Abstract
Background: Mutations in the metabolic enzyme isocitrate dehydrogenase 1 (IDH1) are frequently found in patients with acute myeloid leukemia (AML) and several other tumors. Mutant IDH1 produces R-2-hydroxyglutarate (R-2HG), which induces histone and DNA hypermethylation through inhibition of epigenetic regulators, and leads to a block in differentiation to promote tumorigenesis.
Methods: We developed a novel, highly active oral pan-IDH1 inhibitor, BAY-1436032, for clinical evaluation. Its inhibitory potency was evaluated in primary human AML cells in vitro for the five major IDH1R132 mutation types and in two patient derived AML xenograft (PDX) models in vivo, in which BAY-1436032 cleared leukemic blasts in peripheral blood and prolonged survival by induction of differentiation and inhibition of leukemia stem cell self-renewal.
Results: R-2HG production by mutant IDH1 was effectively inhibited in patient derived AML cells with all reported IDH1R132 mutations ex vivo by BAY-1436032 with an IC50 between 3 to 16 nM. AML cells cultured ex vivo showed morphologic differentiation and marked upregulation of the myeloid differentiation markers CD14 and CD15.
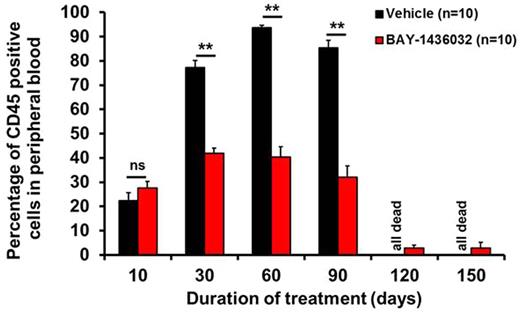
For in vivo experiments, human AML cells from two patients were transplanted into sublethally irradiated NSG mice. After stable engraftment at 17 (PDX1) or 90 (PDX2) days post transplantation, mice were treated with BAY-1436032 orally every day at a dose of 150 mg/kg or vehicle for 100-150 days (n=10 per group). The R/S-2HG ratio in serum was reduced to near normal levels by BAY-1436032. Leukemic cell counts in peripheral blood constantly increased in control mice, while leukemic cells declined from day 30 of BAY-1436032 treatment onwards with morphologic and immunophenotypic evidence of differentiation (Figure). Importantly, all BAY-1436032 treated PDX1 mice survived until the end of treatment at 150 days. In contrast, vehicle-treated mice died with a median latency of 91 days (range 70-95, P<.001). In an independent second model (PDX2) 6 of 10 BAY-1436032 treated mice survived until the end of treatment at day 100 with a median of 15% leukemic cells in peripheral blood, while all vehicle-treated mice suffered from high leukemic burden and died from leukemia with a median survival of 62 days (P=.014). Early mortality was increased with 4 mice dying in the BAY-1436032 group reminiscent of clinical differentiation syndrome in AML patients treated with the IDH1 inhibitor AG-120.
To assess the effect of BAY-1436032 on leukemic stem cell self-renewal we treated PDX1 mice with 150 mg/kg BAY-1436032 or vehicle for 4 weeks and performed a limiting dilution transplantation experiment in secondary recipient mice. LSC frequency was 100-fold lower in BAY-1436032 treated compared to control mice. Gene expression profiling showed that stemness associated genes were downregulated, while genes associated with myeloid differentiation like PU.1 and CEBPA were upregulated upon treatment with BAY-1436032. In addition, cell cycle progression was slowed and E2F transcription factors concomitantly inhibited. In accordance with gene expression profiling results, methylation of the PU.1 promoter decreased, while E2F1 promoter methylation increased upon treatment with BAY-1436032. Finally, histone trimethylation levels at residues H3K4, H3K9, H3K27, and H3K36 decreased in both IDH1R132C and IDH1R132H mutant AML cells but not in IDH1 wildtype cells upon BAY-1436032 treatment.
Conclusion: In summary, the novel oral pan-mutant IDH1 inhibitor BAY-1436032 is active against all IDH1R132 mutation types and shows strong anti-leukemic activity in two independent AML PDX mouse models. Clinical development is ongoing with a first in man study with BAY-1436032 in IDH1 mutant solid tumors.
* M. Heuser and L. Herbst contributed equally to this article
#A. Krämer and A. Chaturvedi share senior authorship
Human leukemic cells in peripheral blood of mice treated with BAY-1436032. ** P<.001, ns, not significant.
Human leukemic cells in peripheral blood of mice treated with BAY-1436032. ** P<.001, ns, not significant.
Heuser:Tetralogic: Research Funding; BerGenBio: Research Funding; Karyopharm Therapeutics Inc: Research Funding; Bayer Pharma AG: Research Funding; Celgene: Honoraria; Novartis: Consultancy, Research Funding; Pfizer: Research Funding. Pusch:German Cancer Research Center: Patents & Royalties: WO2013/127997A1. Kaulfuss:Bayer Pharma AG: Employment. Panknin:Bayer Pharma AG: Employment. Zimmermann:Bayer Pharma AG: Employment, Patents & Royalties: WO2015/121210 . Toschi:Bayer Pharma AG: Employment. Neuhaus:Bayer Pharma AG: Employment, Patents & Royalties: WO2015/121210. Haegebarth:Bayer Pharma AG: Employment, Equity Ownership. Rehwinkel:Bayer Pharma AG: Employment, Equity Ownership, Patents & Royalties: WO2015/121210. Hess-Stumpp:Bayer Pharma AG: Employment. Bauser:Bayer Pharma AG: Employment. Ho:Sanofi-Aventis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. von Deimling:German Cancer Research Center: Patents & Royalties: IDH1R132H mutant specific antibody H09; BRAF V600E mutant specific antibody VE1; BAY-1436032 patent.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal