Response
Our recent paper in Blood provoked some discussions.1 We fully agree with Seddiki et al who suggest further studies on miRNA in different, isolated lymphocyte subpopulations.2 In fact, we intend to analyze miRNA signatures within Treg cell subsets.
However, we do not agree with Witwer and Clements who seem unsettled by our findings that dispute previous results.3
The analysis of complex data, especially in a relatively new field and in a pathology where little proven fact is available, could allow for different interpretations, but a correct methodologic approach is crucial. Some technical points were raised, the first regarding the quality of RNA. We extracted total RNA using the mirVana miRNA Isolation Kit, designed for the purification of miRNAs, strictly following the recommended procedures. The purity of RNA was assessed from the ratio of A260/A280 and A260/A230 (both above 1.80 in all samples) and its quality was evaluated on 1% denaturing agarose gel, stained with ethidium bromide, known to be a standard correct procedure.
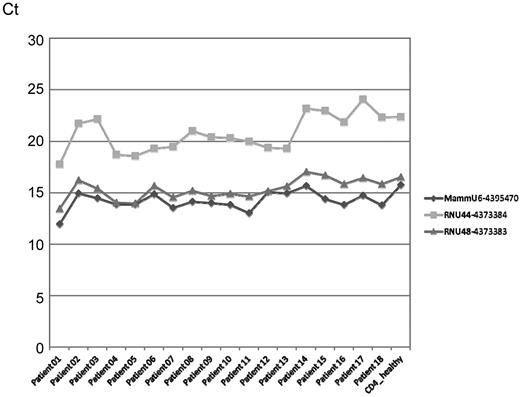
The stability of the endogenous control MammU6 was also questioned. The Taqman microRNA Low Density Array includes the following endogenous controls: MammU6–4395470, RNU44–4373384, and RNU48–4373383. The mammalian U6 assay was selected as a reference for data normalization since it was present in 4 wells, different from all other targets, and its mean Ct value was used for normalization. The variation of MammU6 among different samples was observed as being acceptable also by other authors.4
Considering the 3 endogenous controls it is evident that MammU6 (Figure 1) showed overall the lowest Ct values with a behavior similar to RNU48–4373383, while RNU44–4373384 had the highest Ct values and also the largest range of variation. Therefore MammU6 was the most appropriate reference assay for the set of data studied.
Endogenous control profile across samples. The Mammalian U6 assay was selected for data normalization. Endogenous controls included MammU6-4395470, RNU44-4373384, RNU48-4373383.
Endogenous control profile across samples. The Mammalian U6 assay was selected for data normalization. Endogenous controls included MammU6-4395470, RNU44-4373384, RNU48-4373383.
The “healthy control” is not inadequate, because: (1) RNA was extracted from a pool of 6 healthy donors with a similar CD4+ T count and a documented history of uninfection with HIV, HTLVs, HCV or HBV (confirmed also 6 months after enrollment); and (2) the analysis on TaqMan Low Density Array was repeated twice starting from several total RNA extractions. The Ct values used for data normalization, reported in GEO file, were the mean Ct obtained from these analyses.
The proposed octapartite exposure signature was also criticized. We considered that the 5 members of this signature matched our inclusion criteria, as other authors have.5 Undetermined raw Ct values were set to 40, so these 5 miRNAs were down-regulated in 76.5%-94.1% of the patients classes.
The unsupervised hierarchical clustering suggested that miRNAs could act as critical regulators of the complex interaction between HIV-1 and the immune system. Using a different approach for data analysis, Witwer and Clements highlighted multiple naive-to-ES differences. They cannot establish that their approach is the only one valid as there is no proof for such a statement.
Concerning subjects defined as MEU (multiple-HIV exposed uninfected), the definition was not a “conjecture” at all. As clearly written, the enrolled MEU subjects had had unprotected penetrative sexual intercourse at least twice a week for at least 2 years with an HIV-1+ partner. The seronegativity of MEU subjects and seropositivity of their partners was monitored before and after enrollment by p24 antigen ELISA test, plasma RNA detection and viral DNA load on lymphocytes. The crucial point is not whether the HIV seropositive partner was in therapy or how the therapy worked, but if each partner had the virus. This critique surprised us, as our group has a solid scientific reputation and a established clinical expertise in the field of MEU.6,–8
Authorship
All authors listed on the paper Bignami et al1 have reviewed and approved the response.
Conflict of interest disclosure: The authors declare no competing financial interests.
Correspondence: Claudio Casoli, GEMIB laboratory, Vicolo delle Asse 1 Parma I-43121, Italy; e-mail: claudio.casoli@gemiblab.com.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal