Abstract
Background: PKD causes a defect in glycolysis resulting in a hereditary non-spherocytic hemolytic anemia. The prevalence of iron overload is not well described for PKD.
Aim: We aim to describe the demographic features and prevalence of iron overload in transfusion dependent and transfusion independent patients with PKD.
Methods: Between March 2014 and April 2016, 203 patients enrolled on the PKD Natural History Study at 29 IRB approved sites. All patients were confirmed to have two compound heterozygous or homozygous mutations in the PKLR gene. Children < 1 year of age (n=9) were excluded from this analysis, because elevated ferritin levels are less reliably related to iron overload. Patients were designated with iron overload at the time of enrollment if the plasma ferritin was >1000 ng/mL or the patient was on chelation therapy at any time during the prior 12 months. Patients were designated with having had iron overload if a MRI ever showed liver iron content (LIC) >3 mg/g dry weight or if they had ever received chelation therapy. Tests of association were performed using Fisher's exact test (categorical) and Wilcoxon rank sum test (continuous). Linear associations between variables were measured by Pearson correlation coefficient. P-values <0.05 were considered statistically significant.
Results: Of the 194 patients, 111 (57%) were adults ≥18 years and 83 (43%) were children. The median age of enrollment was 20.6 y (range: 1.3-69.9). Splenectomy had been performed in 65% (126/194). Screening ferritin levels were available for 72% (140/194) and LIC for 32% (62/194). At enrollment, 48% (70/147) had iron overload as defined by ferritin and/or current chelation. Using the LIC criterion, iron overload had been present at some point in 86% (95/110) of patients. Ferritin positively correlated with LIC (n=45); r=0.62, p<0.0001. However, of 29 patients with an LIC measurement and a mean ferritin <1000 ng/mL, 15 (52%) had a LIC >3 mg/g DW.
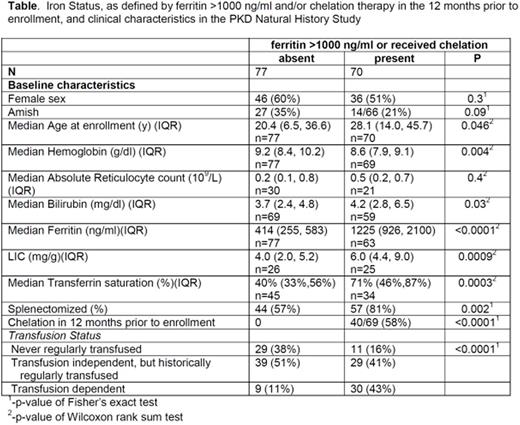
Baseline characteristics in patients with and without iron overload are shown in the Table. Notably, even in patients that were never regularly transfused and had a hemoglobin (Hb) >8.7 g/dl, the prevalence of iron overload was 26% (8/31). The frequency of iron overload was significantly higher in patients who had a prior splenectomy (p<0.0001), even after controlling for transfusion history (p<0.0001). Age was associated with iron overload (p=0.046); although, the age range of patients with iron overload was broad (1.3-69.9 years). The frequency of iron overload was significantly higher in those with a lower baselineHb(p=0.004) and higher bilirubin (p=0.03).
Data on cardiac iron status was available for 66 patients.Only 2 had cardiac iron overload (defined as T2*<20ms); they were age 5 (T2* 17.8ms, LIC 5 mg/g) and 22 years (T2* 19.7ms, LIC 14 mg/g) at the time of the MRI.
Of the 194 patients, 52 (27%) were from the Pennsylvania Amish community. These patients were managed differently than the non-Amish, in that only 2% of the Amish patients were on iron chelation therapy in the 12 months prior to enrollment compared with 43% among the non-Amish cohort. In addition, the Amish had a significant higher prevalence of splenectomy (96% vs 52%, p<0.0001) and proportion who had been historically transfused (79% vs 32%, p<0.0001). Despite these differences, the Amish patients had a lower prevalence of iron overload (34% vs. 51%). There was no significant difference inHbor enrollment age between the Amish and non-Amish cohorts.
Conclusion: Iron overload is a common, serious complication in PKD, regardless of age, disease severity, or transfusion status. Although ferritin correlates with LIC for the PKD population overall, at the individual patient level, ferritin is not a good predictor of LIC and a ferritin <1000 ng/ml does not exclude hepatic iron overload. Therefore, we recommend that all patients with PKD starting at age 1 year should be screened annually for iron overload using ferritin and, at least once, using MRI.
Barcellini:Agios: Consultancy. Neufeld:Novartis: Consultancy. Morton:Agios Pharmaceuticals: Research Funding. Yaish:Octapharma: Other: Study investigator. Kuo:Agios Pharmaceuticals, Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees; Alexion: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Apotex: Other: unrestricted educational grant; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Thompson:bluebird bio: Consultancy, Research Funding; Eli Lily: Research Funding; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Mast: Research Funding; Baxalta (now part of Shire): Research Funding; ApoPharma: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding; Amgen: Research Funding. Grace:Agios Pharmaceuticals: Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal