Abstract
A regimen comprising of fludarabine, cytarabine, G-CSF and gemtuzumab ozogamicin (FLAG-GO) has been our frontline treatment regimen for patients with core binding factor acute myelogenous leukemia (CBF-AML) and among 50 patients with median follow up of over 3 years, this regimen resulted in overall survival (OS) and relapse free survival (RFS) of 78% and 85% respectively. This is clearly better than our historical data with idarubicin and cytarabine based regimens (Borthakur et al. JCO. Vol 28, No 15 suppl; 2010:6552). After withdrawal of GO from the market, it has been substituted by low dose idarubicin at 6 mg/m2 on days 3 and 4 in induction and in one post remission cycle during cycles 3-6 (FLAG-Ida). So far 38 patients have been treated with FLAG-Ida (median follow up 1 year) with all patients achieving complete remission. The current report is part of the planned analysis to ensure that patient outcomes have not been compromised by the change in regimen.
Univariate and multivariate (MVA) Cox proportional hazards regression was used to identify association of the clinical variables with overall survival (OS), event free survival (EFS) and time to relapse (TTR). Event is defined as death from any cause or relapse. Treatment regimen (FLAG-GO or FLAG-Ida) were included as variables in the analysis. Apart from relevant clinical variables, reduction in fusion transcript ratio (in reference to ABL gene transcript compared to that at diagnosis) at time points 1 month (≥3 log reduction yes/no) and 3 month (≥3 log reduction yes/no) and presence of any mutation (RAS, KIT, FLT3 yes/no) were also added as variables. Stepwise backwards selection method was used to remove variables that did not remain significant in the multivariate model (p ≥ 0.15).
T(8;21) is the cytogenetic abnormality in 52% of all 88 patients. Median age of all patients is 51 (range, 19-78 years) and 48% are female. Median time to 3 log reduction in transcript ratio was 1 month (range, 1-22 months). Complete remission rate with or without platelet recovery has been 98% with 2 induction deaths.
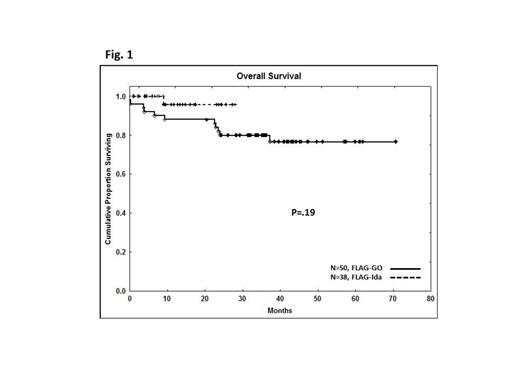
Kaplan-Meier analysis showed OS (Fig.1), EFS and TTR were not significantly different FLAG-GO and FLAG-Ida regimens. By MVA, OS, EFS and TTR are not different among these regimens (p > 0.5). Three log or more reduction in transcript ration at 1 month was associated with better OS (p=.03) and EFS (p=.008) (not TTR) in MVA. Presence of KIT, RAS or FLT3 gene mutation did not impact outcomes studied.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal