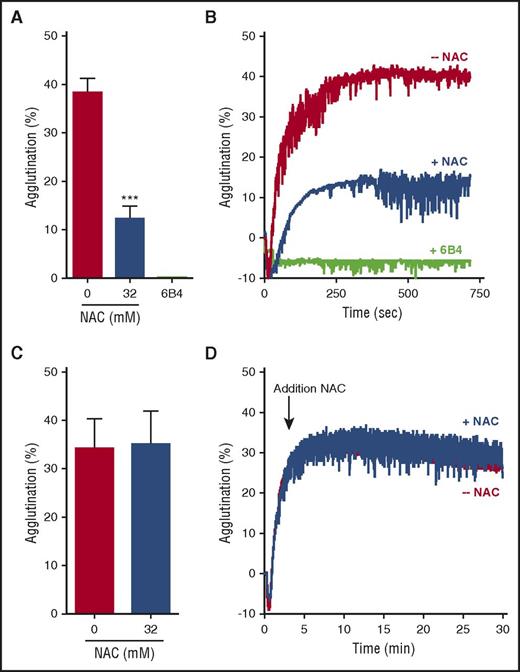
NAC does not dissolve preexisting VWF-rich thrombi in vitro
We have demonstrated that NAC is able to reduce VWF multimers in plasma. Nevertheless, this reduction in circulating VWF multimers was demonstrated not to be sufficient to resolve TTP signs. Because treatment with NAC in mice and baboons was initiated when TTP signs were present, VWF-rich microthrombi had already formed in these animals. We therefore tested whether NAC was able to reduce VWF when present within a VWF-rich thrombus, resulting in thrombus resolution. First, NHP was incubated with 32 mM NAC before platelet agglutination was started by using ristocetin. VWF in plasma was efficiently reduced from 38.3% to 12.3% platelet agglutination, inhibiting normal platelet agglutination (Figure 5A; representative traces are shown in Figure 5B). However, complete inhibition, as observed when using a potent VWF-platelet inhibitor (eg, anti-GP1b antibody 6B416,24 ), was not achieved. These data demonstrate that NAC is less potent in blocking the VWF-platelet interaction, compared with inhibitory anti-GP1b antibodies like 6B4 (Figure 5B)16,24 and inhibitory anti-VWF antibodies like caplacizumab.25 Next, NAC was added to stably formed VWF-rich agglutinates. Despite the ability of NAC to reduce VWF multimers in solution, addition of 32 mM NAC to a stable agglutinate did not result in agglutinate breakdown (Figure 5C, representative traces shown in Figure 5D). Thrombus resolution therefore was not achieved upon addition of NAC, a fact that provides a potential explanation for the unsuccessful recovery from preexisting TTP signs although prophylactically administered NAC prevented the onset of severe TTP signs.
NAC does not dissolve preexisting VWF-rich thrombi in vitro. (A) NHP was incubated with 0 or 32 mM NAC, and plasma was subsequently added to lyophilized platelets (n = 3 per condition). Platelet agglutination was started by addition of ristocetin. Anti-GP1b antibody 6B4 was added at 20 nM to block the VWF-platelet interaction. The percentage of aggregation reached at 12 minutes was used for quantification. (B) Representative traces from 3 different experiments are shown. (C) Platelet agglutination was induced by addition of ristocetin to a mixture of NHP and lyophilized platelets. NAC (0 or 32 mM) was added when the agglutinate was stably formed (indicated with an arrow). The percentage of aggregation reached at 30 minutes was used for quantification. (D) Representative traces from 3 different experiments are shown. Graphs represent the mean ± standard deviation. Statistical significance is indicated with asterisks: ***P < .001.
NAC does not dissolve preexisting VWF-rich thrombi in vitro. (A) NHP was incubated with 0 or 32 mM NAC, and plasma was subsequently added to lyophilized platelets (n = 3 per condition). Platelet agglutination was started by addition of ristocetin. Anti-GP1b antibody 6B4 was added at 20 nM to block the VWF-platelet interaction. The percentage of aggregation reached at 12 minutes was used for quantification. (B) Representative traces from 3 different experiments are shown. (C) Platelet agglutination was induced by addition of ristocetin to a mixture of NHP and lyophilized platelets. NAC (0 or 32 mM) was added when the agglutinate was stably formed (indicated with an arrow). The percentage of aggregation reached at 30 minutes was used for quantification. (D) Representative traces from 3 different experiments are shown. Graphs represent the mean ± standard deviation. Statistical significance is indicated with asterisks: ***P < .001.
Discussion
So far, a limited number of patients suffering from TTP and nonresponsive to plasma exchange therapy have been treated with NAC.9-14 The results of these case reports are variable, however, and always examine the effect of NAC treatment as an adjuvant to standard plasma exchange. Therefore, more studies are needed to demonstrate the effectiveness of NAC to treat TTP. For this reason, we used preclinical animal models to study if administration of NAC, without concurrent plasma infusion or plasma exchange and immunosuppressive therapy, is effective in preventing and/or resolving severe TTP.
Mice deficient in ADAMTS13, either congenital (Adamts13−/−) or acquired (anti-ADAMTS13 monoclonal antibodies26 ), do not spontaneously develop TTP signs. An additional trigger, such as rVWF,17 is required to start the onset of TTP. In the currently used murine model, excessively elevated levels of exogenous VWF triggered TTP signs. Although this experimental setup is not analogous to the disease onset in patients, it is currently not known what triggers acute TTP in humans, and therefore more appropriate mouse models are not yet available. The validity of this murine TTP model was previously established when it was used to demonstrate the therapeutic potential of recombinant ADAMTS13.17,23 This possible new treatment strategy for acute TTP is currently being tested in clinical trials. The baboon model is physiologically more relevant, as the injection of anti-ADAMTS13 monoclonal antibody 3H9 was enough to spontaneously trigger early onset of TTP in baboons. However, no clinical end-points (eg, pain, distress, severe organ failure, or mortality) were reached in this baboon model. The validity of the baboon model was demonstrated with the anti-VWF antibody caplacizumab, which demonstrated promising results in treating TTP in baboons,21,22 a finding recently confirmed in acute TTP patients in the TITAN trial.25 Therefore, even though we understand the clear limitations of these animal models, and although clinical studies are required to make any meaningful extrapolations toward treatment of TTP patients, these limitations do not challenge the validity of our findings.
NAC is a precursor of the amino acid l-cysteine and a precursor in the synthesis of glutathione. With its free thiol group, NAC is able to reduce disulfide bonds. Via this mechanism, it is able to reduce disulfide bonds between mucin polymers, serving as a mucolytic agent.8 As previously reported, NAC is also able to reduce disulfide bonds within VWF, thereby reducing VWF multimer size and removing UL VWF strings from the endothelial surface.7 Both intra- and interchain disulfide bonds are found throughout the entire VWF protein.
Based on these findings, we first investigated whether prophylactic administration of NAC might be able to prevent the onset of TTP. Currently, some patients are preventively treated with plasma infusion or plasma exchange to replenish ADAMTS13 activity, remove ADAMTS13 antibodies, and reduce the thrombotic potential of UL VWF. As the trigger for acute TTP onset is not known, this prophylactic treatment is thus far the only optional preventive strategy. We administered a single dose of NAC to Adamts13−/− mice in which TTP signs were next triggered with rVWF. We found that prophylactic administration of NAC prevented the development of severe signs of TTP. These results are in agreement with the previously published results describing decreased thrombus formation after stimulation of the mesenteric venules by calcium ionophore in Adamts13−/− mice that received prophylactic administration of NAC.7 Unfortunately, due to ethical limitation of the number of baboons we could use, we were not able to test the prophylactic aspects of NAC in the prevention of TTP in the more clinically relevant TTP baboon model. Nevertheless, our current results suggest that NAC might be beneficial when added to plasma infusion in patients with congenital TTP during prophylactic treatment, as NAC reduces the prothrombotic potential of circulating UL VWF. Although plasma infusion remains indispensable to replenish ADAMTS13 activity, adding NAC might reduce the number of plasma infusions needed. Nevertheless, due to the limitations of this mouse model, future clinical case-control studies are necessary to further investigate the prophylactic potential of NAC by comparing the effect of prophylactic plasma infusion or exchange with and without NAC.
We next investigated whether NAC could also be used to reverse ongoing TTP. Eight case reports of patients with acquired TTP have been published thus far and 3 of the 8 patients were unresponsive to NAC treatment.12-15 The other 5 patients demonstrated recovery from TTP signs during treatment that included NAC.9-11 It was suggested that these variable results could be caused by variation in dosing,14 as a high dose of 150 to 300 mg/kg NAC was used in all 5 patients responding to treatment. The NAC dosage protocol used was similar to that used to treat acetaminophen overdose27 : 150 to 300 mg/kg NAC was administered over a period of 24 hours, and patients were treated for several days. However, of the 3 patients without an apparent response, 1 patient also received a high dose of NAC (18 g daily) for several days. To ensure that we administered an adequate dose, we adopted a dosing strategy of 400 mg/kg every 12 hours in the baboon study group. This concentration would correspond to a plasma concentration of 59 mM NAC, taking into account a circulating blood volume of 60 mL/kg and hematocrit of 45%, which is sufficient to reduce VWF, based on the results of our in vitro experiments (Figure 1; supplemental Figure 2). A reduction in circulating UL VWF multimers was indeed observed, demonstrating that the NAC concentration was high enough to exert its reducing effect on VWF. However, a successful recovery from TTP signs with NAC could not be demonstrated in mice or in baboons. Interestingly, our in vitro data provide a possible explanation for the lack of efficacy of NAC to resolve TTP signs in these animal models: once a VWF-rich microthrombus was formed, NAC was not able to break down the preexisting thrombus.
In all clinical case reports, NAC treatment was adjunctive to standard plasma exchange, and in some patients NAC was combined with other drugs, such as rituximab, steroids, or vincristine. Our study demonstrates that NAC alone is not sufficient to resolve TTP signs. Even though these animal models have their limitations, our results suggest that the observed recovery of some patients could be attributed to the effects of concurrent treatment strategies that were applied. It is possible that NAC reduces VWF multimer size to a level sufficient to reduce TTP signs when it is administered adjunctively with plasma infusion or exchange. However, this hypothesis was beyond the scope of our study and should be investigated in a randomized clinical trial. Additionally, as NAC is also known for its antioxidant and free radical–scavenging properties, these could also influence the patient outcome. Indeed, NAC prevents thrombosis not only by reducing VWF multimers but also by acting as a direct reactive oxygen species scavenger.28,29 The redox status of the patient will therefore determine the effect of NAC via its antioxidant and free radical–scavenging properties.
From all available data, it is clear that NAC is effective in reducing the size of VWF multimers in vivo. However, in this study we demonstrated that the reducing capacity of NAC alone (without plasma exchange) seems insufficient to resolve acute TTP signs in preclinical animal models. Our in vitro data suggest that this might be explained by the inability of NAC to break down a preexisting VWF-rich thrombus. Currently, a pilot study is enrolling patients with acute TTP that will be treated with a bolus injection of 150 mg/kg NAC followed by an infusion of 150 mg/kg NAC over 17 hours after plasma exchange (ClinicalTrials.gov ID: NCT01808521). It will be highly interesting to learn the results of this study observing the effects of NAC in combination with plasma infusion or exchange therapy in a larger number of patients.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
C.T. received support as a postdoctoral fellow from the Research Foundation–Flanders (FWO), Belgium (12N0715N). The research also was supported by an FWO–Red Cross grant (G0D1315N) and KU Leuven grants (PF/10/014 and OT/14/071).
Authorship
Contribution: C.T. designed research, performed experiments, collected, analyzed, and interpreted data, and co-wrote the manuscript; J.R. designed research, performed experiments, collected and analyzed data, and co-wrote the manuscript; W.J.V.R., C.D., N.V., I.P., A.V., and S.L. performed experiments, analyzed data, and reviewed the manuscript for scientific content; B.P., H.D., J.A.L., and S.F.D.M. interpreted data and reviewed the manuscript for scientific content; K.V. designed and supervised research, interpreted data, and reviewed the manuscript for scientific content.
Conflict-of-interest disclosure: B.P. is an employee of Shire. The remaining authors declare no competing financial interests.
Correspondence: Karen Vanhoorelbeke, Laboratory for Thrombosis Research, IRF Life Sciences, KU Leuven Campus Kulak Kortrijk, Etienne Sabbelaan 53, 8500 Kortrijk, Belgium; e-mail: karen.vanhoorelbeke@kuleuven.be.
References
Author notes
C.T. and J.R. are joint first authors.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal