Abstract
The POEMS syndrome (polyradiculoneuropathy, organomegaly, multiple endocrinopathies, monoclonal protein, skin changes) is a rare disease associated with a plasma cell dyscrasia. Patients with disseminated POEMS can be treated with high-dose therapy and autologous stem cell transplantation (ASCT). While clinical improvement is nearly universal in these patients, the long-term outcomes after transplantation are unclear. We therefore assessed the long-term clinical outcomes of 59 POEMS patients treated with ASCT at our institution. With a median follow-up of 45 months, 14 patients have progressed with a progression-free survival of 98% and 75% at 1 and 5 years, respectively. Factors associated with progression have included an IgG-λ monoclonal component (hazard ratio [HR] 7.5; 95% confidence interval [CI], 2.3-28.3; P = .0008), fluorodeoxyglucose-avid lesions on baseline positron emission tomography (HR 6.4; 95% CI, 1.2-120; P = .03), lack of complete hematologic response (HR 5.4; 95% CI, 1.8-16.7; P = .003), and patients aged 50 years or younger at transplantation (HR 4.4; 95% CI, 1.3-20; P = .01). The most common progression events have been radiologic followed by rising VEGF. Symptomatic progression has been rare. Most patients could be salvaged with immunomodulatory drugs or radiation. The 5-year survival is 94%. Herein, we describe a system of monitoring response and progression among patients with POEMS after ASCT.
Introduction
The POEMS syndrome (osteosclerotic myeloma) is a paraneoplastic disorder secondary to a plasma cell dyscrasia. It affects multiple organ systems, as evident by the acronym POEMS representing polyradiculoneuropathy, organomegaly, multiple endocrinopathies, monoclonal protein, and a myriad of skin changes such as hypertrichosis, skin thickening, cherry angiomata, and hyperpigmentation. Other important clinical features that are not represented in the acronym but commonly seen in patients include sclerotic bone lesions, extravascular fluid overload resulting in pleural effusions and/or ascites, papilledema, thrombocytosis, plasmacytomas, clubbing, pulmonary hypertension, multicentric Castleman disease, and fatigue.1 The optimal treatment of POEMS syndrome involves eliminating the plasma cell clone, and this may include local therapy of a plasmacytoma with surgery or radiation therapy, or using systemic chemotherapy in patients with disseminated disease, that is, medullary disease, or multiple plasmacytomas. Patients with POEMS syndrome have a superior overall survival (OS) compared with that of multiple myeloma. Previously published data from the Mayo Clinic has shown that patients have a median OS of 13.8 years.2 There have been multiple reports of high-dose chemotherapy with peripheral blood stem cell transplantation in patients with POEMS, but most studies are small and long-term outcomes are not well delineated.3-13 Since 1999, we have expanded our experience with the use of autologous stem cell transplantation (ASCT) in patients with POEMS syndrome. In this article, we report the long-term outcomes of our cohort of POEMS patients treated with ASCT with an emphasis on identifying factors that were significantly associated with a risk of progression or relapse of POEMS syndrome after peripheral blood stem cell transplantation.
Methods
The study was approved by the Mayo Foundation Institutional Review Board, and data were collected in accordance with Minnesota state regulations. We queried our bone marrow transplantation database to identify all patients with POEMS syndrome who underwent high-dose therapy followed by ASCT at the Mayo Clinic (Rochester, MN). Between March 1999 and October 2011, a total of 145 new patients with POEMS syndrome were seen at the Mayo Clinic in Rochester. Seventy-nine (54%) received ASCT: 59 patients at our institution and 20 at home institutions. Disposition of the remaining patients included: a recommendation for ASCT that was not followed at home institution (n = 14); radiation for localized disease, that is, 1-3 plasmacytomas (n = 22); and other therapies because of perceived ASCT ineligibility, that is, no active disease at the time of evaluation, age > 70 years, coexisting solid cancers, and severe dysfunction of single or multiple organ systems (n = 30). The 59 patients receiving ASCT at the Mayo Clinic were considered our study population and are the subject of this analysis. At least 12 months of follow-up data were available in 56 patients (95%).
Transplantation details
Mobilization of stem cells was performed in accord of institutional standards during the time. The choice and dose of the conditioning regimen was based on the patient's clinical condition and the treating physician's judgment. Posttransplantation, a periengraftment syndrome, was defined based on the modified Spitzer criteria, as follows.
Major criteria.
A temperature of > 38.3°F without an identifiable infectious etiology, rash involving > 25% body surface area, and noncardiogenic pulmonary edema.
Minor criteria.
Bilirubin greater than twice normal, doubling of transaminases, weight gain ≥ 2.5% of baseline, or transient encephalopathy.
Periengraftment syndrome included the presence of all 3 major criteria or 1 major and 2 minor criteria. The modification used—the requirement for a neutrophil count of > 0.5 × 106/cubic mL around 96 hours of the criteria—was relaxed because of the temporal disconnect of neutrophil engraftment and periengraftment seen in POEMS syndrome.3
Definition and evaluation of response
Evaluations were performed as follows: physical examination, blood tests, and plasma VEGF levels were obtained on day 100 after ASCT, at 6 months, 1 year, and yearly subsequently. A BM examination was performed on day 100 and at year 1. Subsequent positron emission tomography (PET) scans were performed in patients with baseline PET abnormalities on day 100, year 1, and yearly. All of the above tests were also considered regardless of the timing if a patient developed new symptoms concerning for a relapse, based on the treating physician's judgment.
If patients did not meet criteria for a measurable parameter, they were considered inevaluable for that parameter and noted as such. Four types of response were assessed: hematologic, fluorodeoxyglucose (FDG) avidity on PET, VEGF, and clinical.
Hematologic responses.
Hematologic responses were modified from uniform response criteria for multiple myeloma14 and included complete response (CRH), negative BM, and negative immunofixation of the serum and urine; very good partial response (VGPRH), 90% reduction in M-protein or immunofixation positive only as long as M-protein was at least 0.5 g/dL at baseline; partial response (PRH), 50% reduction in M-protein or immunofixation positive as long as baseline M-protein was at least 1.0 g/dL; no response (NRH), no response.
PET responses.
PET responses included, complete radiologic response (CRR), which referred to initial FDG avidity on a baseline PET scan that disappears after ASCT; partial radiologic response (PRR), which referred to initial FDG avidity that was ≥ 50% improved after ASCT; and no radiologic response (NRR), which referred to all cases that had initial FDG avidity but did not meet criteria for CRR or PRR.
VEGF responses.
VEGF responses included CRV, normalized VEGF; PRV, VEGF improved by at least 50%, assuming baseline was ≥ 200 pg/mL15 ; and NRV, not meeting either CRV or PRV. Plasma VEGF levels were performed using plasma samples collected in EDTA tubes by the ELISA method. Testing was performed at the Quest Diagnostics Nichols Institute (San Juan Capistrano, CA). Normal values ranged between 31 and 86 pg/mL.
Clinical responses.
Clinical responses were based on objective improvements in any of the POEMS syndrome features, that is, improvement in peripheral neuropathy, extravascular fluid overload (ascites/effusions/edema), or pulmonary function tests,16 as clinical improvements (IC) or no clinical improvement (NIC).
Definition of progression/relapse
Progression was defined as follows: hematologic progression, increase in the M-component (serum or urine) by > 25% from the lowest value14 ; PET progression, definite increase in size or FDG avidity of existing plasmacytomas on PET scan; VEGF progression, persistent increase in plasma VEGF level > 200 pg/mL on at least 2 occasions; and clinical progression, new symptoms attributable to POEMS syndrome. Similarly, relapse was defined as: hematologic relapse, reappearance of serum/urine M-protein by electrophoresis/immunofixation14 ; radiographic (PET) relapse, new bone/soft tissue plasmacytomas; VEGF relapse, rising plasma VEGF level > 200 pg/mL on at least 2 occasions; and clinical relapse, new symptoms attributable to POEMS syndrome.
Statistical analysis
Overall survival was defined as the time from transplantation to death from any cause. Progression-free survival (PFS) was defined as the time from transplantation to the time of progression, relapse, or death from any cause. Using the Kaplan-Meier method, curves for PFS and OS were plotted. The following factors were assessed by univariate analysis using Cox modeling to assess their risk for progression after ASCT: age at transplantation, sex, race, POEMS features, subtype of Ig heavy chain, baseline PET scan, VEGF level at transplantation, achievement of CR. Data analysis was conducted using JMP version 8.0 statistical software (SAS Institute Inc). A P value < .05 was considered to be statistically significant.
Results
Baseline clinical and laboratory characteristics
Table 1 summarizes the presenting features at diagnosis of the 59 patients, 30 of whom have been reported in part previously.3 The median age at transplantation was 51 years (range, 20-70 years), with a male predominance at 64%. High-dose therapy was the first-line treatment in 25 patients (42%), while the remaining 34 patients had received prior chemotherapy and/or radiation treatment. One patient had a prior chemoembolization procedure for a large hepatic plasmacytoma. The median time to ASCT from the time of diagnosis of POEMS was 4.9 months (range, 2.4-260.9 months). The Eastern Cooperative Oncology Group (ECOG) performance score was 3 in 20% of patients.
Clinical and laboratory baseline characteristics of patients with POEMS
| . | All, N = 59 (%) . | No progression, N = 45 (%) . | Progression, N = 14 (%) . | P . |
|---|---|---|---|---|
| Median age at diagnosis, y (range) | 50 (20-70) | 51 (20-70) | 42 (36-59) | .02 |
| Median age at transplantation, y (range) | 51 (20-70) | 52 (20-70) | 46 (36-59) | .01 |
| Time to transplantation from diagnosis, mo (range) | 4.9 (2.4-261) | 5.2 (2.4-261) | 4.1 (3-122) | |
| Male sex | 38 (64) | 28 (62) | 10 (71) | |
| White race | 49 (83) | 36 (80) | 13 (93) | |
| ECOG PS 2/3 | 41 (70) | 34 (76) | 7 (50) | |
| Polyneuropathy | 57 (97) | 44 (98) | 13 (93) | |
| Organomegaly | 39 (66) | 32 (73) | 7 (50) | |
| Endocrinopathy | 53 (90) | 43 (96) | 10 (71) | |
| Monoclonal plasma cell disorder* | 58 (98) | 44 (98) | 14 (100) | |
| Skin changes | 54 (92) | 44 (98) | 10 (71) | |
| Extravascular fluid overload | 53 (90) | 43 (96) | 10 (71) | |
| Castleman disease | 11 (19) | 8 (18) | 3 (21) | |
| Plasma VEGF level at transplantation (normal 31-86 pg/mL) | 452 | 436 (N = 33) | 555 (N = 8) | |
| IQR | 232-788 | 206-788 | 369-794 | |
| Ig heavy chain | ||||
| IgA | 32 (54) | 28 (62) | 4 (29) | |
| IgG | 17 (29) | 9 (20) | 8 (57) | .0008 |
| IgM | 2 (3) | 2 (4) | 0 | |
| Biclonal (IgG + IgA) | 1 (2) | 1 (2) | 0 | |
| No heavy chain | 7 (12) | 5 (11) | 2 (14) | |
| Ig light chain | ||||
| Lambda | 56 (95) | 42 (93) | 14 (100) | |
| Kappa | 3 (5) | 3 (7) | 0 | |
| BM involvement by monoclonal plasma cells, % | ||||
| ≥ 10% | 11 (19) | 7 (16) | 4 (29) | |
| 5%-9% | 11 (19) | 10 (22) | 1 (7) | |
| < 5% | 17 (29) | 12 (26) | 5 (35) | |
| Polytypic plasma cell aggregates | 20 (34) | 16 (36) | 4 (29) | |
| Pretransplantation PET/CT scan with increased FDG uptake | 18 (31) | 10 (42) | 8 (80) | .03 |
| (Baseline PET/CT scan performed, N = 34) | (N = 24) | (N = 10) | ||
| Previous treatment | ||||
| None | 25 (42) | 20 (44) | 5 (36) | |
| Previous chemotherapy | 28 (47) | 20 (44) | 8 (57) | |
| Cyclophosphamide-based | 14 | 10 | 4 | |
| Melphalan-based | 2 | 0 | 2 | |
| IMID | 9 | 6 | 3 | |
| Others | 13 | 12 | 1 | |
| Previous radiotherapy | 10 (17) | 8 (18) | 2 (14) |
| . | All, N = 59 (%) . | No progression, N = 45 (%) . | Progression, N = 14 (%) . | P . |
|---|---|---|---|---|
| Median age at diagnosis, y (range) | 50 (20-70) | 51 (20-70) | 42 (36-59) | .02 |
| Median age at transplantation, y (range) | 51 (20-70) | 52 (20-70) | 46 (36-59) | .01 |
| Time to transplantation from diagnosis, mo (range) | 4.9 (2.4-261) | 5.2 (2.4-261) | 4.1 (3-122) | |
| Male sex | 38 (64) | 28 (62) | 10 (71) | |
| White race | 49 (83) | 36 (80) | 13 (93) | |
| ECOG PS 2/3 | 41 (70) | 34 (76) | 7 (50) | |
| Polyneuropathy | 57 (97) | 44 (98) | 13 (93) | |
| Organomegaly | 39 (66) | 32 (73) | 7 (50) | |
| Endocrinopathy | 53 (90) | 43 (96) | 10 (71) | |
| Monoclonal plasma cell disorder* | 58 (98) | 44 (98) | 14 (100) | |
| Skin changes | 54 (92) | 44 (98) | 10 (71) | |
| Extravascular fluid overload | 53 (90) | 43 (96) | 10 (71) | |
| Castleman disease | 11 (19) | 8 (18) | 3 (21) | |
| Plasma VEGF level at transplantation (normal 31-86 pg/mL) | 452 | 436 (N = 33) | 555 (N = 8) | |
| IQR | 232-788 | 206-788 | 369-794 | |
| Ig heavy chain | ||||
| IgA | 32 (54) | 28 (62) | 4 (29) | |
| IgG | 17 (29) | 9 (20) | 8 (57) | .0008 |
| IgM | 2 (3) | 2 (4) | 0 | |
| Biclonal (IgG + IgA) | 1 (2) | 1 (2) | 0 | |
| No heavy chain | 7 (12) | 5 (11) | 2 (14) | |
| Ig light chain | ||||
| Lambda | 56 (95) | 42 (93) | 14 (100) | |
| Kappa | 3 (5) | 3 (7) | 0 | |
| BM involvement by monoclonal plasma cells, % | ||||
| ≥ 10% | 11 (19) | 7 (16) | 4 (29) | |
| 5%-9% | 11 (19) | 10 (22) | 1 (7) | |
| < 5% | 17 (29) | 12 (26) | 5 (35) | |
| Polytypic plasma cell aggregates | 20 (34) | 16 (36) | 4 (29) | |
| Pretransplantation PET/CT scan with increased FDG uptake | 18 (31) | 10 (42) | 8 (80) | .03 |
| (Baseline PET/CT scan performed, N = 34) | (N = 24) | (N = 10) | ||
| Previous treatment | ||||
| None | 25 (42) | 20 (44) | 5 (36) | |
| Previous chemotherapy | 28 (47) | 20 (44) | 8 (57) | |
| Cyclophosphamide-based | 14 | 10 | 4 | |
| Melphalan-based | 2 | 0 | 2 | |
| IMID | 9 | 6 | 3 | |
| Others | 13 | 12 | 1 | |
| Previous radiotherapy | 10 (17) | 8 (18) | 2 (14) |
Statistically significant factors are shown with P values.
ECOG indicates Eastern Cooperative Oncology Group; PS, performance score; IQR, interquartile range; PET, positron emission tomography; FDG, fluorodeoxyglucose; IMID, immunomodulatory agents (thalidomide, lenalidomide); and others, rituximab, vincristine, adriamycin, azathioprine, mycophenolate mofetil, interferon, tamoxifen, infliximab.
M-protein in serum, urine, or plasmacytoma.
Transplantation details
Table 2 provides the details of the ASCT. Peripheral blood stem cells were collected using either cyclophosphamide with growth factors, growth factors alone, or growth factors with plerixafor as mobilizing agents. The conditioning chemotherapy was melphalan in 58 of the 59 patients: 41 patients received melphalan 200 mg/m2, and 17 received melphalan 140 mg/m2. Patients with pronounced lung function impairments as evidenced by a low diffusion capacity of the lung for carbon monoxide (DLCO) and/or an ECOG performance score of 3 received low-dose melphalan, according to the transplantation physician's judgment. One of the patients receiving melphalan 200 also received a bone-seeking radioisotope 153Sm-EDTMP (samarium-153 ethylene diamine tetramethylene diphosphonate). One patient with Castleman disease and generalized marked lymphadenopathy was conditioned using BEAM (carmustine, etoposide, cytarabine, melphalan). The median stem cell dose was 4.43 × 106 CD34 cells/kg. The median duration of hospitalization was 9 days (range, 0-175 days).
Transplantation details of patients with POEMS syndrome who underwent ASCT
| . | Number, N = 59 (%) . | No progression, N = 45 (%) . | Progression, N = 14 (%) . |
|---|---|---|---|
| Stem cell mobilization | |||
| Cyclophosphamide/growth factor | 6 (10) | 4 (9) | 2 (14) |
| Growth factor alone | 50 (85) | 38 (84) | 12 (86) |
| Growth factor/plerixafor | 3 (5) | 3 (7) | 0 |
| Conditioning regimen | |||
| Melphalan, 200 mg/m2 | 41 (69) | 29 (64) | 12* (86) |
| Melphalan, 140 mg/m2 | 17 (29) | 15 (33) | 2 (14) |
| BEAM | 1 (2) | 1 (2) | 0 |
| Median stem cell dose, ×106 CD34 cells/kg (IQR) | 4.4 (3.7-5.3) | 4.4 (3.7-5.2) | 4.9 (3.3-5.9) |
| Median days in hospital (range) | 9 (0-175) | 9 (0-175) | 3.5 (0-20) |
| Periengraftment syndrome | 22 (37) | 19 (42) | 3 (21) |
| Engraftment days (IQR) | |||
| Neutrophils | 15 (11-35) | 15 (14-17) | 16 (15-19) |
| Platelets > 20 000 | 13 (8-26) | 14 (12-18) | 11 (11-14) |
| Platelets > 50 000 | 15 (11-28) | 16 (14-19) | 15 (13-17) |
| . | Number, N = 59 (%) . | No progression, N = 45 (%) . | Progression, N = 14 (%) . |
|---|---|---|---|
| Stem cell mobilization | |||
| Cyclophosphamide/growth factor | 6 (10) | 4 (9) | 2 (14) |
| Growth factor alone | 50 (85) | 38 (84) | 12 (86) |
| Growth factor/plerixafor | 3 (5) | 3 (7) | 0 |
| Conditioning regimen | |||
| Melphalan, 200 mg/m2 | 41 (69) | 29 (64) | 12* (86) |
| Melphalan, 140 mg/m2 | 17 (29) | 15 (33) | 2 (14) |
| BEAM | 1 (2) | 1 (2) | 0 |
| Median stem cell dose, ×106 CD34 cells/kg (IQR) | 4.4 (3.7-5.3) | 4.4 (3.7-5.2) | 4.9 (3.3-5.9) |
| Median days in hospital (range) | 9 (0-175) | 9 (0-175) | 3.5 (0-20) |
| Periengraftment syndrome | 22 (37) | 19 (42) | 3 (21) |
| Engraftment days (IQR) | |||
| Neutrophils | 15 (11-35) | 15 (14-17) | 16 (15-19) |
| Platelets > 20 000 | 13 (8-26) | 14 (12-18) | 11 (11-14) |
| Platelets > 50 000 | 15 (11-28) | 16 (14-19) | 15 (13-17) |
POEMS represents polyradiculoneuropathy, organomegaly, multiple endocrinopathies, monoclonal protein, and a myriad of skin changes such as hypertrichosis, skin thickening, cherry angiomata, and hyperpigmentation; ASCT, autologous stem cell transplantation; 153Sm-EDTMP, samarium-153 ethylene diamine tetramethylene diphosphonate; BEAM, carmustine, etoposide, cytarabine, melphalan; and IQR, interquartile range.
One patient got 153Sm-EDTMP with melphalan.
A periengraftment syndrome was recognized in 22 patients (37%) and treated with a high dose of corticosteroids. Over time, the median number of hospitalization days was noted to have shortened, dropping from 11 days (range, 0-175, n = 33) pre-2008 to 5 days (range, 0-67, n = 26) since 2008. There was no difference in the incidence of periengraftment syndrome based on previous treatment with cyclophosphamide either as prior chemotherapy or as a part of chemomobilization.
Response
The best initial responses were as follows. Hematologic response: CRH 34 (57%), VGPRH 2 (3%), PRH 11 (19%), NRH3 (5%), inevaluable 8 (14%), not available 1 (2%). PET response: CRR 10 (17%), PRR 4 (7%), NRR 5 (8%), inevaluable 17 (29%), no PET scan at baseline 23 (39%). VEGF response: CRV28 (48%), PRV 3 (5%), NRV 4 (6%), no baseline VEGF 24 (41%). Clinical response: IC 54 (92%), NIC 0 (3%), not available 4 (7%).
The fastest responses were seen in VEGF and clinical status (improvements in extravascular fluid overload); these were seen as early as day 100 after transplantation. Most hematologic and radiologic responses were seen by 1 year, except 4 patients in whom responses were delayed to as long as 2 years (3 patients) and 3 years (1 patient). Maximal improvements in neuropathic symptoms were often not seen until 3 years, but subjective improvements were reported as early as 3 months after transplantation.
Because all patients could not be evaluated by each of our proposed response criteria, aggregate responses were also measured based on the current international myeloma working group criteria.14 Based on these criteria, the best initial responses seen were the following: CR in 38 (64%), VGPR in 6 (10%), PR in 7 (12%), less than PR in 4 (7%), and 4 patients were not evaluable.
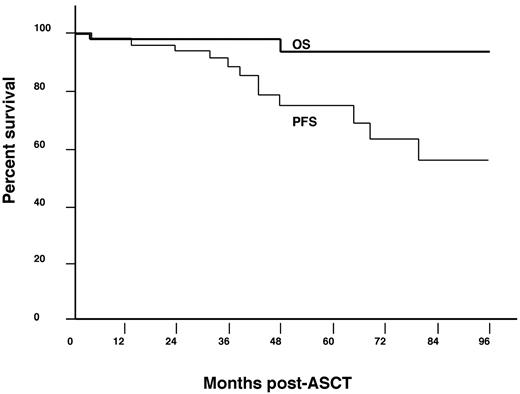
Survival
The median duration of follow-up from the time of the ASCT was 45 months (range, 0-149). The 5-year OS rate was 94%. There were 3 deaths, which included 1 early death and 2 deaths that occurred late (patients 1 and 9 in Figure 1). Patient 9 died because of relapsed POEMS syndrome 4 years after his ASCT. After initially attaining a CR by day 100 after ASCT, he was noted to have an elevated plasma VEGF level 39 months after ASCT. He was observed as there were neither new clinical symptoms nor evidence of active disease by immunofixation (monoclonal protein) or on imaging with PET scan. Four months later, he presented with florid POEMS symptoms (worsening neuropathy, extravascular fluid gain, new endocrinopathies), 10% monoclonal plasma cells in the BM, and high VEGF, and was treated with 2 cycles of lenalidomide with dexamethasone. Because of continued clinical worsening, bevacizumab with cyclophosphamide/prednisolone was also given, but he continued to deteriorate and succumbed to his illness 5 months after documented relapse. The third death (patient 1) was not because of POEMS syndrome. Patient 1 was our first transplanted POEMS patient.5 He was conditioned with 153Sm-EDTMP and full-dose melphalan which resulted in both complete hematologic response and marked clinical improvement. Five years after his transplantation, he presented with low back pain, and was found to have a new lytic lesion in the sacrum with a recurrence of the monoclonal protein, but no clinical worsening. He was treated with radiation and then received thalidomide for 3 years. Nine years after his ASCT for POEMS syndrome, he was diagnosed with anaplastic large-cell lymphoma in his parotid gland. For his lymphoma, he was treated with chemotherapy, ASCT, additional radiation and chemotherapy, but died from progressive lymphoma 13 years after his ASCT for POEMS syndrome. He had no evidence of POEMS syndrome at the time of his demise.
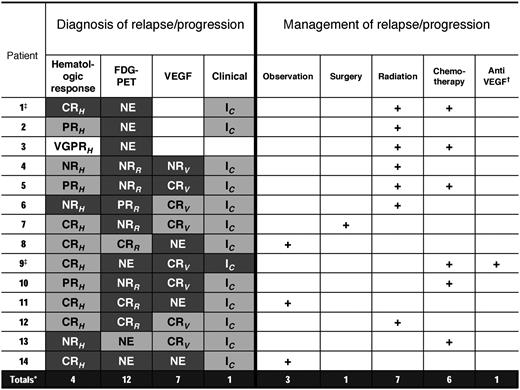
Best response and progression of POEMS after transplantation. Letters in boxes for columns 2-5 refer to initial best responses after ASCT. CRH indicates complete hematologic response; VGPRH, very good partial hematologic response; PRH, partial hematologic response; NRH, no hematologic response; CRR, complete radiologic response; PRR, partial radiologic response; NRR, no radiologic response; CRV, complete VEGF response; PRV, partial VEGF response; NRV, no VEGF response; IC, clinical improvement; NIC, no clinical improvement; and NE, not evaluable, that is, a baseline test was not available. Shading gradient in columns 2-5 refers to progression status. Dark gray shading indicates progression; light gray shading, no progression; white box, missing data; and + (in columns 6-10) refers to treatment given. *Totals refers to number of progressions; †, bevacizumab; and ‡, deceased.
Best response and progression of POEMS after transplantation. Letters in boxes for columns 2-5 refer to initial best responses after ASCT. CRH indicates complete hematologic response; VGPRH, very good partial hematologic response; PRH, partial hematologic response; NRH, no hematologic response; CRR, complete radiologic response; PRR, partial radiologic response; NRR, no radiologic response; CRV, complete VEGF response; PRV, partial VEGF response; NRV, no VEGF response; IC, clinical improvement; NIC, no clinical improvement; and NE, not evaluable, that is, a baseline test was not available. Shading gradient in columns 2-5 refers to progression status. Dark gray shading indicates progression; light gray shading, no progression; white box, missing data; and + (in columns 6-10) refers to treatment given. *Totals refers to number of progressions; †, bevacizumab; and ‡, deceased.
Relapse/progression of POEMS after high-dose therapy
Fourteen patients have progressed to date, including the 2 patients described above (Figure 1). The 5-year PFS is 75% (Figure 2). Among the progressing patients, the median time to progression was 43 months (range, 5-84 months). Factors associated with progression on univariable analysis included: (1) an IgG-λ monoclonal protein (hazard ratio [HR] 7.5; 95% confidence interval [CI], 2.3-28.3; P = .0008); (2) a baseline PET scan showing FDG-avid bony lesions (HR 6.41; 95% CI, 2-120; P = .03); (3) lack of complete hematologic response (HR 5.4; 95% CI, 1.8-16.9; P = .003); and (4) age ≤ 50 years at transplantation (HR 4.4; 95% CI, 1.3-20; P = .01; Table 1).
In addition, patients who did not have a PET CR (CRR) were also at a higher risk of progression (HR 6.4; 95% CI, 1-123.7; P = .05). With regards to patients aged 50 years or younger, these patients had a higher incidence of lytic bone lesions (P .007), IgG-λ M-component (P = .04), and extravascular fluid overload at baseline (P = .05).
On attempting to tease out differences between patients with an IgG heavy-chain isotype from the more common IgA heavy-chain isotype, we found that the former group tended to be younger, with a median age at transplantation of 44 years versus 51 years (P .03), less likely to have organomegaly, 38% versus 80% (P .004), and with little BM involvement by monoclonal plasma cells, with a median of 0% versus 5% (P .03). Six (67%) of 9 IgG-λ patients had PET-avid lesions at baseline as opposed to 7 (44%) of 16 IgA-λ. Although this did not meet statistical significance, the analysis was restricted by the small number of patients with baseline PET scans. No differences were seen in these 2 groups with other POEMS features, or baseline plasma VEGF levels.
The details of the 14 patients who progressed, with their best initial responses, nature of progression, and the subsequent management are shown in Figure 1. The majority of patients had either a radiographic or a biochemical progression or relapse. Only 1 patient has had a symptomatic progression. Another patient had symptoms of back pain secondary to a sacral lytic lesion, without any clinical symptoms of recurrent POEMS syndrome. As shown in Figure 1, many of the patients who progressed never achieved a CRH (7 of 14) or CRR (5 of 8) after ASCT. It is worth noting that all of these patients had an improvement in their peripheral neuropathy, although 2 patients had evidence of persistent extravascular fluid overload at 1 year. There was a significant relationship between VEGF and relapse and PET relapse. Of the 12 patients with radiographic relapse, only 3 had normal VEGF, and of the 29 patients without documented PET relapse, only 2 had elevated VEGF (P < .001).
All but 1 of the patients (patient 9; Figure 1) has done well despite progression. Three patients with radiographic and/or biochemical progression are currently being observed without any intervention. One patient was treated with surgical resection of a residual hepatic plasmacytoma with resultant CR (patient 7). The most common interventions have been radiation (7 patients) and chemotherapy or immunomodulatory therapy (6 patients). The most commonly used drug has been lenalidomide (4 patients). Other treatments used have been thalidomide (1) and bortezomib (1) patient. Aggregate response to second-line treatment has been CR in 4 patients, VGPR in 2, and PR in 3. Two patients had no response to treatment, and 3 patients are being observed without any treatment.
Secondary myelodysplastic syndrome
Two patients have developed therapy related myelodysplastic syndrome (MDS) at 3 and 7 years after ASCT. One patient had Castleman disease with POEMS syndrome and had received previous chemotherapy with lenalidomide, and then with combination of rituximab, cyclophosphamide, and mitoxantrone followed by conditioning with melphalan 200 mg/m2 and transplantation. She was found to be anemic without leukopenia or thrombocytopenia ∼ 3 years after the ASCT. At this time, she also had a rise in her M-protein. BM examination showed megakaryocytic atypia, and cytogenetics revealed a new 20q− in 15 of 20 metaphases. She is currently being observed without therapy for either disorder. The second patient with therapy-related MDS had been treated with intravenous gammaglobulin, steroids, and azathioprine for presumed chronic inflammatory demyelinating polyneuropathy for 3 years before a diagnosis of POEMS was made. She then received thalidomide with prednisone for 19 months. For her ASCT, she was conditioned using melphalan 140 mg/m2, and had a very complicated posttransplantation course including periengraftment syndrome requiring 3 months of mechanical ventilation, 5 months of hemodialysis, and possible mucormycosis. Despite this complicated course, she achieved a CR and showed a good response in clinical features, until the development of a bicytopenia (neutropenia and thrombocytopenia) 7 years later. BM evaluation revealed high-grade myelodysplasia with 15% blasts, and a complex karyotype with 5q−, monosomy 7, and tetraploidy. At the time of this analysis, she was being considered for 5-azacytidine treatment.
Discussion
High-dose chemotherapy with autologous peripheral blood stem cell transplantation has been established as a treatment modality in POEMS syndrome with disseminated disease.17 In this article, we have analyzed the long-term clinical outcomes in a cohort of POEMS patients who received transplants. Historically, patients with POEMS syndrome have been reported to have a median OS of > 13 years,2 even before the era of ASCT. This extremely good OS statistic in combination with the rarity of the disorder makes alternate endpoints all the more important. For the first time, a large enough treatment cohort of patients with POEMS syndrome undergoing ASCT has been amassed to clarify not only response rates, but also PFS rates and salvage strategies after ASCT. The results are encouraging in that only 25% of patients have progressed 5 years after ASCT. In addition, the 5-year OS rate of 94% was not surprising based on prior data,7-9 but it was very encouraging as was patients' ability to be salvaged with radiation or lenalidomide. The failure of bevacizumab was not unprecedented18,19 nor was the success of lenalidomide.20,21
Because response and progression in this syndrome is so multifaceted, we parsed response and progression into categories including: (1) hematologic, (2) radiologic, (3) VEGF, and (4) clinical (symptomatic). These categories were in part based on works of others,22-24 but mostly were constructs based on prior experience.16 Similar to patients with Ig light-chain amyloidosis, POEMS syndrome is a disease of low tumor burden, and much of patients' clinical phenotype is characterized by tissue damage remote from the site of the clone. Factoring objective organ responses (eg, neuropathy, extravascular fluid gain, and pulmonary function) into the measured responses would also appear to be valuable in defining a response to therapy.16
The conundrum of persistent M-spike posttherapy was also evident in this series, illustrating the limitations of applying myeloma response criteria to this patient population. We and others have shown that even patients with no apparent hematologic response by uniform myeloma response criteria can have dramatic and durable clinical responses.3 This observation continues to be valid even in this larger series of patients, but our new data also suggest that the patients who do not achieve CR are the same patients who are at higher risk for VEGF and radiologic progression, but not symptomatic clinical progression, at least so far. Surprisingly, the 1 patient who had progression of the symptoms of POEMS syndrome—that is, worsening neuropathic symptoms, anasarca, and pulmonary function—had achieved hematologic CR after ASCT.
VEGF levels have been shown to correlate well—but not perfectly—with clinical response, and this held true in this series. Rising VEGF predicted less well for symptomatic clinical progression and rising serum M-protein, but quite well for radiographic progression as measured by FDG avidity on PET scan. Radiographic responses are more accessible in patients who have FDG-avid lesions.
Although relapses of POEMS syndrome after ASCT have been described as individual cases,25,26 within the present study we were able to provide a more robust denominator to calculate rates progression. Armed with putative response tools, we were able to characterize various types of progression in 25% of patients by 5 years. Among the patients who progressed, the most common progressions were radiographic, that is, new or growing bony lesions defined on a PET scan followed by VEGF and hematologic progression. Fortunately, clinical deterioration has been a rare event, with only one of our patients having a florid POEMS relapse. An intriguing and novel observation in this series was that the 29% of patients with POEMS having an IgG heavy-chain isotype were statistically more likely to progress. Previously, our group had noted that POEMS patients with an IgA heavy-chain isotype were more likely to have BM involvement than those patients with an IgG heavy-chain isotype who tended to have more of a multiple or solitary plasmacytoma-type picture.27 In the 14 patients who progressed in our study, occurrence of new plasmacytomas or increasing activity in known lesions (N = 12) comprised an important feature of progression. Consistent with our prior report, it appears that patients with IgG heavy-chain isotype have a higher incidence of larger, FDG-avid plasmacytomas than those patients with an IgA isotype. The IgA patients appear to have more of a diffuse—albeit low level—medullary involvement by the plasma cell clone. Additional differences between the IgA and IgG heavy-chain isotypes we found in our cohort were a younger age (more likely with IgG), organomegaly (more likely with IgA).
Patients who had an abnormal baseline PET/CT scan with intense uptake or multiple FDG-avid lesions before high-dose therapy were also more likely to require additional therapy after transplantation. Thus, we speculate that high-dose chemotherapy alone may not be sufficient for patients with larger FDG-avid plasmacytomas and adjunctive treatment with radiation to dominant “hot” lesions on PET/CT scan or immunomodulatory drugs may be necessary. Further studies will be required to substantiate this concept.
We also observed that younger patients were at a higher risk of progression. On univariate analysis, age ≤ 50 years at the time of transplantation was significantly associated with a risk of relapse. This finding is difficult to explain. It is possible that this is a biased finding given the small sample size; however, we did find that younger patients were also more likely to have IgG-λ M-component, more lytic bony lesions, as well as more extravascular fluid overload, and thus may suggest a more aggressive biology of POEMS.
Management strategies for patients who progressed have been successful in most instances. None of the patients in our series who progressed were treated for an elevated VEGF, unless they had accompanying clinical symptoms, a rising monoclonal protein, or PET changes. However, it is worth noting that the only patient in our cohort with a florid POEMS relapse (patient 9; Figure 1) did present with an elevated VEGF months before clinical relapse. At the current time, however, we do not recommend initiating treatment in asymptomatic patients based only on VEGF elevation. We have occasionally seen a mildly elevated VEGF that returns to normal on repeat testing. For asymptomatic patients with PET progression, as shown in Figure 1, we have treated 4 patients (nos. 2, 3, 7, and 12) on the basis of PET progression alone. Typically, these patients tend to have 1-2 dominant lesions on PET imaging that are amenable to radiation therapy (or surgery in patient 7 who had a persistent hepatic plasmacytoma).
In terms of short-term morbidity, a periengraftment syndrome was seen in 22 (37%) patients. Previously, we have reported that patients with POEMS syndrome have a high incidence of periengraftment syndrome, albeit at a different temporal point in regards to neutrophil engraftment, that is, symptoms of engraftment syndrome have been noted to predate time of neutrophil engraftment by as long as 1 week.3 Since 2008, these patients have been watched carefully for the early signs of the periengraftment syndrome (fever, weight gain, pulmonary infiltrates, skin rash, and diarrhea) and have been treated with short bursts of high doses of corticosteroids when periengraftment syndrome was suspected. This has resulted in a shorter duration of hospitalization of patients as seen before and after 2008. It has been suggested both by us and others that prior cyclophosphamide use may decrease the incidence of periengraftment syndrome. We were not able to validate this finding in this larger group of our patients, but notably our practice has shifted away from cyclophosphamide mobilization diminishing our ability to address the question adequately.
We have seen little long-term morbidity in this series of patients. Only 2 cases of therapy-related MDS have been seen. This is lower than the risk of therapy-related MDS/acute leukemia in a series of myeloma patients who received high-dose melphalan therapy (12.2%).28 The majority of our patients were treatment naive, which may have influenced the low incidence of MDS. A longer follow-up will be important in detecting additional cases. It is also interesting to have encountered a patient with a second hematologic malignancy, namely a non-Hodgkin lymphoma (NHL) after the initial diagnosis of POEMS syndrome. Incidentally, this patient received 153Sm-EDTMP for BM ablation before his autologous transplantation. Rare cases of NHL have been described in POEMS.29,30 In our cohort of 59 patients, we have seen another patient with follicular lymphoma before the diagnosis of POEMS syndrome.
Lastly, because patients with POEMS syndrome are known to have long OS, it will be of interest to follow our cohort over a longer time period to determine whether patients who received transplants have improved survival over patients treated with alternate therapy. If an improvement is found in patients who received transplants, one would also need to account for the possibility of selection bias in that younger and fitter patients may be selected to undergo a transplantation over alternate therapy.
Conclusions
We show the long-term outcomes of patients with POEMS syndrome treated with high-dose therapy and ASCT. Patients can progress/relapse after transplantation, and therefore should be followed periodically. Risk factors for progression identified in our series included age of 50 years or younger, a baseline IgG-λ monoclonal protein, FDG-avid lesions on PET scan at baseline, and lack of hematologic CR post-ASCT. The median survival has not been reached, and longer follow-up of our cohort will be needed to determine whether patients who received transplants have a better OS compared with historical controls and patients treated contemporaneously without transplantation. A separate system to define and monitor disease response for POEMS is warranted, and our suggested method of parsing response into 4 major groups of response will need to be validated.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
The authors thank Kate Johanns for maintaining the Dysproteinemia database, and their colleagues in the Dysproteinemia and Bone Marrow Transplant groups for their excellent care in the treatment of these patients.
Authorship
Contribution: A. D'Souza and A. Dispenzieri designed the research, collected and analyzed the data, and wrote the manuscript; M. Lacy, M.G., S.K., F.B., S.H., D.D., S.Z., R.K. S.A., D.I., P.J., I.M., L.P., M. Litzow, D.G., and W.H. assisted in data collection and manuscript preparation; and all authors approved the final draft of the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Angela Dispenzieri, MD, Mayo Clinic, Rochester, MN 55905; e-mail: dispenzieri.angela@mayo.edu.