Abstract
The introduction of JAK inhibitors into clinical practice has produced significant clinical benefits for patients with MPNs, including reduction of splenomegaly and improvement in symptom burden. However, JAK inhibitors have demonstrated limited clinical ability to alter the natural history and biology of disease (such as reversal of fibrosis) in MPNs, and have produced only modest reductions in JAK2 V617F allele burden. This has prompted the evaluation of other therapeutic strategies in MPN patients.
Heat Shock Protein 90 (Hsp90) inhibitors have recently emerged as promising potential treatment for MPN patients. Hsp90 physically interacts with JAK2, and inhibition of Hsp90 induces degradation of JAK2. Hsp90 is also a client chaperone for a variety of other proteins involved in pathways known to be critical for cancer cell differentiation, proliferation and survival. Preclinical studies have demonstrated that Hsp90 inhibitor therapy results in dose-dependent degradation of JAK2 and inhibition of downstream signaling pathways including STAT5, STAT3 and MAPK, as well as inhibition of growth of cells expressing mutant JAK2. In vivo treatment with Hsp90 inhibitors in MPN retroviral murine models led to normalized of peripheral blood counts, reduction in organomegaly, and improvement in overall survival.
Based on these promising preclinical data, we conducted an open label phase II trial to assess the efficacy and confirm the safety of a novel Hsp90 inhibitor, AUY922 (Novartis) in patients with primary myelofibrosis (PMF), post-polycythemia vera and post-essential thrombocythemia myelofibrosis (IPSS-2 or higher) and in patients with polycythemia vera or essential thrombocythemia, who were refractory to, intolerant of, or ineligible for conventional therapy. The primary objective of the study was to determine the efficacy of AUY922 in this patient population. Secondary objectives included confirmation of safety and tolerability along with exploration of how treatment modified the biology of the disease.
From 2012-2014, 6 patients were treated, 4 female, median age 55 (53-72), with MPNs; 4 with primary-MF, 1 with PV and 1 with ET, 4 patients were JAK2 V617F positive. Prior treatments included hydroxyurea in 5, anagrelide in 3, interferon in 1, azacitadine and pamidronate in 1. Median length of treatment was 39.5 months (1-145). 3 patients with MF experienced stable disease (including two patients with blast-phase disease), and one experienced an anemia response as measured by the European LeukemiaNet (ELN) response criteria. The patient with ET experienced stable disease as measured by the Revised International Working Group for Myeloproliferative Neoplasms Research and Treatment (IWG-MRT).
Notable (greater than grade 2) adverse events included diarrhea in 5 patients, night blindness in 4, alkaline phosphatase elevation in 2, nausea in 2, emesis in 2, blurred vision in 1 and CPK elevation in 1 patient Serious Adverse Events (SAEs) included nausea, vomiting, diarrhea and altered mental status. As well, three patients experienced gastrointestinal bleeding as an SAE (grade 2 and 3). Two of these events were associated with an ileocecal ulcer. Two events were deemed to be possibly related to exposure to the study drug. The trial was terminated for this reason.
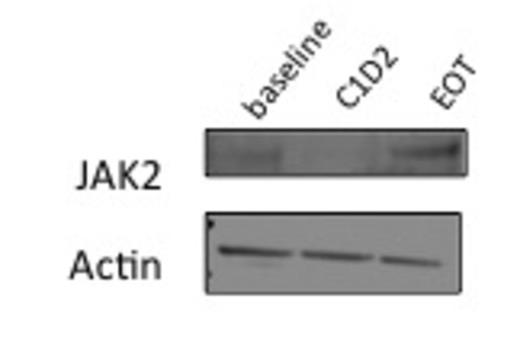
Evaluation of the impact on JAK-STAT signaling is underway with evidence of reduction in total JAK2 protein following infusion (figure 1). Further evaluation of JAK-STAT signaling, impact on cytokine production, and JAK2 allele burden is under evaluation and results will be presented.
In summary, treatment with AUY922 in patients with has demonstrated response including stable disease (including in blast-phase myelofibrosis patients) as well as anemia response and clinical improvement in one myelofibrosis patient. These data indicate that Hsp90 inhibition has clinical activity in MPN patients, and that further clinical therapeutic efforts targeting Hsp90 warrant investigation in this patient population.
Responses to treatment with AUY922 per Revised International Working Group for Myeloproliferative Neoplasms Research and Treatment (IWG-MRT) and European LeukemiaNet criteria (ELN).
Responses to treatment with AUY922 per Revised International Working Group for Myeloproliferative Neoplasms Research and Treatment (IWG-MRT) and European LeukemiaNet criteria (ELN).
Western Blot analysis of total JAK2 from peripheral blood of patient at baseline, cycle 1 day 2, and end of treatment.
Western Blot analysis of total JAK2 from peripheral blood of patient at baseline, cycle 1 day 2, and end of treatment.
Mauro:Pfizer: Consultancy; Novartis Pharmaceutical Corporation: Consultancy, Research Funding; Bristol-Myers Squibb: Consultancy; Ariad: Consultancy. Levine:Foundation Medicine: Consultancy; CTI BioPharma: Membership on an entity's Board of Directors or advisory committees; Loxo Oncology: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal