Abstract
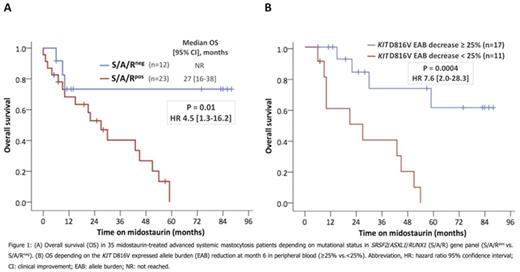
Systemic mastocytosis (SM) arises as a consequence of aberrant activation of KIT signaling, most commonly by acquisition of the KIT D816V mutation (>80-90% of patients). A recently reported phase-II-study exploring the efficacy and safety of midostaurin, a multi-targeted KIT inhibitor, in patients with advanced SM (advSM) has demonstrated major and partial responses in 60% of patients (Gotlib et al., NEJM 374, 2016). However, recent data have highlighted that the molecular pathogenesis of SM is complex. In particular, additional mutations in SRSF2, ASXL1 and/or RUNX1 (S/A/Rpos), seen in 60-70% of patients with advSM, have a significant adverse impact on disease phenotype and prognosis (Jawhar et al., Leukemia 30, 2016). In the current study, we evaluated the impact of molecular markers on response, resistance, progression and overall survival (OS) in midostaurin-treated advSM patients: 1) the S/A/R mutation profile at diagnosis, 2) the reduction of the KIT D816V allele burden (EAB) and 3) the dynamics of the clonal architecture. Between 2009 and 2015, 38 patients (68% male; median age 67 years; range, 48-76) with advSM [SM with an associated hematological neoplasm (SM-AHN), n=22; aggressive SM (ASM), n=3; mast cell leukemia (MCL), n=4; MCL-AHN, n=9] received midostaurin at our institution and were monitored for the KIT D816V EAB by quantitative RT-PCR (RT-qPCR). In addition, serial Next Generation Sequencing (NGS) analyses were performed in 16/25 multi-mutated patients. Median time from diagnosis of advSM to start of midostaurin was 12 months (range, 1-60). Twenty-one of 38 (55%) patients died; median OS was 29 months (range, 0-88) from start of midostaurin and 40 months (range, 5-115) from diagnosis. Three patients (8%) stopped midostaurin early (median 2 months; range, 1-4) because of intolerance. In the remaining 35 (92%) patients, median treatment duration was 13 months (range, 1-88). Progression/death occurred in 6/35 (17%) patients within the first 6 months and 5 (83%) patients were S/A/Rpos. Overall response rate (ORR) according to IWG-MRI-ECNM consensus criteria and OS were significantly different between S/A/Rpos (n=23) vs. S/A/Rneg (n=12) patients [ORR: 35% vs. 75%, P=0.01; OS: P=0.01, HR 4.5 (1.3-16.2), Figure 1A]. The maximal median reduction of KIT D816V EAB in 28 patients treated for >6 months was -29% (range, -100 to 71). Depending on the KIT D816V EAB at month 6, patients were classified as responders (≥25%, n=17) or non-responders (<25%, n=11). Responders were significantly associated with a longer median time on midostaurin (25 vs. 9 months, P=0.01) and achievement of any clinical response according to IWG-MRI-ECNM criteria (13/17 vs. 2/11 patients, P=0.006). All 4 patients who lost their KIT D816V EAB response were S/A/Rpos. In univariate analyses of multiple clinical and molecular response parameters at month 6, three parameters were significantly associated with inferior OS: reduction of KIT D816V EAB <25% (P=0.0004), serum tryptase <50% (P=0.03) and alkaline phosphatase <50% (P=0.04). In multivariate analysis, only KIT D816V EAB reduction <25% remained an independent poor prognostic marker for reduced OS (P=0.004, HR 6.8 [1.8-25.3]) and was superior to the IWG-MRI-ECNM consensus criteria (P=0.005, HR 4.7 [1.5-15.5]) (Figure 1B). Serial NGS analysis of 7 deceased patients revealed acquisition of additional mutations in RUNX1 (n=2), K/NRAS (n=3), IDH2 (n=1) or NPM1 (n=1) and/or increasing mutation allele burdens while KIT D816V EAB was either low (n=2), stable (n=1) or increasing (n=4). In contrast, two patients acquired a JAK2 V617F mutation but remained in remission on midostaurin. In summary, we have found that S/A/Rneg at diagnosis and reduction of the KIT D816V EAB ≥25% at month 6 were the most favorable predictors for survival in midostaurin-treated advSM patients. S/A/Rpos at diagnosis was associated with a more aggressive phenotype, (early) resistance/progression and poor survival. Serial sequencing during treatment also revealed that secondary resistance/transformation may be caused by expansion of sub-clones exhibiting new mutations in critical target genes independent of KIT D816V. In addition to established response parameters, sequential analysis of KIT D816V EAB and other molecular aberrations add relevant information for optimal management and response assessment in midostaurin-treated patients with advSM.
Valent:Celegene: Honoraria, Research Funding; Novartis: Honoraria, Research Funding; Amgen: Honoraria. Meggendorfer:MLL Munich Leukemia Laboratory: Employment.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal