Abstract
Only 5-azacytidine (5Aza) and decitabine (Dec), both DNA methyltransferase (DNMT1) depleting drugs, are FDA-approved to treat all myelodysplastic syndrome (MDS) subtypes. Neither agent is curative. What are the mechanisms of resistance? MDS is genetically heterogeneous, thus, this question has been approached by attempting to correlate MDS genetics with response, or by examining genetics at relapse. Unfortunately, results have been contradictory/inconclusive. Another approach is to consider that achievement of the intended molecular pharmacodynamic (PD) effect of DNMT1-depletion is a minimum requirement for response, superceding other biological considerations including genetics/susceptibility to apoptosis or differentiation. As such, a logical first-step is to examine whether relapse is driven by sub-clones thriving despite DNMT1-depletion, or from failure to deplete DNMT1 in the first place. In both murine and clinical studies, relapse was from failure to deplete DNMT1 (Leukemia 2011;25(11):1739-50; Oncotarget 2012;3(10):1137-45; J Clin Invest 2015;125(3):1043-55). Thus, the question can be reframed as: Why is molecular PD not achieved, and can mechanisms be identified to extend response?
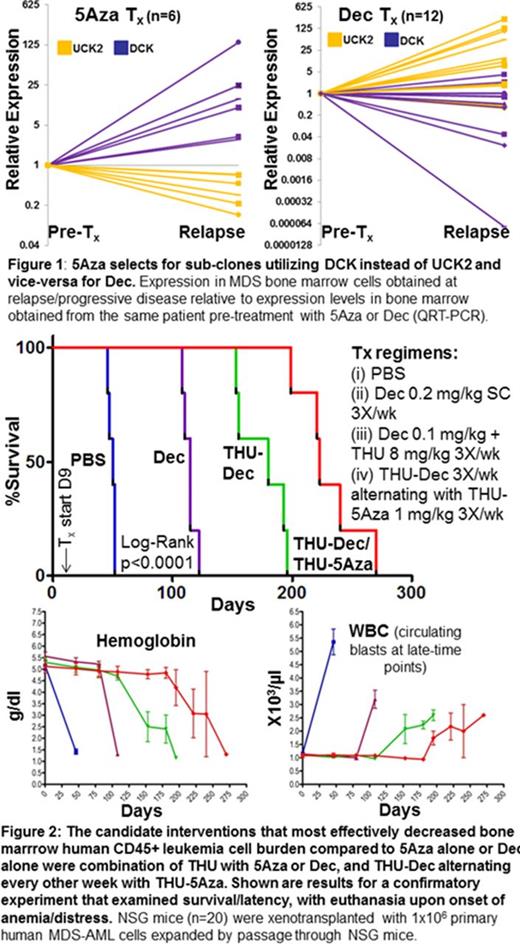
Achievement of molecular PD by 5Aza/Dec depends on intra-cellular exposure/half-life (t½). Intra-cellular t½ depends in turn on extra-cellular exposure time (plasma t½) and pyrimidine metabolism enzymes that phosphorylate 5Aza/Dec, trapping these drugs in cells. There is controversy regarding the identify of the enzymes that phosphorylate and trap 5Aza: UCK1, UCK2 or both. To answer this question, we correlated UCK1, UCK2 and DCK expression with sensitivity of NCI60 cancer cell lines (n=60) to 5Aza or Dec. 5Aza sensitivity correlated strongly with UCK2 expression but not UCK1 or DCK (corr. coeff.: 5AzaGI50 vs UCK2-0.28, p=0.03; vs UCK1 0.22, p=0.1; vs DCK -0.07, p=0.5). Dec sensitivity correlated with DCK expression but not UCK2 or UCK1 (corr. coeff.: DecGI50 versus DCK-0.27, p=0.04; vs UCK1-0.01, p=0.9; vs UCK2-0.19, p=0.2). There was greater than 2-fold variation of UCK2/DCK expression in primary MDS CD34+ bone marrow cells, not linked with WHO-subtypes or genetics, suggesting flexible use of one or the other enzyme for pyrimidine salvage, although a trend for higher DCK expression was noted in MDS containing mutated TP53 (GSE58831). Does treatment select for sub-clones utilizing salvage enzymes that avoid drug? DCK and UCK2 expression was serially measured by QRT-PCR in bone marrow from MDS patients treated with a Dec regimen rationalized for non-cytotoxic DNMT1-depletion (0.1-0.2 mg/kg SC 1-3X/week, n=12) or with conventional 5Aza (n=6). UCK2 consistently increased (up to 100-fold) and DCK consistently decreased at relapse on Dec, with vice-versa for 5Aza (Fig. 1).
Mechanism-based solutions to such resistance were evaluated in xenotransplant models of human MDS-AML that phenocopy human disease with bone marrow infiltration and death by cytopenia. Candidate solutions investigated were (i) Dec + thymidine, or HU, or methotrexate, to increase DCK (by inhibition of ribonucleotide reductase [RNR]); (ii) 5Aza or Dec at lower dose to decrease Cmax and avoid cytotoxicity + tetrahydrouridine (THU) to increase plasma t½/Tmax for DNMT1-depletion (THU inhibits the enzyme cytidine deaminase that rapidly inactivates 5Aza and Dec in vivo); (iii) 5Aza alternating or combined with Dec, since 5Aza-mediated increases in DCK are expected to increase sensitivity to Dec, while Dec-mediated increases in UCK2 are expected to increase sensitivity to 5Aza; Of these interventions, reduced dosage 5Aza or Dec + THU, and alternating THU-5Aza with THU-Dec, but not simultaneous THU-5Aza+THU-Dec, nor the RNR inhibitors, increased response and survival to striking extents (approaching 1 year)(Fig. 2). Bone marrow gH2AX and DNMT1-expression by flow-cytometry and preservation of murine hematopoiesis confirmed the non-cytotoxic, epigenetic mechanism of action (Fig. 2).
We show that straightforward shifts in pyrimidine metabolism demonstrably account for much of the resistance to 5Aza or Dec. Fortunately, such resistance is amenable to logical, non-toxic, clinically applicable solutions, validated in pre-clinical in vivo models. Clinical trials of non-cytotoxic THU-Dec (IND#112914) and alternating THU-Dec/THU-5Aza, incorporating companion PD/metabolism biomarkers, are planned.
Sekeres:Celgene Corporation: Membership on an entity's Board of Directors or advisory committees. Saunthararajah:Not applicable: Other: patent applications around decitabine, 5-azacytidine and tetrahydrouridine. Off Label Use: decitabine used by route of administration and dosages other than FDA approved regimens.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal