In this issue of Blood, Kariminia et al report that serum CXCL10 is the only consistent biomarker for chronic graft-versus-host disease (cGVHD) and is associated with low peripheral blood levels of CXCR3+ natural killer (NK) cells.1
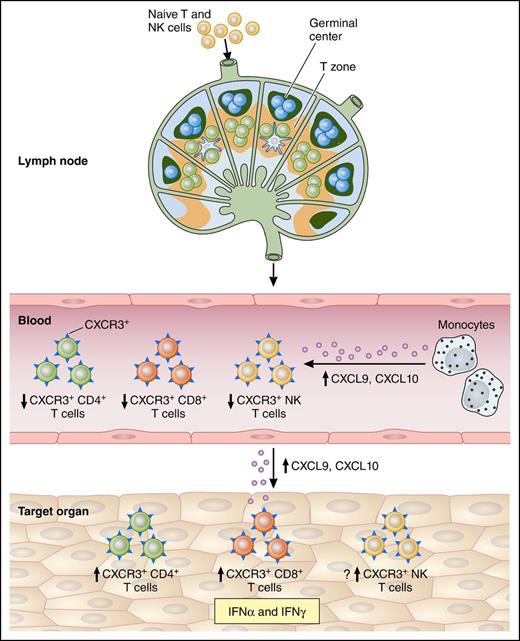
Possible involvement of CXCL9 and CXCL10 in cGVHD pathogenesis. Naive lymphocytes (T and NK) traffic to local lymph nodes, where they recognize recipient minor antigen (mAg) presented by antigen-presenting cells in the T-cell zone. In parallel, B cells are activated in the germinal center. Primed naive cells convert to mAg-specific effector cells and exit the lymph nodes. Upon CXCL9 and CXCL10 release by circulating monocytes, the activated lymphocytes acquire CXCR3 on their surface leading to their recruitment in the target tissues via a CXCL9/CXCL10 gradient and a decrease of their frequencies in the blood. CD4/CD8+ T cells and possibly NK cells recognize recipient mAg expressed on major histocompatibility complex (MHC) I and MHC II or a to-be-determined NK receptor expressed by the epithelium, respectively. Then, effectors will kill tissue targets, promoting local cytokine and chemokine production, and setting up a cycle of tissue destruction and further effector recruitment. Professional illustration by Patrick Lane, ScEYEnce Studios.
Possible involvement of CXCL9 and CXCL10 in cGVHD pathogenesis. Naive lymphocytes (T and NK) traffic to local lymph nodes, where they recognize recipient minor antigen (mAg) presented by antigen-presenting cells in the T-cell zone. In parallel, B cells are activated in the germinal center. Primed naive cells convert to mAg-specific effector cells and exit the lymph nodes. Upon CXCL9 and CXCL10 release by circulating monocytes, the activated lymphocytes acquire CXCR3 on their surface leading to their recruitment in the target tissues via a CXCL9/CXCL10 gradient and a decrease of their frequencies in the blood. CD4/CD8+ T cells and possibly NK cells recognize recipient mAg expressed on major histocompatibility complex (MHC) I and MHC II or a to-be-determined NK receptor expressed by the epithelium, respectively. Then, effectors will kill tissue targets, promoting local cytokine and chemokine production, and setting up a cycle of tissue destruction and further effector recruitment. Professional illustration by Patrick Lane, ScEYEnce Studios.
CXCL9 and CXCL10 are interferon (IFN)-inducible chemokines that bind to CXCR3, their only known receptor. Both IFNα and IFNγ can induce these chemokines. CXCR3 expression can be rapidly induced in a variety of immune cells, including T lymphocytes and NK cells, following dendritic cell activation of naive lymphocytes.2 Binding of these chemokines to CXCR3 promotes lymphocyte migration to inflamed tissues. CXCR3 is also critical for the recruitment of alloreactive T cells in acute GVHD.3
There is a critical need for biology-based biomarkers to confirm or aid the diagnosis of cGVHD after allogeneic hematopoietic stem cell transplantation or monitor its progression to facilitate evaluation of new therapies.4 The 2014 Biomarker Working Group of the National Institutes of Health Consensus Development Project on criteria for trials in cGVHD established a critical framework for biomarker studies that includes 4 distinct steps and is designed to avoid previous mistakes.4 The first step is identification in discovery experiments of candidate biomarkers of well-matched cases and controls for a specific outcome (eg, diagnosis of all cGVHD, diagnosis of skin cGVHD, predicting future occurrence of cGVHD at an early time point such as day 100 posttransplant). The second is verification of the test’s analytical validity including reproducibility and accuracy. At this stage, cutoff values and sample collection procedures are locked down and cannot be changed without reverification of the test under the revised conditions. The third step is qualification to assess the robustness of the test in all-comer samples from the intended use population for the specific clinical outcome as used in the discovery experiments. The qualification cohort for step 3 should be independent from the identification cohort previously studied in step 1. The final step is application in the clinic.
In the present study, Kariminia et al evaluate previously identified potential biomarkers for cGVHD and search for other markers through discovery approaches followed by validation of 11 candidates in 2 independent cohorts (n = 198 and n = 83). In their series, both CXCL9 and CXCL10 are significantly correlated in multivariate analysis with cGVHD diagnosis in the first replication cohort, but only CXCL10 in the second. In addition, other candidates are significant in 1 but not both the replication cohorts. They also study, on a subcohort of patients, the correlation between CXCL9/CXCL10 and circulating CXCR3+ immune cells (CD4+, CD8+, and CD3−CD56bright). They find CXCR3+ NK cells are significantly lower in the peripheral blood of patients with cGVHD. This population’s frequency cannot be used as a “biomarker” at this point because it is only tested in a subset of patients without using the strict criteria described herein, but it gives a hint in terms of possible biological correlation. Additionally, the frequency of these cells in affected tissues, which should be increased, is not tested as previously done by Croudace et al in the case of T lymphocytes.5
Several groups have now reported CXCL9 or CXCL10 as potential biomarkers of cGVHD with more or less rigor in following the biomarker criteria described herein.5-7 Hakim et al recently and elegantly reported gene expression profiling of circulating monocytes from cGVHD patients. They found significant upregulation of IFN-inducible (including CXCL10) and damage-response genes in cGVHD patients as compared with controls. These pathways were further substantiated in plasma enzyme-linked immunosorbent assays showing elevated levels of CXCL9 and CXCL10 (see figure).8 Altogether, the IFN-inducible chemokines CXCL9 and CXCL10, responsible for CXCR3-expressing Th1/NK lymphocyte recruitment into tissues, are upregulated at diagnosis5,6,8 and are worth being pursued and tested in prospectively collected samples with the rigorous criteria mentioned herein. In addition, IFNs enhance the production of another recognized cGVHD biomarker, the homeostatic B-cell cytokine B-cell activating factor (BAFF) and thus may contribute to B-cell activation during cGVHD development.9
Many questions related to CXCL10 and CXCR3+ cells remain unanswered and we would argue against the notion that CXCL10 is the most consistent biomarker for cGVHD due to the following reasons: (1) there are differences in sample collection timing and techniques between the 2 replication cohorts which could affect biomarker levels. For example, timing of sample acquisition relative to the onset of cGVHD caused significant differences of median CXCL9 concentrations.6 (2) Some samples from cGVHD patients were collected after diagnosis. It is not clear whether some of these patients were already on glucocorticoids that could also affect biomarker levels (ie, BAFF4 ). (3) It is biologically evident that these proteins augment each other’s function. Therefore, a biomarker panel would likely be more accurate in predicting outcomes. This is evident from Figure 2B in the study by Kariminia et al where CXCL9 and CXCL10 combination has a higher area under the receiver operating characteristics curve than either biomarker alone. (4) Most important, clear cutoff values with high positive and negative predictive values for patients at high or low risk of developing cGVHD have not been defined or validated.
cGVHD is a complex and heterogeneous immunologic disease which complicates the study of its biomarkers. Several important advances have been made in the field so far but what is needed next? First, the diagnostic value of CXCL9 and CXCL10 should be validated in large prospective multicenter cohorts with careful consideration to all different covariate and biomarker criteria. Second, the prognostic and predictive value of these chemokines should be tested. Third, the role of these biomarkers in monitoring the response to therapy needs evaluation and the timing of measurement of markers should be optimized. Last, the correlation between these markers and some other important clinical outcomes should be studied, including cGVHD severity, nonrelapse mortality, and/or overall survival.
In conclusion, this study further confirms our perception that the CXCL9/CXCL10/CXCR3 axis is important for the pathophysiology of cGVHD. The closer a biomarker relates to the underlying pathology, the more reliably it can serve as a marker. Future approaches focusing on abrogation of the interaction between these chemokines and CXCR3 may prove successful in preventing or treating cGVHD.10
Conflict-of interest disclosure: S.P. has a patent on “Methods of detection of graft-versus-host-disease” licensed to Viracor-IBT Laboratories. M.A.Z. declares no competing financial interests.