Key Points
Pom-Dex is active and well tolerated in adverse cytogenetic patients with early RRMM, particularly in those with del(17p).
Pom-Dex prolonged OS in adverse cytogenetic patients with early RRMM.
Abstract
The combination of pomalidomide and low-dose dexamethasone (Pom-Dex) can be safely administered to patients with end-stage relapsed/refractory multiple myeloma (RRMM). However, we observed a shorter median progression-free survival (PFS) and overall survival (OS) in these patients when characterized with adverse cytogenetics (deletion 17p and translocation [4;14]) in the Intergroupe Francophone Myélome (IFM) 2009-02 trial. We then sought to determine whether MM with adverse cytogenetics would benefit more from Pom-Dex if exposed earlier in the multicenter IFM 2010-02 trial. The intention-to-treat population included 50 patients, with a median age of 63 years (38% were ≥65 years). Interestingly, there was a striking difference in time to progression (TTP), duration of response, and overall response rate (ORR) according to the presence of del(17p) compared with t(4;14) (TTP, 7.3 vs 2.8 months; duration of response, 8.3 vs 2.4 months; and ORR, 32% vs 15%). OS was prolonged after Pom-Dex, particularly in t(4;14), given the short TTP, suggesting that patients were rescued at relapse with further lines of therapy. Pom-Dex, a doublet immunomodulatory drug–based regimen, is active and well tolerated in adverse cytogenetic patients with early RRMM, particularly in those with del(17p), who are characterized by a high and rapid development of a refractoriness state and known for their poor prognosis. Future studies will determine the underlying mechanisms of Pom-Dex activity in del(17p). This trial is registered at www.clinicaltrials.gov as #NCT01745640.
Introduction
Multiple myeloma (MM) is heterogeneous, with divergent outcomes driven by the biological characteristics of the disease, especially the cytogenetic features.1,2 Various studies have demonstrated that the cytogenetic characteristics as detected by fluorescent in situ hybridization (FISH) are one of the most powerful prognostic markers in MM, especially when identifying the presence of deletion of 17p13 (del[17p])2,3 and/or translocation (4;14) (t[4;14]),2,3 which represents ∼20% to 25% of patients in the series.4 MM with adverse FISH cytogenetics, del(17p) and/or t(4;14), is characterized by short survival related to an early relapse rate (median time to progression [TTP] <4 months) and rapid development of mechanisms of resistance to multiple agents.2,5-7 This shorter survival was observed in patients treated with lenalidomide and/or bortezomib-based regimens, which are considered the 2 most active MM therapeutics.8-11 There is a clear unmet medical need for patients with adverse FISH cytogenetics, as the pace of relapse is rapid, even in the early relapse (relapsed/refractory multiple myeloma [RRMM]) setting (eg, 1 to 3 lines of therapy).
Several studies have reported that pomalidomide and low-dose dexamethasone (Pom-Dex) produce a 30% to 40% response rate (overall response rate [ORR], partial response and greater) with prolonged duration of response (DOR) and median TTP in RRMM patients who have progressed after multiple treatments.12-15 However, the median TTP and progression-free survival (PFS) were much shorter (<4 months in most studies) for patients with del(17p) and/or t(4;14).12,13,16 These studies were conducted in advanced end-stage MM patients who had been previously exposed to a median of 5 or 6 lines of therapy, and therefore it is difficult to determine whether this lack of activity was related to the end-stage feature or the adverse genomic profile.
We sought to demonstrate that Pom-Dex was active in patients with adverse FISH cytogenetic profiles if used earlier in the relapse setting. We therefore designed a phase 2 IFM multicenter, open-label study aimed to determine the efficacy and safety profile of Pom-Dex in early RRMM patients with del(17p) and/or t(4;14).
Materials and methods
Eligibility
Patients were eligible to enter into the study if they had RRMM following at least 1 prior regimen of myeloma treatment.17,18 The patients were to be exposed to lenalidomide (minimum 2 cycles). Patients were required to have measurable disease using either intact immunoglobulin and light-chain immunoglobulin or a serum immunoglobulin free light chain of >100 mg/L and an abnormal free light chain ratio.17,18 Patients were also required to have a platelet count ≥75 × 109/L, neutrophil count ≥1.0 × 109/L, and creatinine clearance ≥50 mL/min. All previous myeloma therapy must have been discontinued ≥2 weeks before the start of treatment. The FISH cytogenetic analysis of del(17p) (positive if ≥60%) and/or t(4;14) was performed centrally by H.A.-L. on selected bone marrow plasma cells.2,10 Laboratory analyses to determine MM response were performed centrally at the Centre Hospitalier Régional Universitaire (CHRU) of Lille.
The study was approved by the Intergroupe Francophone du Myélome (IFM) and the CHRU of Lille review board in accordance with national regulations in France, and it was conducted according to the Declaration of Helsinki. This trial is registered at www.clinicaltrials.gov (#NCT01745640).
Treatment schedule
Pomalidomide 4 mg was given orally daily on days 1 to 21 of each 28-day cycle along with dexamethasone 40 mg, which was given orally to all patients on days 1, 8, 15, and 22 of each cycle. The treatment was given until progression. It was mandatory to provide a thromboprophylaxis to patients in the study. Granulocyte colony-stimulating factor was allowed. Dose adjustments were permitted based on grade 3 or 4 adverse events (AEs) or based on an investigator’s decision and involved lowering the dose of pomalidomide to 3 mg, 2 mg, or 1 mg per day and the dose of dexamethasone to 20 mg once weekly. Patients unable to tolerate the lowest doses of pomalidomide or dexamethasone had to stop therapy with that agent permanently.
Objectives of the study
The primary objective was to determine TTP. Secondary objectives were to determine safety, ORR (partial response and better), very good partial response (VGPR) + complete response rate, clinical benefit rate (minor response and better), time to response and response duration, overall survival (OS), event-free survival, response, and TTP with respect to cytogenetic abnormalities in bone marrow tumor plasma cells.
Response and toxicity criteria
Responses and progression were assessed according to International Myeloma Working Group (IMWG) criteria17,18 and required 2 consecutive assessments made at any time. The National Cancer Institute Common Terminology Criteria for Adverse Events (version 4.0) was used to grade AEs and assign perceived attribution of these events to the study treatment regimen.
Protocol outlines
An interim analysis was performed when 15 patients were enrolled and had received at least 1 cycle. The analysis was performed and reviewed by a data management committee. The data management committee confirmed the treatment was safe for the studied population and allowed the study to fully recruit.
Statistical design and analysis
The median TTP published in relapsed or refractory MM patients with deletion 17p and t(4;14) is 4 months2,3,10 For the primary efficacy variable, TTP, a 100% improvement in the median, from 4 months to 8 months, for the Pom-Dex regimen is considered clinically relevant. The total number of patients is equal to 47. With the expectation that 15% of patients could be unevaluable, 55 patients (47/0.85) were to be included. The primary analysis was conducted on the intention-to-treat (ITT) population. A data cutoff was made on September 1, 2013. Statistical analysis was performed using SAS software.
Results
Patient population
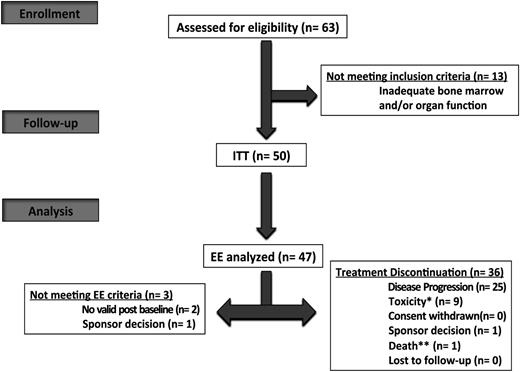
A total of 63 patients (male-to-female ratio, 1.5; median age, 63 years [range, 31-83 years]; 19 patients (38%) were >65 years) were enrolled from 27 centers between January 2012 and July 2013 (Figure 1). Thirteen patients were screening failures. The ITT population included 50 patients. All patients had loss of 17p (46%) and/or t(4;14) (64%); of note, 2 patients displayed both 17p and t(4;14). The efficacy evaluable population comprised 47 patients. Patient demographics and baseline characteristics for the ITT population are described in Table 1. Overall, the median age was 59 (range, 30-80) years. The median time from diagnosis to enrollment was 3 years (interquartile range, 2-4). All patients had prior exposure to lenalidomide with 84% being refractory, and 96% had received a proteasome inhibitor with 54% being refractory; details on previous lines of therapy are summarized in Table 2.
IFM 2010-02 flow diagram. *Toxicity related to study treatment. **Death not related to toxicity and progression. EE, efficacy evaluable.
IFM 2010-02 flow diagram. *Toxicity related to study treatment. **Death not related to toxicity and progression. EE, efficacy evaluable.
Patient demographics and baseline characteristics at entry into IFM 2010-02
| Characteristic . | n = 50 . |
|---|---|
| ISS stage at diagnosis, n (%) | |
| II/III | 20 (59) |
| Lytic bone lesions, n (%) | |
| Yes | 34 (87) |
| Number of lytic lesions: 3-6 | 3 (7.7) |
| Number of lytic lesions: >6 | 16 (41) |
| Osseous fracture | 8 (20.5) |
| Medullary compression | 1 (2.6) |
| Plasmacytoma | 6 (12) |
| Median β2-microglobulin, mg/L (range) | 3.2 (1-13) |
| 3.5-5.5 mg/L, n (%) | 16 (44) |
| >5.5 mg/L, n (%) | 19 (53) |
| Median albumin, g/L (range) | 39 (25-48) |
| Median serum creatinine, µmol/L (range) | 76.5 (53-138) |
| >115 µmol/L, n (%) | 1 (2) |
| Median hemoglobin, g/dL (range) | 11.1 (8.6-15) |
| <10 g/dL, n (%) | 14 (28) |
| Median neutrophils, ×109/L (range) | 2.9 (.6-13) |
| <1000 g/L, n (%) | 2 (4) |
| Median platelets, ×109/L (range) | 180 (65-458) |
| <100 g/L, n (%) | 6 (12) |
| Circulating plasma cells (yes, %) | 3 (6) |
| Adverse FISH cytogenetics | |
| At study entry, n (%) | 50/50 |
| del(17p), n (%) | 22 (46)* |
| t(4;14), n (%) | 32 (64)* |
| At diagnosis, n (%) | 45/50 |
| del(17p), n (%) | 14 (39) |
| t(4;14), n (%) | 26 (70) |
| Characteristic . | n = 50 . |
|---|---|
| ISS stage at diagnosis, n (%) | |
| II/III | 20 (59) |
| Lytic bone lesions, n (%) | |
| Yes | 34 (87) |
| Number of lytic lesions: 3-6 | 3 (7.7) |
| Number of lytic lesions: >6 | 16 (41) |
| Osseous fracture | 8 (20.5) |
| Medullary compression | 1 (2.6) |
| Plasmacytoma | 6 (12) |
| Median β2-microglobulin, mg/L (range) | 3.2 (1-13) |
| 3.5-5.5 mg/L, n (%) | 16 (44) |
| >5.5 mg/L, n (%) | 19 (53) |
| Median albumin, g/L (range) | 39 (25-48) |
| Median serum creatinine, µmol/L (range) | 76.5 (53-138) |
| >115 µmol/L, n (%) | 1 (2) |
| Median hemoglobin, g/dL (range) | 11.1 (8.6-15) |
| <10 g/dL, n (%) | 14 (28) |
| Median neutrophils, ×109/L (range) | 2.9 (.6-13) |
| <1000 g/L, n (%) | 2 (4) |
| Median platelets, ×109/L (range) | 180 (65-458) |
| <100 g/L, n (%) | 6 (12) |
| Circulating plasma cells (yes, %) | 3 (6) |
| Adverse FISH cytogenetics | |
| At study entry, n (%) | 50/50 |
| del(17p), n (%) | 22 (46)* |
| t(4;14), n (%) | 32 (64)* |
| At diagnosis, n (%) | 45/50 |
| del(17p), n (%) | 14 (39) |
| t(4;14), n (%) | 26 (70) |
High-risk cytogenetics by FISH consisted of deletion 17p or t(4;14) at diagnosis and/or at entry in IFM 2010-02 trial. Patients considered not measurable based on serum intact Ig and urine light-chain excretion, were measurable if they had serum immunoglobulin free light chain (FLC) more than 100 mg/L and an abnormal FLC ratio.
Two patients were both del(17p) and t(4;14).
Details on previous lines of therapy
| Therapy details . | n = 50 . |
|---|---|
| Time from diagnosis to randomization, y | |
| Median (95% CI) | 3 (2-4) |
| ≤3 y, n (%) | 23 (50) |
| 3-6 years, n (%) | 18 (39) |
| Number of lines, median (range) | 3 (1-10) |
| 2, n (%) | 16 (32) |
| 3, n (%) | 19 (38) |
| ≥3, n (%) | 11 (22) |
| Prior lines of therapy, exposed/refractory, (%) | |
| Lenalidomide | 100/86 |
| Bortezomib | 96/56 |
| Alkylator | 100/90 |
| Anthracyclines | 26/50 |
| Last prior therapy | 76* |
| Autologous transplantation | 76† |
| Allogeneic transplantation | 2† |
| Therapy details . | n = 50 . |
|---|---|
| Time from diagnosis to randomization, y | |
| Median (95% CI) | 3 (2-4) |
| ≤3 y, n (%) | 23 (50) |
| 3-6 years, n (%) | 18 (39) |
| Number of lines, median (range) | 3 (1-10) |
| 2, n (%) | 16 (32) |
| 3, n (%) | 19 (38) |
| ≥3, n (%) | 11 (22) |
| Prior lines of therapy, exposed/refractory, (%) | |
| Lenalidomide | 100/86 |
| Bortezomib | 96/56 |
| Alkylator | 100/90 |
| Anthracyclines | 26/50 |
| Last prior therapy | 76* |
| Autologous transplantation | 76† |
| Allogeneic transplantation | 2† |
Last prior therapy. Only refractory percentage is provided.
Percentages provided are exposed patients.
Duration of treatment
The median follow-up was 10 months. At the data cutoff, a total of 36 patients (72%) had discontinued treatment (Figure 1). The most common reason for treatment discontinuation was disease progression (n = 25, 69%). The duration of treatment, dose reduction, and dose interruption data are summarized in supplemental Table 1, available on the Blood Web site (12% and 64% of patients had pomalidomide dose reduction and interruption). Overall, the median number of cycles administered was 4 (range, 1-17), with 26% (n = 13) and 8% (n = 4) with >6 and 12 cycles, respectively. Pomalidomide therapy was manageable in this population characterized with poor prognosis related to intrinsic plasma cells features, because the median relative dose intensity was 87.8% for pomalidomide (95% confidence interval [CI]: 73, 100) and 87.5% for dexamethasone (95% CI: 75, 100).
Efficacy
The ORR was 22% (n = 11) for the ITT population, including 6% (n = 3) >VGPR, and 60% (n = 30) had stable disease (including minor response, n = 6 [13%]). Details of response rates are presented in Table 3 along with the median time to first response and the median duration of response. In the efficacy evaluable population (n = 47), 23% of patients responded, and 62% (n = 29) had stable disease. The ORR according to presence of del(17p) and/or t(4;14) is summarized in Table 3 and showed a striking difference with ORR in 32% vs 15% in del(17p) and t(4;14), respectively. The median duration of response was 5.5 months (95% CI: 0.9, —) overall, with 44% of patients maintaining response beyond 8 months. Similarly to ORR, there was a striking difference in duration of response between the del(17p) and t(4;14), with a median of 8.3 months (1.8, —), with 67% still responding at 8 months vs 2.4 months (1;8), with only 25% at 8 months.
Summary of response to treatment in the total study population and per group (n = 50)
| . | Total . | del(17p) . | t(4;14) . |
|---|---|---|---|
| Response rate, ITT (n = 50), n (%) | |||
| ORR (≥PR) | 11 (22) | 7 (32) | 5 (15) |
| ≥VGPR | 3 (6) | 2 (9) | 1 (3) |
| PR | 8 (16) | 5 (23) | 4 (12) |
| Stable disease | 30 (60) | 9 (41) | 22 (69) |
| CBR (≥MR) | 17 (34) | 7 (32) | 11 (34) |
| Progressive disease | 7 (14) | 4 (18) | 5 (15) |
| Not evaluable | 2 (4) | 2 (9) | 0 |
| Time to first response, mo | |||
| Median (95% CI) | 4.1 (4;8) | — | — |
| Range | 3-28 | — | — |
| Duration of response, mo | |||
| Median (95% CI) | 5.5 (0.95;–) | 8.3 (1.9;–) | 2.4 (0.95;8.3) |
| One-year free, % | 44 | 67 | 25 |
| Response rate, EE (n = 47), n (%) | |||
| ORR | 11 (23) | — | — |
| CBR | 17 (36) | — | — |
| . | Total . | del(17p) . | t(4;14) . |
|---|---|---|---|
| Response rate, ITT (n = 50), n (%) | |||
| ORR (≥PR) | 11 (22) | 7 (32) | 5 (15) |
| ≥VGPR | 3 (6) | 2 (9) | 1 (3) |
| PR | 8 (16) | 5 (23) | 4 (12) |
| Stable disease | 30 (60) | 9 (41) | 22 (69) |
| CBR (≥MR) | 17 (34) | 7 (32) | 11 (34) |
| Progressive disease | 7 (14) | 4 (18) | 5 (15) |
| Not evaluable | 2 (4) | 2 (9) | 0 |
| Time to first response, mo | |||
| Median (95% CI) | 4.1 (4;8) | — | — |
| Range | 3-28 | — | — |
| Duration of response, mo | |||
| Median (95% CI) | 5.5 (0.95;–) | 8.3 (1.9;–) | 2.4 (0.95;8.3) |
| One-year free, % | 44 | 67 | 25 |
| Response rate, EE (n = 47), n (%) | |||
| ORR | 11 (23) | — | — |
| CBR | 17 (36) | — | — |
The median time to first response and response duration are also provided.
CBR, clinical benefit rate; EE, efficacy evaluable; MR, minor response; PR, partial response.
Interestingly, patients who reached a response, particularly a VGPR, independently of the adverse FISH cytogenetic subgroup had an improved TTP outcome in this study (supplemental Table 2). As such, it might be a good surrogate marker for improved long-term outcome in patients with adverse FISH cytogenetics.
TTP
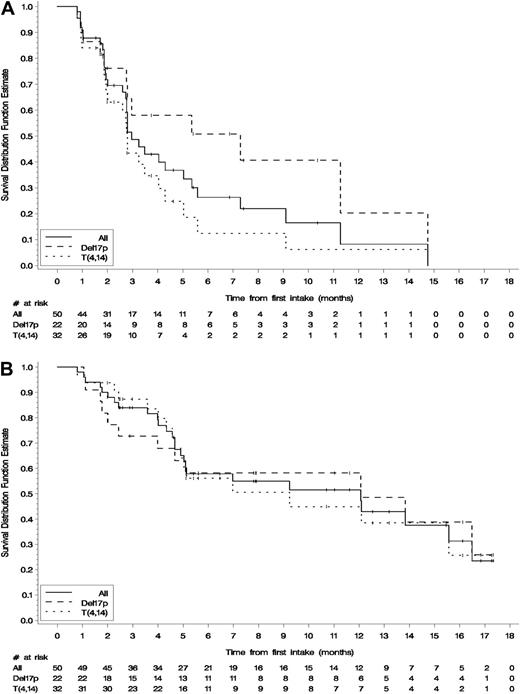
At the cutoff of September 1, 2013, 33 patients (66%) had progressed. The primary end point was median TTP, with the objective to improve the median TTP from 4 months to 8 months. The median TTP of the studied cohort as a whole was 2.96 (95% CI: 2.7, 5.0) months, with 22% of patients free of relapse at 8 months (Figure 2A). We then sought to study TTP per cytogenetic feature, and we found a marked difference between del(17p) and t(4;14). The median TTP was 7.3 (2.7, 14.7) months, with 41% at 8 months vs 2.8 (1.9;4.0) and 12.4% at 8 months in del(17p) and t(4;14), respectively (Figure 2A). The median PFS was very similar to TTP (2.8 [2, 4] months with 19.3% at 8 months).
Survival end points for the cohort as a whole and in either subgroup, deletion 17p or t(4;14). Kaplan-Meier estimates (ITT, n = 50). (A). TTP. (B). OS.
Survival end points for the cohort as a whole and in either subgroup, deletion 17p or t(4;14). Kaplan-Meier estimates (ITT, n = 50). (A). TTP. (B). OS.
Variables to explain shorter TTP
As we observed a prolonged TTP for some patients across the 2 subgroups, del(17p) and t(4;14), we sought to identify the characteristics that could explain, and in the future predict, differential sensitivity to the combination of Pom-Dex in this specific population. Variables that significantly explained the shorter TTP on univariate analysis, independently of the adverse cytogenetic profile, are summarized in supplemental Table 2. The cytogenetic profile, either del(17p) or t(4;14), did not explain the shorter TTP. We then performed a multivariate analysis entering all variables based on selected P value ≤ .15 on univariate analysis. We found that ISS3 (hazard ratio [HR] = 4.24; 95% CI: 1.33, 13.50; P = .014), time between initial diagnosis and study entry ≤2 years (HR = 5.00; 95% CI: 1.59, 15.72; P = .005), and refractory to the last prior line (HR = 4.77; 95% CI: 1.38, 16.47; P = .013) had a significantly shorter TTP.
Survival
A total of 26 patients (52%) had died during the study at the cutoff date. The median OS for the cohort as a whole is 12 months (95% CI: 5, 15), with an 8-month event-free survival rate of 55% (Figure 2B). Interestingly, del(17p) patients benefited more from Pom-Dex compared with t(4;14) patients, similar to TTP and PFS, but to a lesser extent (Figure 2B). The median OS was 12 months (2, —) with 58% at 8 months vs 9.2 months (5, —) and 50.5% at 8 months. These data suggest that most patients in the study, including those with t(4;14), were able to receive the appropriate rescue therapy after progression on the Pom-Dex regimen.
Toxicity profile of the pomalidomide-dexamethasone combination
The toxicity profile of the Pom-Dex combination consisted primarily of myelosuppression, as previously reported, and appeared manageable in these fragile RRMM patients. A total of 49 patients (98%) experienced an AE, of which 44 (88%) were treatment related. The incidence rate of grade 3 and 4 AEs was 45 (90%), including hematologic AEs, and 32 (64%) experienced a serious adverse event (SAE). Ten patients (20%) died of AEs (mostly septic syndrome), all of them in the context of progression of myeloma so more likely to be related to myeloma rather than the Pom-Dex regimen. Similarly, 12 patients (24%) discontinued because of the occurrence of SAEs, but 1 patient discontinued due to a rash. No occurrence or worsening of neuropathy was reported; no occurrence of second primary malignancy and only 1 pulmonary embolism was noted knowing that use of a thromboprophylatic treatment was mandatory in this study. The most common hematologic and nonhematologic (all grades) study drug-related AEs are summarized in Tables 4 and 5.
Summary of study-drug–related AEs (all grades, NCI CTC) that occurred in more than 5% of cases, according to system organ class (n = 50)
| Study-drug–related AEs . | n (%) . |
|---|---|
| Blood and lymphatic system disorders | 36 (72) |
| Anemia | 15 (30) |
| Neutropenia | 31 (62) |
| Febrile neutropenia | 4 (8) |
| Thrombocytopenia | 82 (16) |
| General disorders and administration-site conditions | 21 (42) |
| Asthenia | 7 (14) |
| Musculoskeletal and connective tissue disorders | 6 (12) |
| Cramps | 6 (12) |
| Gastrointestinal disease | 12 (24) |
| Constipation | 4 (8) |
| Diarrhea | 3 (6) |
| Nausea | 3 (6) |
| Infections | 16 (32) |
| Pneumonia/bronchitis | 9 (18) |
| Study-drug–related AEs . | n (%) . |
|---|---|
| Blood and lymphatic system disorders | 36 (72) |
| Anemia | 15 (30) |
| Neutropenia | 31 (62) |
| Febrile neutropenia | 4 (8) |
| Thrombocytopenia | 82 (16) |
| General disorders and administration-site conditions | 21 (42) |
| Asthenia | 7 (14) |
| Musculoskeletal and connective tissue disorders | 6 (12) |
| Cramps | 6 (12) |
| Gastrointestinal disease | 12 (24) |
| Constipation | 4 (8) |
| Diarrhea | 3 (6) |
| Nausea | 3 (6) |
| Infections | 16 (32) |
| Pneumonia/bronchitis | 9 (18) |
AEs leading to discontinuation of pomalidomide or both treatments, according to system organ class (n = 50)
| AEs leading to discontinuation of pomalidomide or both treatments . | n (%) . |
|---|---|
| Blood and lymphatic system disorders | |
| Anemia | 1 (2) |
| Neutropenia | 1 (2) |
| Febrile neutropenia | 1 (2) |
| Thrombocytopenia | 1 (2) |
| Musculoskeletal and connective tissue disorders | 2 (4) |
| Bone pain | 2 (4) |
| Pulmonary embolism | 1 (2) |
| Infections | 4 (8) |
| Septic shock | 3 (6) |
| AEs leading to discontinuation of pomalidomide or both treatments . | n (%) . |
|---|---|
| Blood and lymphatic system disorders | |
| Anemia | 1 (2) |
| Neutropenia | 1 (2) |
| Febrile neutropenia | 1 (2) |
| Thrombocytopenia | 1 (2) |
| Musculoskeletal and connective tissue disorders | 2 (4) |
| Bone pain | 2 (4) |
| Pulmonary embolism | 1 (2) |
| Infections | 4 (8) |
| Septic shock | 3 (6) |
Discussion
This phase 2 study further demonstrates that Pom-Dex is a safe and effective combination for the treatment of RRMM, including in patients with high-risk features and adverse FISH such as del(17p). On the contrary, it seems that patients with t(4;14) will require a significant improvement in the Pom-Dex regimen if used as a backbone in future studies.
The differential activity observed in RRMM with del(17p) compared with t(4;14) exposed to the combination of Pom-Dex remains unexplained to date. This discovery was also reported in a subanalysis of the MM003/Nimbus phase 3 multicenter trial for registration in Europe of the combination of Pom-Dex.16 The authors also noted that the median PFS was significantly longer for patients with del(17p) vs t(4;14) (4.6 vs 2.8 months; P = .011), as was the median OS (12.6 vs 7.5 months, respectively; P = nonsignificant). Interestingly, median OS was similar for patients with del(17p) and standard-risk cytogenetics (12.6 vs 14.0 months).16 These results and ours suggest that the Pom-Dex regimen may partially overcome the poor prognosis of del(17p) in RRMM, even in very advanced and heavily pretreated patients. However, it also clearly showed that t(4;14) patients do not benefit from the doublet Pom-Dex–based regimen and will require further developments, such as studying the activity of triplet Pom-Dex–based regimens, before Pom-Dex can be considered an interesting backbone to build upon in t(4;14).
We reported herein data showing that Pom-Dex given earlier (median 3 lines of therapy instead of 5 to 6 in advanced end-stage studies) in the disease course of relapsed myeloma benefited patients with del(17p) compared with the 4-month median PFS reported in the literature with bortezomib and/or lenalidomide-based regimens in this MM population characterized with rapid relapse rate and development of mechanisms of resistance to all existing agents.2,3,8,10 Similarly, these data also compared favorably to the 4- to 5-month median PFS reported with use of Pom-Dex in end-stage myeloma very advanced in the disease course with a median of 5 to 6 lines of therapy.12-15 The improved survival observed in patients with del(17p) is also impressive in the context of all patients relapsing after lenalidomide. The protocol required inclusion of patients exposed to lenalidomide that is given following European Medicines Agency approval at first relapse until progression in France, meaning most of the patients relapsed with MM refractory to lenalidomide. Questions remain on whether this improved TTP for del(17p) was related to the earlier exposure to the combination of Pom-Dex or to a specific mechanism of action of pomalidomide on MM with del(17p).19,20
An important concern is the role of adverse cytogenetics in RRMM, particularly in advanced end-stage RRMM, where very limited data are available. In patients who failed to improve with bortezomib and IMiDs, the IMWG found that t(4;14) at diagnosis contributed to shorter OS, with del(17p) contributing to a lesser extent; however, data regarding cytogenetic status and FISH results from the IMWG study are very limited.21 In our study, adverse cytogenetics were determined by FISH at diagnosis and study entry for all patients by H.-A-L. (centrally in Toulouse), whose expertise is known internationally, and furthermore, we applied the cutoff at 60% of positive cells to consider the presence of del(17p). Interestingly, 96% of patients had presence of adverse cytogenetics (del[17p] and/or t[4;14]) at both diagnosis and study entry in our study.
The initial hypothesis of the study was to improve TTP of del(17p) and t(4;14) by 100%, going from the 4-month median TTP reported in the literature to 8 months. In that regard, the study did not reach its statistical end point, because the median TTP for the best of the 2 subgroups, del(17p), failed to reach a TTP of 8 months by less than 1 month. However, given the discovery of the intrinsic heterogeneity among and across adverse cytogenetic subgroups, it seems that future studies will have to consider an adverse cytogenetic subgroup-based hypothesis rather than a simple hypothesis that fits all groups.
In our study, we noticed that some patients, although characterized with adverse cytogenetic features, could have a prolonged TTP in both the del(17p) and t(4;14) groups, although it was more frequent in del(17p). We then sought to better understand the characteristics of patients characterized with a shorter TTP and found that patients with ISS3, with a very short time between initial diagnosis and study entry (≤2 years) and refractory to the last prior line displayed a greater reduction in TTP. We also identified several other poor features characterizing MM with high genomic instability, clonal evolution, higher proliferation rate, and escape from the bone marrow microenvironment, although solely identified on univariate analysis. It seems understandable that current approaches with the available drugs might not be able to overcome the adverse and high-risk prognosis of RRMM in these patients.
The difference among and across adverse cytogenetic subgroups may also be related to underlying biological differences with a key impact on the tumor cell behavior, aggressiveness, resistance to treatments and potentially impact survival. IFM has launched a separate study on a larger cohort of patients with either adverse cytogenetic profile, del(17p) or t(4;14), in order to better understand the various biological mechanisms underlying these subgroups and potentially identify some that would clearly explain the survival differences among and across these subgroups. There are currently several ongoing hypothesis. (1) One hypothesis is that the differences might essentially be related to the underlying genetic features, other chromosomal changes that accompany the main genetic alteration and that would positively or negatively impact the outcome, such as trisomies or deletions.22 IFM reported at the 2014 annual meeting of the American Society of Hematology that either del(17p) or t(4;14), del(1p32), and genomic complexity were the most important prognosticators for PFS and OS for these 2 adverse cytogenetic profiles. (2) Other underlying biological mechanisms were also proposed, such as an impact of the type of loss of materials in certain cytogenetic abnormalities (eg, t[4;14]). Certain patients show a loss of one of the telomeric parts, on either chromosome 4 or 14, and we do not yet understand how this affects the underlying biological tumor compartment. It is suspected that part of the prognostic differences among t(4;14) could be explained by such a biological mechanism.22 (3) An impact of the fusion protein based on the given translocation breakpoint (eg, for t[4;14]) is also suspected.23 H.A.-L. has investigated fusion genes involving the immunoglobulin heavy chain to understand their biology and explore a possible effect on survival in myeloma. This study suggested that immunoglobulin heavy-chain–related translocations in myeloma may impact tumor biology via a number of mechanisms, one of which is the generation of fusion proteins with functional consequences. (4) Last, but certainly not least, one mechanism might involve the degree of epigenetic changes that might affect gene expression.24 This study’s findings demonstrated that the hypomethylation of the 1q12 region can potentially amplify any genomic region distal to it and provide evidence for an epigenetic origin of the high-risk 1q21 copy-number alterations related to the progression of myeloma. Although not yet demonstrated for del(17p) or t(4;14), this mechanism and others cannot be ruled out.
Our study further confirms the manageable safety profile of the combination of Pom-Dex in RRMM, independently of which fragile population is treated, either end-stage very advanced patients12-15,25 or patients with high-risk features and adverse cytogenetic RRMM.12,13,16 In the latter group, one might keep in mind when facing septic syndrome, particularly if there is no occurrence of neutropenia, that this event is related to progression or dexamethasone rather than pomalidomide in general.
However, no definite conclusion can be drawn regarding the observed activity in del(17p), and the benefit in terms of PFS observed with Pom-Dex in our study still is limited, considering the Pom-Dex regimen was given to early-relapse patients in our study, and definitely needs improvement. Future studies will have to look into improving further the TTP of RRMM with adverse cytogenetics. Beyond the earlier use of Pom-Dex in RRMM, one would recommend to optimize the Pom-Dex-based regimen, for example with a triplet-based regimen. Many third agents could be combined with Pom-Dex, including the novel generation of proteasome inhibitors. Several publications have shown that bortezomib and carfilzomib overcame the poor prognosis of t(4;14) and/or del(17p).10,26,27 A recent study reported on carfilzomib in patients with RRMM and high-risk cytogenetic abnormalities (defined as del[13] or hypodiploidy by metaphase cytogenetic analysis and/or del[17p13], t[4;14], or t[14;16] by FISH) and suggested that the PFS of this group with adverse cytogenetic features was not significantly reduced compared with standard-risk patients28 ; interestingly, the median PFS and OS were shorter in carfilzomib-treated patients with del(17p) compared with t(4;14).28
Pomalidomide plus low-dose dexamethasone, a doublet newer-generation IMiD-based regimen, is active and well tolerated in this RRMM population, particularly in patients with high-risk features such as del(17p), characterized with high and rapid development of a refractoriness state, and known for their poor prognosis. This study also provides further evidence that IMiD compounds share no cross-resistance, including pomalidomide following lenalidomide. We believe future studies will look into optimizing the pomalidomide combination in the context of triplet pomalidomide-based regimens in adverse FISH myeloma, particularly in combination with a proteasome inhibitor, known for its synergy with IMiDs.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Celgene for financial support and for providing investigational products. They thank Dr Bernadette Hennache, Dr Suzanna Schraen, Mr Jean Luc Faucompret, and Mr Jaime Gutierrez for the central laboratory analysis in CHRU of Lille, France; Christine Cotton and Florence Carrère from Statitec (Toulouse, France) for data management and statistical analysis; and Bella Ohyba, Chanaz Louni, Mehdi Hamza, and Malek Dib, for the Direction de la Recherche Clinique of CHRU of Lille, France. They also wish to thank Pr Noël Milpied (Bordeaux, France), Pr Chantal Doyen (Louvain, Belgium), Pr Véronique Leblond (Assistance Publique–Hôpitaux de Paris, Paris, France), and Pr Gilles Chatellier (Assistance Publique–Hôpitaux de Paris, Paris, France), members of the Data and Safety Monitoring Board.
Authorship
Contribution: X.L. and T.F. conceived and designed the study; X.L., L.K., M.M., C.H., L.G., M.R., B.A., B.P., B.K., A.M.S., S.B., G.M., B.T., C.M., A.B., L. Lacotte, M.T., M.D., J.-G.F., M.O.P., P.R., M.W., B.R., L. Legros, L.B., O.D., M.E.-B., D.C., J.P.F., P.M., M.A., H.A.-L., and T.F. collected and assembled data; C.M. and M.O.P. were responsible for study and CRF conception, administrative support, and coordinated the study; H.A.-L. provided cytogenetic data; B.T. was the central pharmacist; B.O. provided biochemistry data; and X.L. analyzed and interpreted data and wrote the manuscript.
Conflict-of-interest disclosure: The following authors disclose board honorarium and lecture fees from Celgene: X.L., L.K., M.M., C.H., M.R., B.A., L.B., P.M., M.A., H.A.-L., and T.F. The remaining authors declare no competing financial interests.
Correspondence: Xavier Leleu, Service des Maladies du Sang, Hôpital Huriez, Centre Hospitalier Régional Universitaire, 59037 Lille, France; e-mail: xavier.leleu@chru-lille.fr.
References
Author notes
X.L., M.M., C.H., and L.K. contributed equally to this study.