Abstract
Background: Allogeneic stem cell transplantation is a potentially curative procedure to a long list of hematological malignancies, but involves substantial risk of morbidity and mortality. Means for accurately predicting outcome and assessing risk are thus greatly needed. The Disease Risk Index (DRI) is a prognostic tool developed and validated by Armand et al. across a wide range of hematological malignancies (Blood 2012, Blood 2014) on cohorts of American patients. The Index stratifies patients into 4 distinct risk groups (low, intermediate, high, very high) and has yet to be validated in an international cohort. We sought to evaluate the validity of the DRI in a large cohort of European patients.
Methods: This was a retrospective validation study on an independent cohort of patients undergoing allogeneic HSCT and reported the European Society for Blood and Marrow Transplantation (EBMT). Patients included had a hematological malignancy and underwent allogeneic transplantation between the years of 2000 and 2015. Risk groups were coded in accordance with the refined DRI (Blood, 2014). Outcomes were evaluated 4 years after the allogeneic HSCT. Overall survival (OS) was calculated with the Kaplan-Meier method. The log-rank test was used for comparisons of Kaplan-Meier curves. Cumulative incidence curves for nonrelapse mortality (NRM) and relapse with or without death were constructed reflecting time to relapse and time to NRM, respectively, as competing risks. The difference between cumulative incidence curves in the presence of a competing risk was tested with the Gray method. The prognostic effect of the DRI strata was estimated using a Cox proportional hazard model for OS and a Fine and Gray model for NRM and relapse.
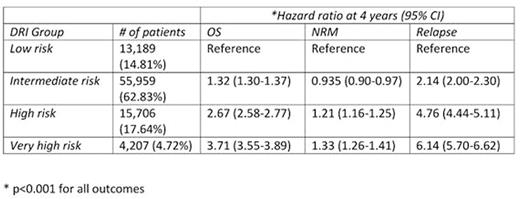
Results: A total of 89,061 patients from 423 transplantation centers were included in the analysis. Median age was 48.3 (IQR 36.2-57.5). The most frequent indication for transplantation was AML (39,530 patients) followed by ALL (16,206) and MDS (9,750); other indications spanned the spectrum of hematological malignancies. The majority of patients were in 1st or 2nd complete remission (54%). The median follow-up period was 3.6 years. Approximately 63% of patients were classified as intermediate risk by DRI, suggesting that this group could be further partitioned. The 4 year overall survival (95% CI) of the low, intermediate, high, and very high risk groups was 60.8% (59.9-61.8), 51.3% (50.8‐51.8), 27.0% (26.1‐27.8), 18.4% (17.1-19.8) (Figure 1). The same groups corresponded with increasing cumulative incidence of relapse; 8.9% (8.3-9.4), 19.3% (18.9-19.7), 39.0% (37.8-39.6), 45.1% (43.4-46.7), respectively. The DRI groups also showed increasing hazard between strata in the overall survival setting; intermediate risk was associated with a hazard ratio of 1.32, high risk 2.67 and very high risk 3.71 relative to low risk. Relapse showed a similar pattern. NRM was less strongly stratified by DRI (Table 1). The DRI groups maintained a similar risk, regardless of whether the transplantation was performed prior or after 2008. DRI was the strongest determinant of overall survival and relapse when introduced to a multivariable model with additional covariates. AUC for the index at 4 years was 62.5 for OS, 58.5 for NRM and 68.2 for relapse.
Conclusions: We have validated the Disease Risk Index in a massive European data set. The groupings suggested by the DRI corresponded with distinct risk groups for overall mortality and relapse. Overall, our results indicate the international applicability of this robust prognostic tool.
Kaplan-Meyer survival curves for overall survival, stratified by DRI
Bader:Medac: Consultancy, Research Funding; Riemser: Research Funding; Neovii Biotech: Research Funding; Servier: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Bonini:Molmed SpA: Consultancy; TxCell: Membership on an entity's Board of Directors or advisory committees. Dreger:Gilead: Consultancy; Janssen: Consultancy; Novartis: Speakers Bureau; Gilead: Speakers Bureau; Novartis: Consultancy; Roche: Consultancy. Kuball:Gadeta B.V,: Membership on an entity's Board of Directors or advisory committees. Montoto:Roche: Honoraria; Gilead: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal