Key Points
Monocytic IDOhi MDSCs are increased in CLL patients, suppress T cells, and promote TReg induction.
CLL cells induce conversion of monocytes into MDSCs suggesting bidirectional crosstalk between CLL cells, MDSCs, and TRegs.
Myeloid-derived suppressor cells (MDSCs) represent a heterogeneous population that shares certain characteristics including an aberrant myeloid phenotype and the ability to suppress T cells. MDSCs have been predominantly studied in malignant diseases and findings suggest involvement in tumor-associated immune suppression. Chronic lymphocytic leukemia (CLL) is the leukemia with the highest incidence among adults. Immune defects occur already at early disease stages and impact the clinical course. We assessed presence, frequency, association to other immune parameters, and functional properties of circulating CD14+ cells lacking HLA-DR expression (HLA-DRlo) in patients with untreated CLL. These monocytic cells represent one of the best-defined human MDSC subsets. Frequency of CD14+HLA-DRlo cells was significantly increased in CLL patients. Furthermore, MDSCs suppressed in vitro T-cell activation and induced suppressive regulatory T cells (TRegs). The MDSC-mediated modulation of T cells could be attributed to their increased indoleamine 2,3-dioxygenase (IDO) activity. CLL cells induced IDOhi MDSCs from healthy donor monocytes suggesting bidirectional crosstalk between CLL-cells, MDSCs, and TRegs. Overall, we identified a MDSC population that expands in CLL. The exact mechanisms responsible for such accumulation remain to be elucidated and it will be of interest to test whether antagonizing suppressive functions of CLL MDSCs could represent a mean for enhancing immune responses.
Introduction
Chronic lymphocytic leukemia (CLL) is predominantly a B-cell-derived disease and represents the leukemia with the highest incidence among adults in the Western world. Cancers are often associated with hosts’ immune defects, thereby preventing an effective tumor immune surveillance and antagonizing immune therapeutic approaches.1 Already at early disease stages, CLL patients display T-cell dysfunctions2 and have an increased susceptibility toward infections.3 Mounting evidence suggests that CLL cells actively cause T-cell deficiencies.4
Current observations suggest a reciprocal interrelationship between malignant B-cells and immune suppressive regulatory T cells (TRegs)5 that are central for physiological immune homeostasis.6 Accumulating TRegs in cancer patients are linked to tumor immune escape.7 TReg frequency is increased in CLL patients and correlates with progressive disease.8,9 A second, emerging population of suppressive cells in cancer biology are the so-called myeloid-derived suppressor cells (MDSCs).10
This heterogeneous cell population is present at very low frequencies in healthy individuals, but accumulates rapidly during inflammatory responses and cancer.11 As yet, studies in various solid malignancies (eg, breast cancer12 and melanoma13 ) and hematologic malignancies (eg, multiple myeloma14 ) have revealed an abundance of MDSCs. Because MDSCs are potent inhibitors of T-cell responses, they can limit intrinsic antitumor immune reactivity and immune therapeutic approaches, as recently emphasized by the results of a cancer vaccine trial.15 Results from clinical correlation studies demonstrated that increased MDSCs could be an independent negative prognostic marker.16
In general, human MDSCs are dichotomized into granulocytic and monocytic subsets. They exert their suppressive function through a plethora of mechanisms, including production of reactive oxygen species (ROS), immune-modulating cytokines (eg, IL-10), cyclooxygenase-2 (COX2), arginase-1 (ARG1), indoleamine-2,3-dioxygenase (IDO), and the depletion of nutrients required for adequate T-cell activation.10
Their phenotypical and functional diversity results, most likely, from the divergent composition of tumor-derived factors (eg, granulocyte macrophage–colony stimulating factor [CSF])17 or tumor necrosis factor [TNF]-α11) that govern MDSC induction, expansion, and activation, and that depend on the cancer-cell type.
Targeting MDSC accumulation in cancer models has shown efficacy in delaying disease progress,17 which is suggestive of its therapeutic exploitation. Understanding the disease-specific MDSC biology is a prerequisite for developing novel therapeutic approaches.
Here, we report IDOhiCD62LhiPD-L1hiHLA-Ghi CD11b+CD33+CD14+HLA-DRlo monocytic cells that are increased in the peripheral blood of untreated CLL patients. These MDSCs are suppressed in vitro T-cell proliferation and interferon (IFN)-γ production, whereas promoting TRegs partly mediated via IDO. In patients, MDSC levels correlated with T cells displaying a less activated phenotype and with TReg frequencies. Primary CLL cells and cell lines induced in healthy donor (HD) derived monocytes a phenotype resembling CLL MDSCs capable of inhibiting T-cell responses and of TReg induction in an IDO-dependent manner. Taken together, our data suggests that CLL cells, MDSCs, and TRegs interact within an immunosuppressive network that could represent a significant source for the hosts’ immune deficiencies.
Material and methods
Patients
The ethics committee of the University Hospital of Erlangen-Nuremberg approved this study (approval number: 200_12). All patients signed an informed consent in accordance with the Declaration of Helsinki. Samples were retrieved from 79 untreated CLL patients with a mean age of 69.37 ± 8.48 years and a male to female ratio of 1.6 to 1. At the time point of sampling, 61 patients had Binet stage A, 6 had stage B, and 12 had newly diagnosed stage C.
Paraffin-embedded B-cell CLL (B-CLL) infiltrated lymph nodes (n = 5) were selected from the archive of the Institute of Pathology (University Hospital Erlangen, Germany). Material was used in accordance with a general statement of our local ethical board on the anonymous use of tissue from the archive of the Institute of Pathology.
Cell lines
MEC-1 and EHEB CLL cell lines were purchased from DSZM (Braunschweig, Germany) and the bone marrow stromal cell line HS-5 was purchased from ATCC (Manassas, VA).
Reagents
Dulbecco's phosphate buffered saline, AIM-V, RPMI 1640, Iscove modified Dulbecco medium, L-glutamine, CFSE, and Trypan blue were purchased from Invitrogen (Carlsbad, CA), 1-methyl-dl-tryptophan (1-MT), catalase, celecoxib, NS398, N-acetyl-l-cysteine, dimethyl sulfoxide, β-mercaptoethanol, phytohemagglutinin (PHA), phorbol-12-myristate-13-acetate, and penicillin–streptomycin from Sigma-Aldrich (St. Louis, MO). VPD450 was purchased from BD Biosciences (San Jose, CA). Fetal bovine serum was purchased from PAN Biotech (Aidenbach, Germany), human serum albumin from Baxter (Deerfield, MA), and EDTA from Merck Millipore (Readington Township, NJ).
Cell isolation
Peripheral blood mononuclear cells (PBMCs) were isolated by density gradient-based Ficoll-Paque (GE Healthcare, Waukesha, WI) and stored in liquid nitrogen. CD14+ monocytes, CD19+ B-/CLL-cells, and CD3+ T cells were purified using magnetic beads (CD14 microbeads/B-cell isolation kit II/Pan-T-cell isolation kit II, Miltenyi Biotec; Bergisch Gladbach, Germany) at a purity level of ≥90%. For certain experiments, CD3+ T cells, CD14+HLA-DRlo cells and CD3+CD4+CD25hiCD127low TRegs were purified using fluorescence-activated cell sorter (FACS)-based sorting (FACS ARIA II; BD Biosciences) (purity ≥95%).
Coculture experiments
VPD450-labeled primary CLL cells or with 30 Gy irradiated cell lines were cocultured with PBMCs (with/without prior monocyte depletion), purified T cells, or monocytes retrieved from HDs. In indicated experiments, purified, autologous monocytes were titrated into cocultures of T cells and CLL cells. Experiments were performed with cells grown either in close contact or Transwell plates (Thermo Scientific, Waltham, MA). T-cell stimulation was performed using activating anti-CD2, -CD3, and -CD28 bead-coupled antibodies (Miltenyi Biotec) or PHA.
T-cell proliferation assay
CD14+HLA-DRlo cells purified from CLL patients’ PBMCs or induced CD14+HLA-DRlo cells purified from coculture experiments were subsequently cocultured with autologous VPD-labeled T cells in different ratios. Purified induced TRegs were similarly cocultured with autologous T cells. The T cells were stimulated using anti-CD2, -CD3, and -CD28 bead-coupled antibodies (Miltenyi Biotec) or PHA.
Antibodies and flow cytometry
Cells were stained according to the manufacturer’s recommendations using fluorochrome-coupled antibodies as detailed in supplemental Table 1 (available on the Blood Web site). For intracellular staining, cells were fixed and permeabilized using BD Cytofix/Cytoperm reagents (BD Biosciences) in line with the manufacturer’s instructions. For the exclusion of dead cells, LIVE/DEAD aqua fixable dead cell staining kit (Invitrogen) was applied. Cells were acquired and analyzed using a FACS Canto II flow cytometer (BD Biosciences) and FlowJo, version 9.0.2 software (TreeStar, San Carlos, CA).
ROS
Cellular ROS were quantified by FACS using CellROX dyes (Life Technologies, Carlsbad, CA).
Light microscopy
For cytological analyses, FACS-sorted CD14+DRlo cells were subjected to cytospin preparation and stained according to Pappenheim (Giemsa/May-Grünwald).
Immunofluorescence
Paraffin sections (2 µm) were dewaxed in xylol and rehydrated through graded ethanol baths. For antigen retrieval, slides were cooked for 5 minutes at 120°C (Biocarta Europe, Hamburg, Germany). Primary antibodies specific for IDO (1:50, MAB5412, mouse IgG3; Merck Millipore) and CD33 (ready to use, PWS44, mouse IgG2b; Cell Marque, Rocklin, CA) were added overnight. Thereafter, fluorescence-labeled secondary antibodies specific for murine IgG3, Alexa Fluor 488 (1:200 115-545-209; Dianova, Hamburg, Germany) and murine IgG2b, Alexa Fluor 594 (1:200 A21145; Life Technologies) were applied. The 4′,6 diamidino-2-phenylindole (Life Technologies) was used for nuclear staining.
Analysis of tryptophan metabolites
Serum specimens were stored at −80°C until liquid chromatography electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) of tryptophan, 3-hydroxykynurenine, nicotinamide, kynurenine, xanthurenic acid, kynurenic acid, indole-3-lactic acid, indole-3-propionic acid, and indole-3-acetic was performed as previously described.18
Cytokine measurements
IFN-γ was quantified in supernatants using an IFN-γ ELISA (R&D Systems, Minneapolis, MN), IL-1β, IL-6, IL-10, granulocyte macrophage–CSF, and TNFα using an Inflammatory Cytokine Multi-Analyte ELISArray kits (Qiagen, Hilden, Germany).
RNA preparation and quantitative polymerase chain reaction
Total RNA was extracted using RNeasy mini kits (Qiagen) and cDNA prepared (random decamers, deoxyribonucleotide triphosphates, ThermoScript, and dithiothreitol were all purchased from Invitrogen) using a Mastercycler Nexus (Eppendorf). Messenger RNA levels were quantified by real-time polymerase chain reaction (PCR) (QuantiTect SYBR Green and Rotor Gene Q; Qiagen). Relative gene expression was determined by normalizing the expression of each target gene to β-actin. Gene-specific primers are detailed in supplemental Table 2.
Statistical analysis
Differences in means and correlation analyses were evaluated with parametric (2-tailed student or paired Student t test and Pearson test) or nonparametric (Mann-Whitney U test or Wilcoxon and Spearman r tests) based on the distribution levels. All statistical analyses were performed using GraphPad Prism, version 5 (GraphPad Prism Software Inc., San Diego, CA) at a significance level of P < .05.
Results
Increased levels of circulating CD14+HLA-DRlo cells in untreated CLL patients
Frequency of CD14+HLA-DRlo monocytes among (non-CLL) PBMCs was similar to previous findings in B-cell derived malignancies,19 significantly increased in CLL patients as compared with HDs (5.80 ± 3.17% in CLL vs 0.54 ± 0.62% in HD) (Figure 1A). This accumulation was a result of an increased proportion of cells with low and/or absent HLA-DR expression among monocytes (51.19 ± 21.97% in CLL vs 3.28 ± 4.53% in HD) and not of a relative monocytosis (13.40 ± 6.19% in CLL vs 10.86 ± 5.42% in HD) (Figure 1B).
Increased frequency of CD14+HLA-DRlo cells in the peripheral blood of untreated CLL patients. (A) Representative dot plots from flow cytometric (FACS) analyses showing the HLA-DRlo cell frequency among peripheral blood CD14+ monocytes obtained from 37 healthy controls (HD) and 41 untreated CLL patients. (B) Frequency of HLA-DRlo cells and CD14+ cells among monocytes and (non-CLL) PBMCs, respectively, were compared between HD (n = 40) and CLL patients (n = 44). (C) Representative histograms of the phenotypic FACS analyses of CD14+HLA-DRlo cells from CLL patient samples (fine black line = isotype control and solid gray line = specific antibody). (D) Representative light microscopy image of cytospin preparations of FACS-sorted CD14+HLA-DRlo cells from CLL patients stained according to Pappenheim. (E) A representative dot plot of the FACS-based DC subset analysis. The linnegHLA-DR+ DCs were further subdivided into CD11c+ myeloid (m)DCs and CD123+ plasmacytoid (p)DCs. (F) Correlation analyses of the peripheral mDC and pDC counts with the proportion of HLA-DRlo cells among CD14+ cells in CLL patients. The bars represent the standard error of the mean. ***P < .001.
Increased frequency of CD14+HLA-DRlo cells in the peripheral blood of untreated CLL patients. (A) Representative dot plots from flow cytometric (FACS) analyses showing the HLA-DRlo cell frequency among peripheral blood CD14+ monocytes obtained from 37 healthy controls (HD) and 41 untreated CLL patients. (B) Frequency of HLA-DRlo cells and CD14+ cells among monocytes and (non-CLL) PBMCs, respectively, were compared between HD (n = 40) and CLL patients (n = 44). (C) Representative histograms of the phenotypic FACS analyses of CD14+HLA-DRlo cells from CLL patient samples (fine black line = isotype control and solid gray line = specific antibody). (D) Representative light microscopy image of cytospin preparations of FACS-sorted CD14+HLA-DRlo cells from CLL patients stained according to Pappenheim. (E) A representative dot plot of the FACS-based DC subset analysis. The linnegHLA-DR+ DCs were further subdivided into CD11c+ myeloid (m)DCs and CD123+ plasmacytoid (p)DCs. (F) Correlation analyses of the peripheral mDC and pDC counts with the proportion of HLA-DRlo cells among CD14+ cells in CLL patients. The bars represent the standard error of the mean. ***P < .001.
Next, we analyzed the phenotype of circulating CD14+HLA-DRlo cells in CLL (Figure 1C). The cells expressed myeloid markers (CD11c, CD13, CD33), adhesion molecules (CD11b, CD62L), and receptors (TNF-receptor type 2/CD120b)11 associated with promotion and activation of myeloid cells. In line with their monocytic commitment CD14+HLA-DRlo cells were positive for the macrophage colony-stimulating factor receptor (CD115) and expressed the IL-4α receptor (CD124), with both molecules having been linked to MDSC activity.20,21 Cells were positive for both the hemoglobin-haptoglobin scavenger receptor CD163, which represents a useful biomarker for anti-inflammatory M2-monocytes,22 and HLA-G, which has been shown to act tolerogeneic in MDSCs.23 When evaluating members of the B7 family, we found the programmed cell death ligand 1 (PD-L1/CD274)24 on CD14+HLA-DRlo cells, but no CD80 and CD86.13 Furthermore, cells expressed the co-stimulatory CD40 that has been shown to impact their ability to promote T-cell anergy and TReg induction.25 Finally, CD14+HLA-DRlo cells were negative for markers characteristic for granulocytes (CD15, CD66b), markers for mature myeloid cells (CD1a) or for nonclassic monocytes (CD16), and the IL-3 receptor (CD123).
To validate whether the monocytic phenotype actually displayed a characteristic monocytic morphology, we FACS-sorted CD14+HLA-DRlo cells from patients’ PBMCs and characterized them on cytospins. Indeed, the purified CD14+HLA-DRlo cell fraction uniformly contained cells resembling monocytes displaying blue-gray cytoplasm, occasional vacuoles, and lobulated nuclei (Figure 1D).
An underlying differentiation arrest of myeloid progenitors responsible for the accumulation of aberrant monocytic cells in patients could also impact dendritic cell (DC) differentiation. To address this question, we evaluated the DC subsets lineagenegHLA-DRposCD11cposCD123neg myeloid (mDCs) and lineagenegHLA-DRposCD11cnegCD123pos plasmacytoid (pDCs) in CLL patients (Figure 1E). As shown in Figure 1F, the HLA-DRlo fraction among CD14+cells correlated negatively with circulating mDCs and pDCs.
CD14+HLA-DRlo cells from CLL patients suppress T cells and are associated with a shorter lymphocyte doubling time
In the following, CLL patients’ aberrant monocytes and T cells were FACS-sorted. For proofing the in vitro suppressive capacity of CD14+HLA-DRlo cells, autologous T cells were bead-stimulated in their presence and proliferation was measured. In fact, CD14+HLA-DRlo cells suppressed T-cell proliferation significantly and in a dose-dependent manner (Figure 2A). This key functional finding allowed us to finally denominate the MDSCs.
Impact (in vitro and ex vivo) of CD14+HLA-DRlo cells from CLL patients on T cells. (A) The dose-dependent suppressive activity of FACS-sorted CD14+HLA-DRlo cells from CLL patients (n = 4) was evaluated in coculture experiments with autologous T cells activated by means of anti-CD2, -CD3, and -CD28 microbeads. Proliferation of T cells was assessed based on the VPD450 dye dilution after 5 days by FACS and compared with stimulated T cells alone (set as 100% T-cell proliferation). (B) Correlation of the T-cell count with the proportion of CD14+HLA-DRlo cells in the peripheral blood of CLL patients. (C) Patients were grouped based on their MDSC (equals CD14+HLA-DRlo cells) frequency (less than the median [n = 24] and greater than the median [n = 24]). Frequency of PD1+ cells among CD4+/CD8+ T cells was compared between the groups. (D) Time until doubling of the patients’ lymphocyte counts was compared in the 2 groups using a Gehan-Breslow-Wilcoxon test. (E) T-cell activation based on mean fluorescence index (MFI) levels of CD69, CD137, and HLA-DR, respectively, in the CD4+ and CD8+ T-cell compartment was compared between the groups. (F) Representative dot plot of a FACS-based T-cell maturation analysis. CD4+ and CD8+ T cells were divided into CD27+CD45RA+ naïve (NA) cells, CD27+CD45RAneg central memory (CM), CD27negCD45RAneg effector memory cells, and CD27negCD45RA+ effector memory RA+ cells (EMRA) cells. Proportions of these T-cell subsets were analyzed in both patient groups. The bars represent the standard error of the mean. *P < .05; **P < .01.
Impact (in vitro and ex vivo) of CD14+HLA-DRlo cells from CLL patients on T cells. (A) The dose-dependent suppressive activity of FACS-sorted CD14+HLA-DRlo cells from CLL patients (n = 4) was evaluated in coculture experiments with autologous T cells activated by means of anti-CD2, -CD3, and -CD28 microbeads. Proliferation of T cells was assessed based on the VPD450 dye dilution after 5 days by FACS and compared with stimulated T cells alone (set as 100% T-cell proliferation). (B) Correlation of the T-cell count with the proportion of CD14+HLA-DRlo cells in the peripheral blood of CLL patients. (C) Patients were grouped based on their MDSC (equals CD14+HLA-DRlo cells) frequency (less than the median [n = 24] and greater than the median [n = 24]). Frequency of PD1+ cells among CD4+/CD8+ T cells was compared between the groups. (D) Time until doubling of the patients’ lymphocyte counts was compared in the 2 groups using a Gehan-Breslow-Wilcoxon test. (E) T-cell activation based on mean fluorescence index (MFI) levels of CD69, CD137, and HLA-DR, respectively, in the CD4+ and CD8+ T-cell compartment was compared between the groups. (F) Representative dot plot of a FACS-based T-cell maturation analysis. CD4+ and CD8+ T cells were divided into CD27+CD45RA+ naïve (NA) cells, CD27+CD45RAneg central memory (CM), CD27negCD45RAneg effector memory cells, and CD27negCD45RA+ effector memory RA+ cells (EMRA) cells. Proportions of these T-cell subsets were analyzed in both patient groups. The bars represent the standard error of the mean. *P < .05; **P < .01.
The percentage of aberrant monocytes that correlated negatively with the absolute number of circulating T cells was similar to findings from patients with B-cell non-Hodgkin lymphomas (B-NHLs)19 (Figure 2B). Data from previous studies in CLL had shown the emergence of T cells with a PD1+ replicative senescence phenotype.26,27 Although data from preclinical models had suggested that MDSCs might play a role in the mechanisms responsible for immune senescence,28 we could not detect differences in circulating PD1+ CD4+ and CD8+ T cells between patients with lower (less than the median) and higher (greater than the median) levels of MDSCs (Figure 2C).
However, we observed that an increased MDSC frequency was associated with a shorter time period until lymphocyte doubling (Figure 2D). Regarding other prognostic markers, we found no significant differences in CD38 expression (P = .18), ZAP-70 expression (P = .26), β2-microglobulin serum concentration (P = .91), and immunoglobulin heavy variable cluster mutation status (P = .32) between the groups with lower and higher levels of MDSCs, respectively.
Analyzing T-cell subsets in terms of expression (based on the so-called MFI) of early and late activation markers29 yielded a significant inverse association between MDSC frequency and the surface density of CD69, CD137, and HLA-DR on CD4+ T cells, and of CD137 and HLA-DR on CD8+ T cells, respectively (Figure 2E). MDSCs can lead to a downregulation of the signal-transducing CD3ζ chain in T cells,29 but this was not the case in our examined cohort (data not shown). Previous reports have shown a skewing of the T-cell compartment toward activated T cells in CLL patients.26,27 Higher MDSC levels were linked to significantly lower frequencies of both CD4+ and CD8+ CD45RA+CD27− effector memory RA+ cells (EMRA) T cells that are indicative for a persistent stimulation (5.78 ± 1.22% CD4+ EMRA T cells in patients with MDSCs less than the median vs 2.90 ± 0.54% in patients with MDSCs greater than the median and 13.72 ± 1.99% CD8+ EMRA T cells in patients with MDSCs less than the median vs 5.83 ± 0.93% in patients with MDSCs greater than the median) (Figure 2F). CD45RA+CD27+ naïve CD8+ T cells were decreased (9.00 ± 1.33% vs 13.92 ± 1.84%) and CD45RA−CD27− effector memory CD8+ T cells increased (47.93+/−4.01% vs 33.96 ± 3.51%) in patients with a higher MDSC fraction (Figure 2F). No correlation between inversion of the CD4/CD8 ratio26 and MDSCs was detected (data not shown). Taken together, this data suggested a potential (negative) role for MDSCs in the immunologic control of CLL-cell expansion.
CLL cells induce MDSCs with a T-cell suppressive and TReg-promoting capacity
Comparing the phenotype of CD14+HLA-DRlo cells from CLL patients and HDs revealed a significantly increased expression of the secondary lymphoid selectin/adhesion molecule CD62L,30 CD274,24 and HLA-G23 on CLL-derived cells (Figure 3A). ROS levels13 were not altered significantly (supplemental Figure 1).
CLL cells induce in vitro CD14+HLA-DRlo MDSCs with T-cell suppressive and TReg-promoting capabilities. (A) Fold-expression of surface markers based on their mean fluorescence intensity (MFI) on CD14+HLA-DRlo cells from patients (CLL [n = 10]) and healthy controls (HD [n = 10]). (B) Expression levels of HLA-DR, CD62L, CD274, and HLA-G, respectively, expressed as their MFI on purified HD-derived CD14+ monocytes cultured for 5 days in absence/presence of primary CLL cells and CLL cell lines (n = 4-22). (C) The dose-dependent suppressive activity CD14+HLA-DRlo cells converted from healthy control monocytes (n = 4) in presence of CLL cells was evaluated in coculture experiments with VPD450-labeled autologous T cells activated using anti-CD2, -CD3, and -CD28 microbeads. Proliferation of T cells was assessed based on the VPD450 dye dilution in activated T cells after 5 days by FACS and compared with stimulated T cells alone (set as 100% T-cell proliferation). (D) Representative dot plot of a TReg FACS analysis from the peripheral blood of CLL patients. Naturally occurring TRegs were defined as CD4+ T cells positive for CD25 and FOXP3 and low/negative for CD127. Frequency of TRegs among CD4+ T cells is shown for healthy controls (HD [n = 13]) and CLL patients (n = 43). (E) Increase of the TReg frequency among CD4+ T cells of HDs after coculture with autologous CLL-induced CD14+HLA-DRlo MDSCs. (F) The CD120b MFI as assessed by FACS on TRegs from CLL patients (n = 9), TRegs induced in the presence of MDSCs (n = 5), and TRegs from HD (n = 14). (G) Correlation between the TReg frequency among CD4+ T cells and the proportion of HLA-DRlo cells among CD14+ cells in the peripheral blood of CLL patients. The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.
CLL cells induce in vitro CD14+HLA-DRlo MDSCs with T-cell suppressive and TReg-promoting capabilities. (A) Fold-expression of surface markers based on their mean fluorescence intensity (MFI) on CD14+HLA-DRlo cells from patients (CLL [n = 10]) and healthy controls (HD [n = 10]). (B) Expression levels of HLA-DR, CD62L, CD274, and HLA-G, respectively, expressed as their MFI on purified HD-derived CD14+ monocytes cultured for 5 days in absence/presence of primary CLL cells and CLL cell lines (n = 4-22). (C) The dose-dependent suppressive activity CD14+HLA-DRlo cells converted from healthy control monocytes (n = 4) in presence of CLL cells was evaluated in coculture experiments with VPD450-labeled autologous T cells activated using anti-CD2, -CD3, and -CD28 microbeads. Proliferation of T cells was assessed based on the VPD450 dye dilution in activated T cells after 5 days by FACS and compared with stimulated T cells alone (set as 100% T-cell proliferation). (D) Representative dot plot of a TReg FACS analysis from the peripheral blood of CLL patients. Naturally occurring TRegs were defined as CD4+ T cells positive for CD25 and FOXP3 and low/negative for CD127. Frequency of TRegs among CD4+ T cells is shown for healthy controls (HD [n = 13]) and CLL patients (n = 43). (E) Increase of the TReg frequency among CD4+ T cells of HDs after coculture with autologous CLL-induced CD14+HLA-DRlo MDSCs. (F) The CD120b MFI as assessed by FACS on TRegs from CLL patients (n = 9), TRegs induced in the presence of MDSCs (n = 5), and TRegs from HD (n = 14). (G) Correlation between the TReg frequency among CD4+ T cells and the proportion of HLA-DRlo cells among CD14+ cells in the peripheral blood of CLL patients. The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.
Monocytes isolated from HD were cocultured for 5 days with primary CLL cells and the human CLL cell-lines EHEB and MEC-1 based on our data from time kinetic experiments on HLA-DR and CD62L expression (supplemental Figure 2). They displayed phenotypic alterations resembling the phenotype of the primary (p)MDSCs found in the peripheral blood of CLL patients. Cells displayed a markedly reduced expression of HLA-DR and significantly upregulated CD62L, CD274, and HLA-G (Figure 3B). Phenotypic changes were reversible as tested for HLA-DR and CD62L (supplemental Figure 2). Presence of bone marrow stromal cells that have been shown to impact the biology of the CLL cells31 exerted no additional effect on these alterations (supplemental Figure 3). The functional assessment of CLL-educated monocytes, in line with their MDSC-like phenotype, showed a strong T-cell suppressive capacity (Figure 3C) that was independent of T-cell receptor activation and classical co-stimulation (supplemental Figure 4).
In accordance with previous studies,32 CLL patients exhibited significantly elevated levels of peripheral CD25hiCD127loFOXP3+ CD4+ TRegs (Figure 3D). Co-culturing purified CD4+ T cells from HD with in vitro generated induced MDSCs (iMDSCs) promoted the TReg increase (Figure 3E and supplemental Figure 5). These cells, similar to their counterparts isolated from CLL patients, displayed higher expression levels of the TReg activation marker CD120b33 compared with TRegs from HD (Figure 3F). Finally, and in line with these in vitro observations, TReg and MDSC frequencies correlated positively ex vivo in CLL patients (Figure 3G).
Myeloid cells enhance the TReg-promoting capacity of CLL cells
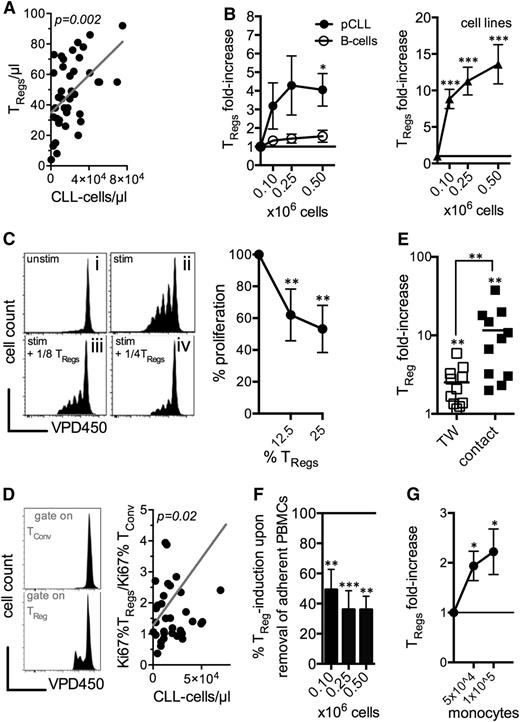
Previous studies had shown that malignant B cells from B-NHLs were capable of converting conventional T cells into TRegs, explaining in part the TReg accumulation in these patients.34 Our data showed a significant correlation of the number of CLL cells with the number of TRegs in the peripheral blood (Figure 4A), implying a comparable interconnection. To study the ability of CLL cells to drive TReg induction, we cocultured HD-derived B-cells (as controls), primary CLL cells, or CLL cell-lines with PBMCs from HD. Only CLL cells led to a significant increase in TRegs in a concentration-dependent fashion (Figure 4B). Presence of HS-5 cells did not further boost TReg induction (supplemental Figure 6). Our functional evaluation of FACS-sorted CD25hiCD127loCD4+ T cells from these cocultures confirmed their activity in suppressing autologous T-cell responses (Figure 4C). The TRegs displayed a preferential cell division35 when compared with conventional T cells after coculture with malignant cells (Figure 4D). This observation was strongly supported by our findings in CLL patients, in whom skewed ratios between proliferating Ki67+ TRegs and conventional T cells (Tconv) correlated significantly with the number of circulating tumor cells (Figure 4D).
Myeloid cells cosynergize with CLL cells in promoting TReg expansion. (A) Correlation of the absolute TReg number with the CLL cell number in the peripheral blood of CLL patients. (B) Fold-increase in the TReg frequency in HD-derived PBMCs cocultured in the presence/absence of an increasing number of autologous B-cells (n = 6), primary (p)CLL-cells (n = 5), and the 2 CLL cell lines (used in n = 12 independent experiments) (the baseline = 1 represents the frequency of TRegs in the absence of cocultured cells). (C) Representative histogram of the dose-dependent suppressive activity of purified TRegs induced in the presence of CLL cells. TRegs suppressed VPD450-labeled autologous T cells activated by anti-CD2, -CD3, and -CD28 microbeads (n = 4). Proliferation of T cells was assessed after 5 days by FACS and compared with activated T cells alone (set as 100% T-cell proliferation). (D) Representative (for n = 4 independent experiments) histogram of the proliferation of conventional CD4+ T cells (TConv) and TRegs based on the VPD450 dilution after coculture with CLL cells. Ratio of the percentages of proliferating Ki67+ TRegs to Ki67+ TConv cells in relation to the number of circulating CLL cells in the patients. (E) Fold-increase in TReg frequencies among PBMCs cocultured in the absence/presence of CLL cells (n = 4) or 2 CLL cell lines (used in n = 7 independent experiments) being separated by a Transwell polyester membrane (TW) or in close cell-to-cell contact (the baseline = 1 represents the frequency of TRegs in the absence of cocultured cells). (F) Percentage induction of TRegs with removal of adherent cells before coculture of PBMCs retrieved from healthy controls with CLL cells (n = 8-10) (TReg induction without removal of adherent cells is set as 100%). (G) CD3+ T cells purified from HD PBMCs were cocultured with CLL cell lines in 4 independent experiments in absence/presence of autologous purified CD14+ monocytes (the baseline = 1 represents the frequency of TRegs in the absence of monocytes). The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.
Myeloid cells cosynergize with CLL cells in promoting TReg expansion. (A) Correlation of the absolute TReg number with the CLL cell number in the peripheral blood of CLL patients. (B) Fold-increase in the TReg frequency in HD-derived PBMCs cocultured in the presence/absence of an increasing number of autologous B-cells (n = 6), primary (p)CLL-cells (n = 5), and the 2 CLL cell lines (used in n = 12 independent experiments) (the baseline = 1 represents the frequency of TRegs in the absence of cocultured cells). (C) Representative histogram of the dose-dependent suppressive activity of purified TRegs induced in the presence of CLL cells. TRegs suppressed VPD450-labeled autologous T cells activated by anti-CD2, -CD3, and -CD28 microbeads (n = 4). Proliferation of T cells was assessed after 5 days by FACS and compared with activated T cells alone (set as 100% T-cell proliferation). (D) Representative (for n = 4 independent experiments) histogram of the proliferation of conventional CD4+ T cells (TConv) and TRegs based on the VPD450 dilution after coculture with CLL cells. Ratio of the percentages of proliferating Ki67+ TRegs to Ki67+ TConv cells in relation to the number of circulating CLL cells in the patients. (E) Fold-increase in TReg frequencies among PBMCs cocultured in the absence/presence of CLL cells (n = 4) or 2 CLL cell lines (used in n = 7 independent experiments) being separated by a Transwell polyester membrane (TW) or in close cell-to-cell contact (the baseline = 1 represents the frequency of TRegs in the absence of cocultured cells). (F) Percentage induction of TRegs with removal of adherent cells before coculture of PBMCs retrieved from healthy controls with CLL cells (n = 8-10) (TReg induction without removal of adherent cells is set as 100%). (G) CD3+ T cells purified from HD PBMCs were cocultured with CLL cell lines in 4 independent experiments in absence/presence of autologous purified CD14+ monocytes (the baseline = 1 represents the frequency of TRegs in the absence of monocytes). The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.
Next, we cocultured both primary CLL cells and the cell lines MEC-1 and EHEB with HD PBMCs, either in direct physical contact or separated by a semipermeable membrane (TW) and compared the degree of TReg induction (increase of TRegs 11.58 ± 10.72-fold [P = .008] with cell-to-cell contact vs 2.51 ± 1.46-fold [P = .006] in TW) (Figure 4E). Our results suggest that cell-to-cell contact was not strictly needed for a significant CLL cell-mediated promotion of TRegs, but obviously significantly (P = .009) augmented the capacity of the CLL cells in this regard.
We adapted our CLL-cell interaction in vitro model by depleting the nonadherent HD PBMCs before conducting the coculture experiments. Absence of antigen-presenting cells, mostly monocytes, led to a significant diminishment of the capability of the CLL cellsto promote TRegs (Figure 4F). As anticipated, adding purified HD CD14+ monocytes into a coculture of purified HD CD3+ T cells and CLL cells further bolstered TRegs induction in a (monocyte-) dose-dependent manner (Figure 4G).
CLL MDSCs express high levels of IDO
MDSCs use numerous immune-regulating mechanisms.36 Therefore, we initially investigated the potential induction of immune-modulating molecules in isolated HD monocytes cocultured for 5 days with CLL cells in cell-to-cell contact, separated by a semipermeable membrane, or in the presence of CLL-cell supernatant. Under all conditions, we detected a significant induction of IDO, which was the highest when cells were grown in direct contact (6217 ± 12 473-fold, 1217 ± 1884-fold, and 4.19 ± 3.45-fold increase in IDO relative gene expression for cell-to-cell contact, membrane-separated growth, and CLL cell supernatant only, respectively) (Figure 5A). In line with this data, we found significantly higher IDO protein levels in CD14+ cells cocultured with primary CLL cells (supplemental Figure 7) and ex vivo in CD14+ cells from CLL patients as compared with cells from HD (Figure 5B). HS-5 cells had no further impact on IDO expression (supplemental Figure 7). Furthermore, the HLA-DRlo cells among the monocytic fraction in CLL PBMCs had the highest intracellular IDO content (equals IDOhi) (Figure 5B). In terms of the underlying mechanisms, we evaluated several (mainly proinflammatory) cytokines and ROS without indications for a relevant contribution (supplemental Figure 8).
CLL CD14+HLA-DRlo MDSCs express high levels of IDO. (A) The relative gene expression of MDSC-related molecules was comparatively assessed in healthy control (HD) monocytes cocultured with CLL cells (n = 7) in cell-to-cell contact, separated by a transwell system (TW) or in the presence of CLL cell supernatant (the baseline = 1 represents gene expression levels in monocytes in the absence of cocultured malignant cells). Arginase 1 and 2 (Arg1/2), inducible nitric oxide synthetase (inos), signal transducer, and activator transcription factor 3 (stat3), indoleamine 2,3 dioxygenase (ido), reduced NAD phosphate (NADPH) subunits p67 and gp91, IL-6 and IL-10, heme-oxygenase-1 (ho1), transforming growth factor β (TGF-β), and cyclooxygenase 2 (cox2) were included in the analyses. (B) Representative dot plots from FACS analyses evaluating the IDO expression in (i) CD14+ cells from HD and patients, as well as in (ii.-iii.) HLA-DRhi and HLA-DRlo CD14+ cells of CLL patients. IDO expression levels shown as the MFI were compared between CD14+ cells from HDs (n = 4) and HLA-DRhi and HLA-DRlo CD14+ cells from CLL patients (n = 4). (C) Coexpression of CD33 (red) and IDO (green) was analyzed in CLL infiltrated lymph nodes (n = 5) as shown in the representative fluorescence microscopy images. The 4′,6 diamidino-2-phenylindole (DAPI) (blue) was used as a nuclear counterstain. Five representative high-power fields (at ×400) were selected for evaluating the number of double-positive CD33+ IDO+ cells (white circles) and the proportion of IDO+ cells among total CD33+ cells. (D) Serum concentrations of tryptophan (Trp) and its degradation products 3-hydroxykynurenine (Hk), nicotinamide (Nam), kynurenine (Kyn), xanthurenic acid (Xa), kynurenic acid (Ka), indole-3-lactic acid (Ila), indole-3-propionic acid (Ipa), and indole-3-acetic (Iaa) were quantified in samples from the CLL patients with the highest MDSC (MDSChi [n = 10]) and the lowest MDSC (MDSClo [n = 10]) frequency by LC-ESI-MS/MS. The bars represent the standard error of the mean. n.d., not detected. *P < .05; **P < .01; ***P < .001.
CLL CD14+HLA-DRlo MDSCs express high levels of IDO. (A) The relative gene expression of MDSC-related molecules was comparatively assessed in healthy control (HD) monocytes cocultured with CLL cells (n = 7) in cell-to-cell contact, separated by a transwell system (TW) or in the presence of CLL cell supernatant (the baseline = 1 represents gene expression levels in monocytes in the absence of cocultured malignant cells). Arginase 1 and 2 (Arg1/2), inducible nitric oxide synthetase (inos), signal transducer, and activator transcription factor 3 (stat3), indoleamine 2,3 dioxygenase (ido), reduced NAD phosphate (NADPH) subunits p67 and gp91, IL-6 and IL-10, heme-oxygenase-1 (ho1), transforming growth factor β (TGF-β), and cyclooxygenase 2 (cox2) were included in the analyses. (B) Representative dot plots from FACS analyses evaluating the IDO expression in (i) CD14+ cells from HD and patients, as well as in (ii.-iii.) HLA-DRhi and HLA-DRlo CD14+ cells of CLL patients. IDO expression levels shown as the MFI were compared between CD14+ cells from HDs (n = 4) and HLA-DRhi and HLA-DRlo CD14+ cells from CLL patients (n = 4). (C) Coexpression of CD33 (red) and IDO (green) was analyzed in CLL infiltrated lymph nodes (n = 5) as shown in the representative fluorescence microscopy images. The 4′,6 diamidino-2-phenylindole (DAPI) (blue) was used as a nuclear counterstain. Five representative high-power fields (at ×400) were selected for evaluating the number of double-positive CD33+ IDO+ cells (white circles) and the proportion of IDO+ cells among total CD33+ cells. (D) Serum concentrations of tryptophan (Trp) and its degradation products 3-hydroxykynurenine (Hk), nicotinamide (Nam), kynurenine (Kyn), xanthurenic acid (Xa), kynurenic acid (Ka), indole-3-lactic acid (Ila), indole-3-propionic acid (Ipa), and indole-3-acetic (Iaa) were quantified in samples from the CLL patients with the highest MDSC (MDSChi [n = 10]) and the lowest MDSC (MDSClo [n = 10]) frequency by LC-ESI-MS/MS. The bars represent the standard error of the mean. n.d., not detected. *P < .05; **P < .01; ***P < .001.
In CLL lymph nodes, we detected IDO+ myeloid cells in line with previous observations in breast cancer patients12 (Figure 5C). The proportion of IDO-expressing cells among infiltrating myeloid cells was 35.60 ± 19.22%. Lymph node homing mediated by an increased CD62L expression on MDSCs (Figure 3A), for example, and/or a CLL-mediated induction (Figure 3B and 5A) from recruited monocytes might be causative. Next, we measured the concentrations of tryptophan and its key degradation products in serum specimens from patients with the highest frequency (MDSChi [n = 10]) and the lowest frequency (MDSClo [n = 10]) of aberrant monocytes, respectively, without detecting any difference (Figure 5D).
CLL MDSCs exert their immune regulatory functions via IDO
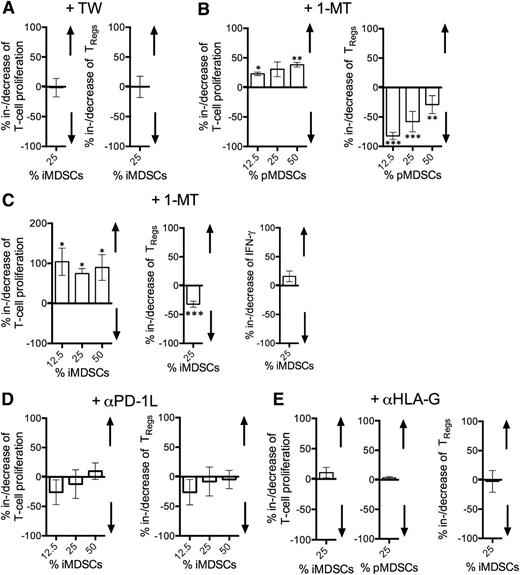
First, we evaluated whether separating iMDSCs from T cells by a semipermeable membrane affected their T-cell suppressive and/or TReg-promoting capacity. There were no differences detectable, suggesting that the mechanisms did not depend on cell-to-cell contact (Figure 6A). To investigate the biological significance of IDO expression, we used its competitive inhibitor 1-MT. We pretreated both, purified primary MDSCs (pMDSCs) and iMDSCs generated by coculturing HD monocytes with CLL cells, before using them in autologous T-cell suppression assays. Blocking IDO resulted in a significant increase in T-cell proliferation and a decrease in TReg induction (Figure 6B). In terms of IFN-γ release, presence of iMDSCs led to a mean reduction of approximately 62% (P = .003). When blocking IDO, we noticed by trend only a slight increase (+16%; P = .062), which suggested at least some efficacy of inhibiting IDO, but also the potential involvement of other suppressive mechanisms (Figure 6C).
CLL CD14+HLA-DRlo MDSCs suppress T cells and induce TRegs through IDO. (A) Induced (i)MDSCs converted from healthy control monocytes in the presence of CLL cells were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without being separated by a semipermeable membrane (TW) (n = 10). T-cell proliferation and TReg induction was assessed by FACS (T-cell proliferation/TReg induction without TW is set as 100%). (B) FACS-sorted primary (p)MDSCs from CLL-patients were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without pretreatment with 500 µM of the IDO inhibitor 1-methyl-dl-tryptophan (1-MT). T-cell proliferation (n = 3) and TReg induction (n = 5) was assessed by FACS (T-cell proliferation/TReg induction without 1-MT application is set as 100%). (C) FACS-sorted iMDSCs were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without pretreatment with 500 µM 1-MT (n = 5). T-cell proliferation, TReg induction, and IFNγ release was assessed by FACS and enzyme-linked immunosorbent assay, respectively (T-cell proliferation/TReg induction/IFN-γ release without 1-MT application is set as 100%). (D) FACS-sorted (i)MDSCs were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without addition of an anti-PD-1L blocking antibody (n = 5). T-cell proliferation and TReg induction was assessed by FACS (T-cell proliferation/TReg induction without the presence of an anti-PD-1L antibody is set as 100%). (E) FACS-sorted pMDSCs (n = 3) and/or iMDSCs (n = 11-16) were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without addition of an anti-HLA-G blocking antibody. T-cell proliferation and TReg induction was assessed by FACS (T-cell proliferation/TReg induction without the presence of an anti-HLA-G blocking antibody is set as 100%). The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.
CLL CD14+HLA-DRlo MDSCs suppress T cells and induce TRegs through IDO. (A) Induced (i)MDSCs converted from healthy control monocytes in the presence of CLL cells were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without being separated by a semipermeable membrane (TW) (n = 10). T-cell proliferation and TReg induction was assessed by FACS (T-cell proliferation/TReg induction without TW is set as 100%). (B) FACS-sorted primary (p)MDSCs from CLL-patients were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without pretreatment with 500 µM of the IDO inhibitor 1-methyl-dl-tryptophan (1-MT). T-cell proliferation (n = 3) and TReg induction (n = 5) was assessed by FACS (T-cell proliferation/TReg induction without 1-MT application is set as 100%). (C) FACS-sorted iMDSCs were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without pretreatment with 500 µM 1-MT (n = 5). T-cell proliferation, TReg induction, and IFNγ release was assessed by FACS and enzyme-linked immunosorbent assay, respectively (T-cell proliferation/TReg induction/IFN-γ release without 1-MT application is set as 100%). (D) FACS-sorted (i)MDSCs were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without addition of an anti-PD-1L blocking antibody (n = 5). T-cell proliferation and TReg induction was assessed by FACS (T-cell proliferation/TReg induction without the presence of an anti-PD-1L antibody is set as 100%). (E) FACS-sorted pMDSCs (n = 3) and/or iMDSCs (n = 11-16) were cocultured for 5 days with autologous VPD450-labeled activated T cells with/without addition of an anti-HLA-G blocking antibody. T-cell proliferation and TReg induction was assessed by FACS (T-cell proliferation/TReg induction without the presence of an anti-HLA-G blocking antibody is set as 100%). The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.
Because pMDSCs from CLL patients, as well as iMDSCs induced in the presence of CLL cells expressed high levels of the potentially T-cell inhibitory ligand PD-L1 (CD274) and HLA-G23 (Figure 3A-B), we performed cocultures with T cells and iMDSCs/pMDSCs in the presence of the corresponding blocking antibodies. In line with previous observations in PD-L1+ MDSCs,24 neutralizing PD-L1 did not efficiently abrogate T-cell suppression and/or TReg induction (Figure 6D). Similarly, blocking HLA-G had no substantial effect on the capacity of CLL MDSCs to inhibit T cells and to promote TRegs (Figure 6E).
Discussion
Here, we show the increased frequency of CD14+ cells lacking HLA-DR expression (CD14+HLA-DRlo) in the peripheral blood of predominantly early stage CLL patients. A recently published brief communication also reported an abundance of cells with an equivalent phenotype in CLL37 but without further in-depth characterization.
We observed that these CD14+HLA-DRlo cells displayed phenotypical and morphological characteristics of monocytes and were devoid of granulocytic markers. Taking into consideration the steadily growing list of proposed MDSC phenotypes, cells were termed MDSCs strictly based on their ability to suppress T cells.38 Concordant with previous studies on monocytic MDSCs,10 cells coexpressed CD11b, CD11c, CD33, CD115 (the macrophage CSF receptor), CD120b (the TNF-α receptor type 2), CD124 (the IL-4Rα), CD274 (PD-1L), HLA-G, and the co-stimulatory CD40 molecule.25 Furthermore, CD14+HLA-DRlo cells were similar to other types of MDSCs39 that were positive for the cell adhesion molecule CD62L (the l-selectin), which might enable them to enter secondary lymphoid organs and to interfere with T-cell priming.
One of the proposed mechanisms leading to MDSC accumulation is a differentiation arrest of myeloid-committed hematopoietic progenitors,10 which can differentiate among others into DCs. In addition, recent data suggests a bidirectional plasticity of already differentiated DCs into MDSCs.40 In accordance with findings from patients with B-NHL19 or after allogeneic stem-cell transplantation,29 we observed a significant inverse correlation between mDCs and pDCs, respectively, with MDSCs. This negative correlation might indicate both an overall skewing of myeloid differentiation toward MDSCs and/or a subsequent conversion of DCs into MDSCs.
MDSCs purified from patients efficiently suppressed T cells, whereas the proportion of MDSCs correlated negatively with the T-cell count in patients.19,29 One might speculate that this association resulted from the T-cell inhibitory effect exerted by MDSCs. However, this hypothesis requires further investigation also in terms of the impact of MDSCs on lymphoid progenitors taking into consideration that tumor-induced myelopoiesis is regularly linked to lymphopenia.41 Emerging data suggests that T-cell exhaustion contributes to T-cell dysfunction in CLL.26,27 Expression of the inhibitory receptor PD1 is of particular interest because expression of its ligand PD-L1 has been demonstrated to be involved in T-cell synapse dysfunctions in CLL41 and is also expressed on CLL MDSCs. Despite the preclinical data on MDSC-mediated immune senescence,28 we found no association between the frequency of PD1+ T cells and MDSCs. Consistent with our in vitro data on the T-cell inhibitory action of the MDSCs, patients with increased MDSC levels exhibited CD4+ and CD8+ T cells with a less activated phenotype and a lower proportion of chronically activated CD45RA+ effector memory (TEMRA, CCR7− CD45RA+) cells.
Recently, Gustafson et al37 reported that the abundance of CD14+HLA-DRlo cells is associated with a shorter time to CLL progression. This finding is supported by our results showing that patients with higher MDSC levels experienced an accelerated accumulation of circulating malignant cells. However, it remains to be elucidated which phenomenon (ie, accumulation of MDSCs or increased tumor burden) is causative, as both solid (eg, melanoma42 ) and hematologic (eg, multiple myeloma14 ) neoplastic cells can directly induce MDSCs.
Comparison of the phenotype of CLL MDSCs with CD14+HLA-DRlo immature myeloid cells from healthy controls13,29,43 revealed a higher expression of CD62L, CD274 (PD-1L), and HLA-G in the patients’ cells. In fact, coculturing CLL cells with HD-derived monocytes induced a similar HLA-DRloCD62LhiCD274hiHLA-Ghi phenotype. Moreover, CD14+HLA-DRlo cells retrieved from the cocultures were T-cell suppressive pointing toward at least a contributing role of CLL cells in MDSC accumulation. In addition to their direct T-cell suppressive effects, monocytic MDSCs can also recruit44 and induce immune suppressive TRegs. The TRegs accumulate in CLL and are often linked to advanced disease stage and worse prognosis.32 MDSCs generated in vitro in the presence of CLL cells were capable of promoting induction of TRegs that displayed an activated CD120bhi profile33 similar to their counterparts in CLL patients. Our ex vivo data, which shows an increased frequency of circulating TRegs in our patients’ collective and positive correlation between MDSCs and TRegs strongly implicates a biologically relevant crosstalk between CLL cells, MDSCs, and TRegs.
Previous work has shown that malignant B-cells can induce TRegs.5,34 In fact, primary CLL cells and cell lines promoted a preferential TReg expansion when cocultured with PBMCs from healthy controls. Accordingly, the ratio of proliferating Ki67+ TRegs to conventional proliferating Ki67+ T cells was skewed toward TRegs in relation to the circulating CLL-cell burden. This finding could reflect an advantageous (in terms of proliferation) environment established by the malignant cells.7 Similar to multiple myeloma,35 cell-to-cell contact was not a strict prerequisite for TReg induction, but rather acted as a significant boost. In this context, we could show that monocytes were a significant enforcer of CLL-cell–mediated TReg induction further corroborating the notion of an active crosstalk between CLL cells and the suppressive immune cell subsets.
It is well established that MDSCs use a broad variety of suppressive mechanisms. When evaluating the gene expression of several candidate molecules in healthy controls’ monocytes cocultured with CLL cells, we observed a significant upregulation of IDO. The IDO catabolizes the initial rate-limiting reaction of the kynurenine pathway. Both tryptophan depletion and kynurenine accumulation exert immune regulatory effects leading to T-cell suppression and TReg induction among others.45 Nowadays, IDO is considered a key endogenous immunologic checkpoint with a pivotal impact on tumor-associated immune tolerance.45 Monocytic MDSCs generated by cytokine treatment display an enhanced IDO expression.46,47 Furthermore, recent studies in patients with breast cancer12 or after stem cell transplantation29 showed phenotypically similar CD14+HLA-DRlo MDSCs expressing and utilizing IDO for T-cell inhibition. CLL patients display an increased IDO activity in their peripheral blood, but the cellular source remained unidentified with CLL cells having been ruled out.48
Here, we provide evidence that IDOhi monocytic MDSCs expand in CLL patients. They resemble the CD14+HLA-DRlo phenotype and the T-cell modulating activity of MDSCs identified in malignant diseases,12 as well as in highly inflammatory conditions.29
Overall, our in vitro and ex vivo data indicates a network of regulatory interactions between CLL cells, MDSCs, and TRegs. It is well-established that immune defects in CLL patients can negatively impact the course of disease, and it will be interesting to prospectively study the interconnection between MDSCs and overall survival.49 Pharmacologic inhibition of IDO may represent an alternative strategy for attenuating MDSC-mediated immune suppression. Furthermore, it remains to be elucidated how treatment with novel immunomodulatory agents such as lenalidomide50,51 affects MDSCs in CLL.
The online version of this article contains a data supplement.
There is an Inside Blood Commentary on this article in this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
We acknowledge the assistance of the Core Unit Cell Sorting and Immunomonitoring Erlangen.
This work was supported by grants from the Interdisziplinäres Zentrum für Klinische Forschung Erlangen (R.J.), the KFO262 (K.D.-W. and A.M.), the European Hematology Association, the Jose Carreras Leukemia Foundation, and the Max-Eder program of the Deutsche Krebshilfe (D.M.).
Authorship
Contribution: R.J., M. Braun, K.D.-W., J.Br., J.Be., S.W.K., P.J.O., and K.L.B. performed research; R.J., M. Braun, and D.M. analyzed data; M. Büttner performed research; M. Büttner provided patient material; M.J.E. collected patient material and provided patient data; A.M. helped design the study; R.J. and A.M. helped write the manuscript; and D.M. designed the study and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Dimitrios Mougiakakos, Department of Internal Medicine 5, Hematology and Oncology, University of Erlangen-Nuremberg, Ulmenweg 18, Erlangen, 90154 Germany; e-mail: dimitrios.mougiakakos@uk-erlangen.de.
References
Author notes
R.J. and M.B. contributed equally to this study.


![Figure 2. Impact (in vitro and ex vivo) of CD14+HLA-DRlo cells from CLL patients on T cells. (A) The dose-dependent suppressive activity of FACS-sorted CD14+HLA-DRlo cells from CLL patients (n = 4) was evaluated in coculture experiments with autologous T cells activated by means of anti-CD2, -CD3, and -CD28 microbeads. Proliferation of T cells was assessed based on the VPD450 dye dilution after 5 days by FACS and compared with stimulated T cells alone (set as 100% T-cell proliferation). (B) Correlation of the T-cell count with the proportion of CD14+HLA-DRlo cells in the peripheral blood of CLL patients. (C) Patients were grouped based on their MDSC (equals CD14+HLA-DRlo cells) frequency (less than the median [n = 24] and greater than the median [n = 24]). Frequency of PD1+ cells among CD4+/CD8+ T cells was compared between the groups. (D) Time until doubling of the patients’ lymphocyte counts was compared in the 2 groups using a Gehan-Breslow-Wilcoxon test. (E) T-cell activation based on mean fluorescence index (MFI) levels of CD69, CD137, and HLA-DR, respectively, in the CD4+ and CD8+ T-cell compartment was compared between the groups. (F) Representative dot plot of a FACS-based T-cell maturation analysis. CD4+ and CD8+ T cells were divided into CD27+CD45RA+ naïve (NA) cells, CD27+CD45RAneg central memory (CM), CD27negCD45RAneg effector memory cells, and CD27negCD45RA+ effector memory RA+ cells (EMRA) cells. Proportions of these T-cell subsets were analyzed in both patient groups. The bars represent the standard error of the mean. *P < .05; **P < .01.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/124/5/10.1182_blood-2013-12-546416/2/m_750f2.jpeg?Expires=1767830672&Signature=G~5RfRltvR7cpvJGVczuRMvTIBIUgNRwQHK9FSBY0NQgp4hMvgYbSc5fKK2oG9FF90dryetb0Kk5PaIN0yHHTD6LnfZZZPxhksrItvMDeFnFqQaLaXN2CC4dqggrL3i-qK9WTKPi-DJNRqhQ6ylOCLPfkeEffAEP~EZceLFm6xW8f4PytzEaQ7bQNMYNOEUVRjFw1k85L1NJfmBRRktN6cbPmsMApSKRIHwrw~K1xjbgpk-PxTcAwOKe2IxUPv0EbD9QkAz~sJHpjPJ~v31drE~Ld8nRT3IXj2h6smzfc7yXolomqXG1SVOO4f5ch0xlkcw-A6kpyOCJD7IbsPL5Iw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 3. CLL cells induce in vitro CD14+HLA-DRlo MDSCs with T-cell suppressive and TReg-promoting capabilities. (A) Fold-expression of surface markers based on their mean fluorescence intensity (MFI) on CD14+HLA-DRlo cells from patients (CLL [n = 10]) and healthy controls (HD [n = 10]). (B) Expression levels of HLA-DR, CD62L, CD274, and HLA-G, respectively, expressed as their MFI on purified HD-derived CD14+ monocytes cultured for 5 days in absence/presence of primary CLL cells and CLL cell lines (n = 4-22). (C) The dose-dependent suppressive activity CD14+HLA-DRlo cells converted from healthy control monocytes (n = 4) in presence of CLL cells was evaluated in coculture experiments with VPD450-labeled autologous T cells activated using anti-CD2, -CD3, and -CD28 microbeads. Proliferation of T cells was assessed based on the VPD450 dye dilution in activated T cells after 5 days by FACS and compared with stimulated T cells alone (set as 100% T-cell proliferation). (D) Representative dot plot of a TReg FACS analysis from the peripheral blood of CLL patients. Naturally occurring TRegs were defined as CD4+ T cells positive for CD25 and FOXP3 and low/negative for CD127. Frequency of TRegs among CD4+ T cells is shown for healthy controls (HD [n = 13]) and CLL patients (n = 43). (E) Increase of the TReg frequency among CD4+ T cells of HDs after coculture with autologous CLL-induced CD14+HLA-DRlo MDSCs. (F) The CD120b MFI as assessed by FACS on TRegs from CLL patients (n = 9), TRegs induced in the presence of MDSCs (n = 5), and TRegs from HD (n = 14). (G) Correlation between the TReg frequency among CD4+ T cells and the proportion of HLA-DRlo cells among CD14+ cells in the peripheral blood of CLL patients. The bars represent the standard error of the mean. *P < .05; **P < .01; ***P < .001.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/124/5/10.1182_blood-2013-12-546416/2/m_750f3.jpeg?Expires=1767830673&Signature=P7yket37iKPp7ZSU6IfqAWKXti55wWIUdKAu3wnshhJ5Hza-fr5-b-C-eR02aK4e9~tPQMG-wjHZoPbBkz2Bqn~CyXTldLifQ36Kg1-kBGXZvqMYGyRoB~9r-BYlHCUR24jgGQKYPlP5cQt4RzwX7P9iWflKxsolWhwBgCUTf2J9iPOxR2koZPNWP16VFKjYeEGggpdWZqKX8lphgYY7oEH9dqt4kdwhMgMdekguWQfD1ymr9hyZt05DWVVubklFm0jWrr9Ky-EvINDuKw9ogS74J8bZGMox5IIkHE9Fdo4NXjKMxA3EilR3T6JfXby0Jxu2OPDSW7ovVBGHKGTBPw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
![Figure 5. CLL CD14+HLA-DRlo MDSCs express high levels of IDO. (A) The relative gene expression of MDSC-related molecules was comparatively assessed in healthy control (HD) monocytes cocultured with CLL cells (n = 7) in cell-to-cell contact, separated by a transwell system (TW) or in the presence of CLL cell supernatant (the baseline = 1 represents gene expression levels in monocytes in the absence of cocultured malignant cells). Arginase 1 and 2 (Arg1/2), inducible nitric oxide synthetase (inos), signal transducer, and activator transcription factor 3 (stat3), indoleamine 2,3 dioxygenase (ido), reduced NAD phosphate (NADPH) subunits p67 and gp91, IL-6 and IL-10, heme-oxygenase-1 (ho1), transforming growth factor β (TGF-β), and cyclooxygenase 2 (cox2) were included in the analyses. (B) Representative dot plots from FACS analyses evaluating the IDO expression in (i) CD14+ cells from HD and patients, as well as in (ii.-iii.) HLA-DRhi and HLA-DRlo CD14+ cells of CLL patients. IDO expression levels shown as the MFI were compared between CD14+ cells from HDs (n = 4) and HLA-DRhi and HLA-DRlo CD14+ cells from CLL patients (n = 4). (C) Coexpression of CD33 (red) and IDO (green) was analyzed in CLL infiltrated lymph nodes (n = 5) as shown in the representative fluorescence microscopy images. The 4′,6 diamidino-2-phenylindole (DAPI) (blue) was used as a nuclear counterstain. Five representative high-power fields (at ×400) were selected for evaluating the number of double-positive CD33+ IDO+ cells (white circles) and the proportion of IDO+ cells among total CD33+ cells. (D) Serum concentrations of tryptophan (Trp) and its degradation products 3-hydroxykynurenine (Hk), nicotinamide (Nam), kynurenine (Kyn), xanthurenic acid (Xa), kynurenic acid (Ka), indole-3-lactic acid (Ila), indole-3-propionic acid (Ipa), and indole-3-acetic (Iaa) were quantified in samples from the CLL patients with the highest MDSC (MDSChi [n = 10]) and the lowest MDSC (MDSClo [n = 10]) frequency by LC-ESI-MS/MS. The bars represent the standard error of the mean. n.d., not detected. *P < .05; **P < .01; ***P < .001.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/124/5/10.1182_blood-2013-12-546416/2/m_750f5.jpeg?Expires=1767830673&Signature=MCScGwGk4cI-Yzr5bdUuZeGGoPnl4N~OqFd01tzWCyD-xM3Yj9v8GFl81zk3CKzR9qfuIfSxtCgmFTA13emxK1R9xOH6cxo251l8wQEY7g9h4fD725EzEEu~87C69XTYhHkTU3AtVJul0iqRfQdqpboVHG6un4Y1a9Y-4iUDxgSaULUSPAveEXtCEUm7QSW72qxppctc9bxPBmDosJtATkyvXBI2z2EAm7wItmxJWDBINv63TLTE3z5RjIQyXttf9SsWsYpUA~F2Z7pjiVMlMbJ5Hs8iJuU7U2eYp~E~7jTQ-8iinSiSou79OvqZvHuoiDMBGoxszxis~ZhEpcb3hw__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)
This feature is available to Subscribers Only
Sign In or Create an Account Close Modal