Abstract
Introduction: In chronic myeloid leukemia (CML), tyrosine Kinase Inhibitors (TKIs) treatment is a potentially life-time therapy for the majority of patients (pts), as few of them, only after achieving a deep and stable molecular response, may discontinue TKIs without recurrence of disease. Available data suggest that relapse after TKIs discontinuation is due to the persistence of leukemic stem cells (LSCs) intrinsically resistant to TKIs. Survival of CML LSCs may be the consequence of activation of several pathways BCR-ABL1 independent. qRT-PCR, the most sensitive assay to monitor disease status in CML pts, may be inappropriate to quantify residual quiescent CML LSCs that are transcriptionally silent. Therefore, the possibility to easily quantify LSCs during TKIs treatment is a great opportunity to better understand the behavior of residual LSCs and potentially to identify those pts candidates to safely discontinue TKIs. Recently, Valent et al described that CD34+/CD38-/Lin- CML LSCs specifically co-express dipeptidylpeptidase IV (CD26) and that CD26 is a potential biomarker for the quantification and isolation of CML LSCs, in bone marrow samples of CML patients. Furthermore, Culen et al. quantified CD26+ LSCs bone marrow compartment in 31 CML patients at diagnosis and their number appears to correlate with response to TKIs treatment. In the present study we wanted to explore the feasibility, rate and potential implication of detecting CD26+ LSCs in peripheral blood (PB) from CML pts during TKI treatment.
Methods: CML pts during first line treatment with any approved TKIs, referring to several Italian Hematology Centers, entered this non interventional cross sectional study after signing a proper informed consent. During a routine follow up visit, in which pts were checked for molecular response by standard PB qRT-PCR BCR-ABL1 analysis, additional 3 mls of PB were collected in EDTA and sent within 24 hours to Siena Hematology Lab to detect CD34+/CD38-/CD26+ LSCs by multicolor flow cytometry. After red blood cells lysis, cells were incubated with anti CD45 (BD Biosciences), CD34 (581), CD38 (HIT2), CD26 (M-A261) (BD Pharmigen). After washing, acquisition and analysis were performed by FACSCanto II (BD Biosciences, NR Nannini) using DIVA 8 software (BD, Biosciences). CD45+ cells acquired for each sample ranged from 500,000 to 1,000,000. Isotype controls were included in each staining. In 5 pts a FISH analysis of PB sorted CML LSCs population was also performed.
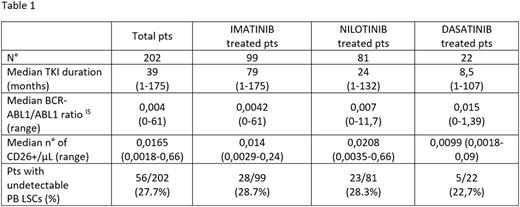
Results: to validate our assay we first performed a FISH analysis of both PB sorted CD34+/CD38-/CD26+ and CD34+/CD38-/CD26- in 5 CML patients at 3-6 months after starting treatment, confirming Ph+ cells only in the CD26+ fraction. Afterward, we checked for circulating CML LSCs a total of 202 CML pts in first line treatment with TKIs for a median of 39 months (range 1-175). Type of TKI, length of treatment, molecular response and quantification of LSCs are summarized in Table 1. PB CML LSCs were detectable in 146/202 (72.3%) pts with a median number of CD26+ of 0,0165 cells/µL (range 0,0018-0,66). Kendall rank correlation coefficient used to analyze the relation between the measurable variables showed no correlation between BCR-ABL/ABLIS ratio (median 0,004 range 0-61) and number of residual LSCs (r 0.118 p=0.097). In 56/202 (27.7%) pts CD26+ LSCs were undetectable, yet we found no correlation with the concomitant degree of molecular response.
Conclusions: this study represents the first attempt to measure in a large cohort of CML patients residual circulating LSCs during TKIs treatment. In our hands PB LSCs flow-cytometry assay appeared feasible, specific and sensitive and thus suitable for routine monitoring. As expected, the majority of CML patients, even in deep molecular response, still harbor residual LSCs and the number of PB CD26+ did not correlate with the number of BCR-ABL1 copies. This evidence suggests that the molecular response refers to transcriptionally active CML progenitor cells and not to quiescent, TKIs resistant, CML LSCs. Prospective studies evaluating the behavior of PB CML LSCs during different TKIs treatment, as well as studies monitoring PB CD26+ in CML pts that discontinued TKIs treatment are ongoing. Our goal is to rule out the impact, if any, of a "stem cell response" in addition to the standard molecular response in the management of CML patients mainly to identify those pts candidates for a safe TKI discontinuation.
Bocchia:Janssen: Honoraria; Novartis: Honoraria; Bristol-Myers Squibb: Honoraria. Aprile:Novartis: Honoraria. Castagnetti:Pfizer: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; ARIAD Pharmaceuticals: Consultancy, Honoraria; Novartis: Consultancy, Honoraria. Tiribelli:Ariad Pharmaceuticals: Consultancy, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Bristol-Myers Squibb: Consultancy, Speakers Bureau. Breccia:Novartis: Consultancy, Honoraria; Bristol Myers Squibb: Honoraria; Celgene: Honoraria; Ariad: Honoraria; Pfizer: Honoraria. Rosti:Roche: Honoraria, Research Funding, Speakers Bureau; Incyte: Honoraria, Research Funding, Speakers Bureau; Pfizer: Honoraria, Research Funding, Speakers Bureau; BMS: Honoraria, Research Funding, Speakers Bureau; Novartis: Honoraria, Research Funding, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal