Abstract
Introduction: The complement system can be activated via three pathways: classical pathway (CP), lectin pathway (LP) and alternative pathway (AP). While the CP and LP are triggered in the solid phase upon interaction of a pattern-recognition molecule with a target surface, the AP can be activated in the fluid phase. In fact, under normal physiological conditions, the AP is constitutively activated at a low level in the fluid phase via a mechanism of "C3 tickover" which leads to production of the C3b that can bind covalently to adjacent target cells or its binding activity is lost very rapidly. On the normal red cells, the bound C3b molecules are rapidly inactivated by an array of membrane-expressed or fluid phase-recruited complement regulators. However, due to the deficiency of two membrane-expressed negative regulators CD55 and CD59, the bound C3b molecules on the PNH red cells are not only amplified via the AP loop but also proceed to form the C3 convertase, the C5 convertase and ultimately the membrane attack complex, which causes hemolysis of PNH red cells. Hence, the blockade of both AP activation in the fluid phase and AP amplification on the surface of PNH red cells with a complement factor D (CFD) inhibitor is expected to be efficacious for PNH indication since hemolysis is due to the constitutive fluid phase AP activation.
In this report, we evaluated whether hemolysis of PNH red cells occurs under the conditions in which the CP, LP and/or AP are activated by various pathogens in vitro. Our objective was to determine whether there is "bystander" hemolysis due to massive pathogen-driven complement activation and the potential for resulting C3b to bind PNH red cells and lead to complement-mediated hemolysis through AP amplification.
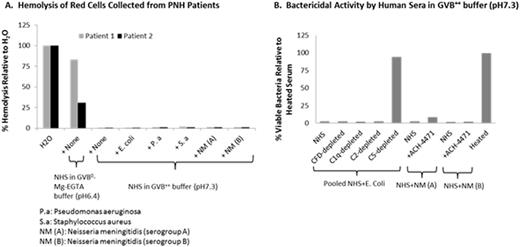
Methods: Blood was obtained from PNH patients with written informed consent. Pathogen inoculums, such as E. coli and Neisseria meningitidis(NM), were prepared by standard methods. Hemolysis of PNH red cells was assessed with ~80% ABO blood type-matched pooled normal human serum (NHS) in GVB0-Mg-EGTA buffer (pH6.4) as well as in GVB++ buffer (pH7.3) in the presence and absence of a small molecule CFD inhibitor (ACH-4471). The extent of bystander hemolysis of PNH red cells was assessed with ~80% ABO blood type-matched pooled NHS in GVB++ buffer (pH7.3) for all pathogens for all pathogens except the NM isolates, where individual sera with bactericidal activity were used. Parallel evidence of complement activation by the pathogens was evaluated by 1) bactericidal activity of E. coli with pooled NHS-depleted of C1q, C2, CFD or C5 in GVB++ buffer (pH7.3), 2) bactericidal activity of NM isolates with the sera in GVB++ buffer (pH7.3) in the presence of ACH-4471, a CFD inhibitor; 3) measurement of the complement components and their activation products.
Results: The extent of hemolysis of red cells harvested from PNH subjects was approximately equal to the Type II/III clone size when assessed with NHS in GVB0-Mg-EGTA buffer (pH6.4) ranging from ~30% to 90%; hemolysis was effectively blocked by ACH-4471, consistent with the data shown previously(Gavriilaki et. al. ASH 2015, Abstract No 275). No significant hemolysis of red cells harvested from PNH subjects was seen when assessed with NHS in GVB++ buffer (pH7.3) and furthermore hemolysis was not increased upon addition of bacteria (Fig. A). For E. coli, the bactericidal activity was unaffected by disruption of the CP (C1q depletion), the AP (CFD depletion), the CP and LP (C2 depletion) although, as expected, bactericidal activity was abrogated by disruption of the terminal pathway (C5 depletion) (Fig. B), confirming that complement activation was initiated via multiple pathways by E. coli. For NM isolates, the bactericidal activity was unaffected by disruption of the AP (Fig. B), confirming that the complement activation is initiated via CP or LP by NM isolates. Complement activation for other pathogens will be presented.
Conclusion: We demonstrated that PNH red cells were not subjected to theoretical "bystander hemolysis" when incubated with the bacteria tested herein, suggesting no increased risk of pathogen-induced hemolytic breakthrough in PNH patients if treated with a complement alternative pathway inhibitor.
Yang:Achillion: Employment, Equity Ownership. Thanassi:Achillion: Employment, Equity Ownership. Galvan:Achillion: Employment, Equity Ownership. Podos:Achillion: Employment, Equity Ownership. Huang:Achillion: Employment, Equity Ownership. Brodsky:Achillion Pharmaceuticals: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Apellis Pharmaceuticals Inc: Membership on an entity's Board of Directors or advisory committees; Alexion Pharmaceuticals Inc: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal