Abstract
Translocation t(12;21) (p13;q22), giving rise to the ETV6-RUNX1 fusion gene, is the most common genetic abnormality in childhood B-cell precursor acute lymphoblastic leukemia (BCP-ALL). The ETV6-RUNX1 translocation arises in utero, but its expression is insufficient to induce leukemia and requires other cooperating genetic lesions for BCP-ALL to develop. Deletions affecting the transcriptional coregulator BTG1 are commonly observed in BCP-ALL (9%), but are significantly enriched in ETV6-RUNX1-positive leukemia (25%). The BTG1 protein displays no intrinsic enzymatic activity but may act by recruiting effector molecules like protein arginine methyltransferase 1 (PRMT1) to specific transcription factors. Here, we show that ETV6-RUNX1 interacts both with BTG1 and PRMT1, and this interaction is lost in c-Kit+Ter-119-Btg1-/- fetal liver (FL) derived hematopoietic progenitors (HPCs). Moreover, targeted deletion of Btg1 enhanced the proliferative capacity of ETV6-RUNX1 in FL-HPCs as measured by enhanced colony-forming and serial replating capacity (Figure 1). The combined loss of Btg1 function and ETV6-RUNX1 expression correlated with strong upregulation of the proto-oncogene Bcl6 and downregulation of BCL6 target genes, such as p19Arf and Tp53 (Figure 2). Similarly, ectopic expression of BCL6 promoted both proliferation and replating capacity of FL-derived progenitor cells in the presence of SCF, FLT3L and IL-7. This phenotype correlated with a fivefold suppression of p19Arf and a twofold suppression of Tp53 expression. Inhibition of BCL6 in a variety of human BCP-ALL cell lines by the peptide inhibitor RI-BPI resulted in decreased proliferation and induction of apoptosis as measured by Annexin-V staining. These included the ETV6-RUNX1-positive cell lines UOC-B6, AT-2 and REH, the BCR-ABL1-positive cell line SD1, as well as Nalm6. Together our results point to a novel role for BCL6 in promoting cell proliferation of primitive progenitor B cells and suggest that targeted inhibition of BCL6 may be effective in the treatment of various BCP-ALL subtypes.
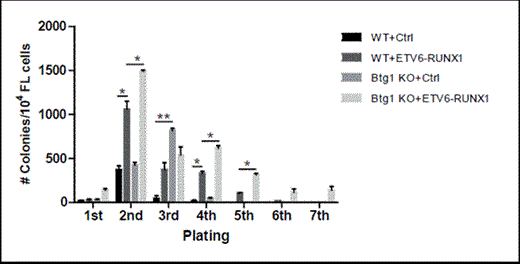
Btg1-deficiency enhances the proliferative capacity of early FL-HPCs expressing ETV6-RUNX1. FL-derived hematopoietic progenitor cells (FL-HPCs) (cKit+Ter119-) were isolated from wild-type and Btg1-/- embryos at day 13.5dpc and transduced with control and ETV6-RUNX1 virus. Control and ETV6-RUNX1 transduced FL-HPCs (1x104 cells) were added 48 hours after transduction in B cell specific methylcellulose in the presence of FLT-3L, IL-7 and SCF. Serial replating was performed under identical conditions. Mean colony counts (and SEM) were determined (>30 cells/colony) after 6-10 days of culture. Data is a representative of 2 independent experiments. *, P< 0.05, **, P< 0.01.
Btg1-deficiency enhances the proliferative capacity of early FL-HPCs expressing ETV6-RUNX1. FL-derived hematopoietic progenitor cells (FL-HPCs) (cKit+Ter119-) were isolated from wild-type and Btg1-/- embryos at day 13.5dpc and transduced with control and ETV6-RUNX1 virus. Control and ETV6-RUNX1 transduced FL-HPCs (1x104 cells) were added 48 hours after transduction in B cell specific methylcellulose in the presence of FLT-3L, IL-7 and SCF. Serial replating was performed under identical conditions. Mean colony counts (and SEM) were determined (>30 cells/colony) after 6-10 days of culture. Data is a representative of 2 independent experiments. *, P< 0.05, **, P< 0.01.
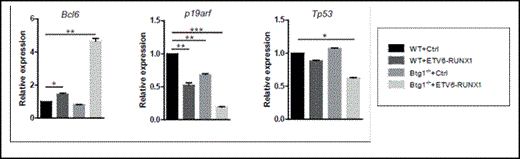
Targeted deletion of Btg1 cooperates with ETV6-RUNX1 in regulating critical effector pathways implicated in leukemia. Relative expression levels of Bcl6, Tp53 and p19arf in empty control (Ctrl) and ETV6-RUNX1 transduced wild-type and Btg1-deficient fetal liver-derived hematopoietic progenitor cells by real-time PCR and normalized to the expression of the housekeeping gene TATA box binding protein (TBP). Data represent the mean and SEM of three independent experiments. *, P< 0.05, **, P< 0.01, ***, P< 0.001.
Targeted deletion of Btg1 cooperates with ETV6-RUNX1 in regulating critical effector pathways implicated in leukemia. Relative expression levels of Bcl6, Tp53 and p19arf in empty control (Ctrl) and ETV6-RUNX1 transduced wild-type and Btg1-deficient fetal liver-derived hematopoietic progenitor cells by real-time PCR and normalized to the expression of the housekeeping gene TATA box binding protein (TBP). Data represent the mean and SEM of three independent experiments. *, P< 0.05, **, P< 0.01, ***, P< 0.001.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal