Abstract
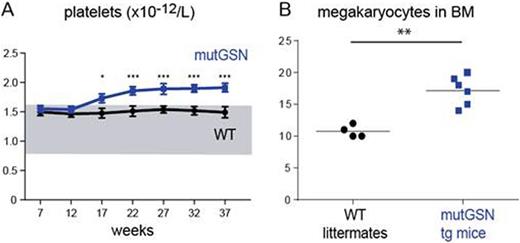
Hereditary thrombocytosis (HT) is a familial myeloproliferative disorder with clinical features resembling sporadic essential thrombocythemia. In some families germline mutations causing HT have been identified in the genes for thrombopoietin (THPO) and its receptor, MPL. However, in many HT families the disease-causing genetic lesion remains unknown. Here we studied a HT pedigree with 15 affected family members in 5 generations. Thrombocytosis is transmitted as an autosomal dominant trait with high penetrance (Figure 1). Genome-wide linkage analysis was performed on DNA from 21 family members using microsatellites and single nucleotide polymorphism arrays. A single co-segregating region with a LOD score of 4.3 was found on chromosome 9. All exons and splice junctions of genes within the co-segregating region were sequenced by classical DNA sequencing and by Next Generation Sequencing (Illumina). We found a candidate mutation in the gelsolin gene (GSN). This C/T transversion was not reported in any SNP database. Computational analysis predicts that the resulting Gly to Cys amino acid change will be damaging to the protein structure. Platelet biogenesis in vitro assays in DAMI cell line stably transfected with the mutant GSN showed that the candidate alteration increased the release of platelets-like particles. To study the in vivo role of the candidate mutation, we generated transgenic mice expressing the mutant GSN gene. These mice developed thrombocytosis and showed increased numbers of megakaryocytes in bone marrow (Figure 2). Thus, our genetic and functional data strongly suggest that GSN mutation is causing thrombocytosis in this family. The exact mechanism of how this newly identified genetic alteration leads to increased megakaryopoiesis needs further investigation.
Disclosures:
No relevant conflicts of interest to declare.
Author notes
*
Asterisk with author names denotes non-ASH members.
© 2013 by The American Society of Hematology
2013


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal