Abstract
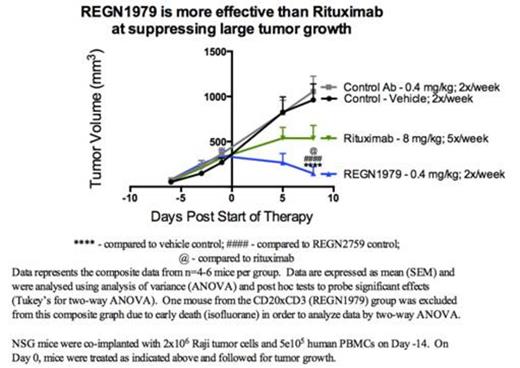
Bispecific antibodies that redirect effector T cells to kill tumor cells have shown considerable promise in both pre-clinical and clinical studies. However, these bispecific formats can have short half-lives necessitating constant infusion of the molecules into patients. We report here on a novel full-length human IgG CD20xCD3 bispecific antibody (REGN1979) that targets CD20 expressed on normal and malignant B cells and CD3 expressed on T cells in humans and cynomolgus monkeys. Our results demonstrate CD20-target cell-dependent activation and cytokine release by T cells, and efficient redirected T cell lysis of target tumor cells. Raji B cell lymphomas grown as tumors in NOD SCID IL2R gamma deficient (NSG) mice and co-implanted with human peripheral blood mononuclear (PBMC) cells were completely inhibited when treated at the time of implantation with a low dose (0.004 mg/kg; 2x/week) of REGN1979. As expected, T cells were required for this tumor inhibition, since treatment in the absence of human T cells was not effective. REGN1979 bispecific antibody also demonstrated potent activity against other tumor cells expressing CD20, as it significantly delayed CD20-transduced B16F10.9 tumor growth in immune-competent mice. Most importantly, REGN1979 induced dramatic tumor regression in large advanced (500-900 mm3) Raji tumors, associated with long-lasting tumor control. The tumor-infiltrating lymphocytes (TILs) in B cell lymphomas in these untreated NSG mice were found to express the inhibitory receptors Tim-3 and PD-1 and were the predominant fraction of T cells in the tumors and in the circulation. T cells in mice treated with REGN1979 showed decreased Tim-3 and PD-1 expression in the circulation accompanied by complete tumor regression. In further studies, REGN1979 (dosed at 0.4 mg/kg; 2x/week) was superior to rituximab therapy (dosed at 8 mg/kg; 5x/week) and comparable to the CD19xCD3 BiTE (dosed at 0.5 mg/kg; 5x/week) in suppressing established Raji tumors (200-400mm3). Pre-clinical studies in cynomolgus monkeys to assess activity of the bispecific antibody for depleting B cells in circulation and various lymphoid organs showed that a single injection of REGN1979 (0.1 mg/kg) was more potent at depleting CD20+ B cells in the mesenteric lymph nodes than a high dose of rituximab (30 mg/kg). In separate studies, REGN1979 was also found to have a long half-life (>14 days) in the circulation of monkeys following depletion of B cells. These studies show potent activity of a new class of fully human bispecific antibodies for treating tumors, and support clinical testing of REGN1979 in patients with CD20+ cancers.
Varghese:Regeneron Pharmaceuticals: Employment, Equity Ownership. Menon:Regeneron Pharmaceuticals: Employment, Equity Ownership. Rodriguez:Regeneron Pharmaceuticals: Employment, Equity Ownership. Haber:Regeneron Pharmaceuticals: Employment, Equity Ownership. Olson:Regeneron Pharmaceuticals: Employment, Equity Ownership. Duramad:Regeneron Pharmaceuticals: Employment, Equity Ownership. Oyejide:Regeneron Pharmaceuticals: Employment, Equity Ownership. Smith:Regeneron Pharmaceuticals: Employment, Equity Ownership. Thurston:Regeneron Pharmaceuticals: Employment, Equity Ownership. Kirshner:Regeneron Pharmaceuticals: Employment, Equity Ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal