Abstract
Chronic GVHD is one of the most severe complications of allogeneic HSCT. The sclerotic skin manifestations of cGVHD (ScGVHD) result from inflammation and fibrosis of the dermis, subcutaneous tissue, or fascia, leading to significant functional disability. Risk factors and clinical markers associated with ScGVHD remain largely unexamined. By using a single-visit, cross-sectional design, we evaluated 206 patients with cGVHD at the National Institutes of Health. Most patients manifested severe (ie, 63% National Institutes of Health score “severe”), refractory disease (median treatments = 4). ScGVHD was detected in 109 (52.9%) patients. ScGVHD was associated with greater platelet count (P < .001) and C3 (P < .001), and decreased forced vital capacity (P = .013). Total body irradiation (TBI) was associated with development of ScGVHD (P = .002). TBI administered in reduced-intensity conditioning was most strongly associated with ScGVHD (14/15 patients, P < .0001). Patients with ScGVHD had significant impairments of joint range of motion and grip strength (P < .001). Greater body surface area involvement was associated with poorer survival (P = .015). We conclude that TBI, particularly in reduced-intensity regimens, may be an important risk factor for ScGVHD. Widespread skin involvement is associated with significant functional impairment, distressing symptoms, and diminished survival. This trial is registered at http://www.clinicaltrials.gov as NCT00331968.
Introduction
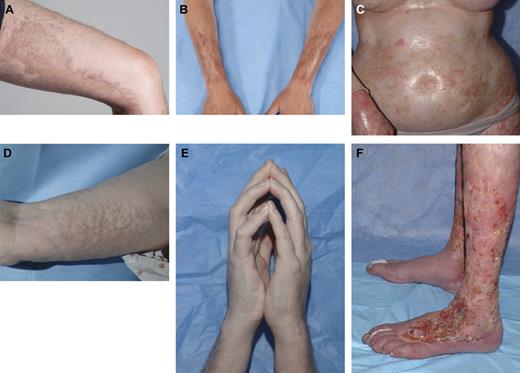
Chronic GVHD (cGVHD) is one of the most severe complications of allogeneic HSCT and commonly affects the skin.1 Sclerotic-type chronic GVHD (ScGVHD) of the skin encompasses several cutaneous presentations characterized by inflammation and progressive fibrosis of the dermis and subcutaneous tissues, resembling morphea, systemic sclerosis, or eosinophilic fasciitis (Figure 1) and may or may not occur in the setting of concurrent overlying epidermal GVHD. ScGVHD is generally a late manifestation of cGVHD, with a mean onset > 1 year after transplantation.2 The precise incidence of ScGVHD is not known but may occur in ∼ 13% of patients who develop cGVHD.3,4 Although ScGVHD is not an acute life-threatening manifestation, widespread involvement may lead to significant functional disability and morbidity.1 Skin ulceration and poor wound healing associated with skin fibrosis increases the risk of infection.5 ScGVHD responds poorly to topical interventions and often is recalcitrant to systemic therapy.4,6,7
The spectrum of cutaneous findings in ScGVHD. Localized bound-down thickened hyperpigmented plaques on the thigh (A) and forearms (B) resemble morphea/localized scleroderma. Widespread shiny indurated skin on the torso resembles generalized systemic sclerosis and may lead to restricted chest wall expansion (C). Subcutaneous and fascial fibrosis results in an irregular, rippled appearance to the skin, resembling eosinophilic fasciitis (D) and may result in joint contractures, including the prayer sign (E). Skin breakdown and poor wound healing is a complication of long-standing ScGVHD, particularly of the lower extremities, and may lead to increased risk of systemic infection (F).
The spectrum of cutaneous findings in ScGVHD. Localized bound-down thickened hyperpigmented plaques on the thigh (A) and forearms (B) resemble morphea/localized scleroderma. Widespread shiny indurated skin on the torso resembles generalized systemic sclerosis and may lead to restricted chest wall expansion (C). Subcutaneous and fascial fibrosis results in an irregular, rippled appearance to the skin, resembling eosinophilic fasciitis (D) and may result in joint contractures, including the prayer sign (E). Skin breakdown and poor wound healing is a complication of long-standing ScGVHD, particularly of the lower extremities, and may lead to increased risk of systemic infection (F).
Despite its major impact on long-term morbidity, the risk factors, clinical markers, and mechanism of sclerotic skin involvement remain largely unknown. CD3 T-cell dose in the graft, eosinophilia, positive antinuclear antibodies (ANAs), and antecedent nonsclerotic cGVHD skin involvement have been proposed as markers of ScGVHD4 ; however, previous series included only small samples of patients and retrospective study designs, and registry and cancer center data fail to distinguish between sclerotic and nonsclerotic cutaneous manifestations of cGVHD.2-4,8-11
The National Institutes of Health (NIH) Consensus Project recently proposed clear definitions of the sclerotic features of cGVHD,12 but it will take many years to prospectively collect a significant number of patients with this rare cGVHD subset. The NIH cGVHD Natural History cohort comprises a unique referral population of cGVHD patients with significant (63% NIH global score of “severe”) refractory disease (median previous therapies = 4) who undergo a comprehensive prospective multisystem evaluation. Many enrolled patients manifest disease that is refractory to conventional management, including ScGVHD, and as a result, the incidence of skin sclerosis in this cohort (53%) is much greater than described in other reviews (13.2%-14%).3,4 This population, therefore, provides a unique opportunity to comprehensively evaluate historical, clinical, laboratory, and serologic markers that may be associated with ScGVHD.
Methods
Subjects
A total of 217 consecutive adult and pediatric patients with a history of cGVHD, according to the definition of the NIH Consensus Group Criteria12 involving 1 or more organ systems after allogeneic HSCT, were referred to and evaluated by an interdisciplinary team at the NIH Clinical Center from 2004 to 2010 via a cross-sectional protocol in which the authors studied the natural history of cGVHD (http://www.clinicaltrials.gov identifier: NCT00331968). This research was approved by the Institutional Review Board of the National Cancer Institute, and all human subjects provided written informed consent in accordance with the Declaration of Helsinki.
All evaluations were collected during a 1-week single visit to the NIH by the use of predefined subspecialist organ-specific evaluations forms by a team that included experts in dermatology, ophthalmology, dentistry, rehabilitation medicine, gynecology, nursing, pain and palliative care, and transplantation medicine. Patients determined to have late acute GVHD or an inconclusive diagnosis of cGVHD as determined by the interdisciplinary panel were excluded from analysis. Skin involvement was assessed by comprehensive dermatologic examination by a dermatologist with expertise in cGVHD (E.W.C), and percent body surface area (BSA) skin involvement was documented. Clinical evidence of sclerotic-type cGVHD was determined by the presence of skin thickening, rippling, or nodularity of subcutaneous tissues on deep palpation, and range-of-motion (ROM) limitation. BSA involvement was recorded for the presence of epidermal cGVHD (nonsclerotic), or dermal and subcutaneous tissue sclerosis (sclerotic). Histologic confirmation of ScGVHD was obtained by 6-mm punch skin biopsy in most patients. Patients categorized as “sclerotic” were compared with patients with “nonsclerotic” skin involvement and those without cutaneous cGVHD.
Protocol evaluation involved collection of comprehensive demographic, historical, clinical, histopathologic, imaging, laboratory, and survival data. Self-reported symptoms were assessed with the use of skin-specific items from the Lee cGVHD Symptom Scale13 and included abnormal skin color, skin thickness, sores on skin, and limited joint movement. Joint involvement was determined by a physiatrist (G.O.J.) with expertise in cGVHD and included measurement of joint ROM and grip strength, as well as walking assessment. Measured ROM was compared with predicted ROM for each individual joint by the use of values established by the American Academy of Orthopedic Surgeons.14 For each patient, the joint with most restriction in ROM was used in the analysis. Percent-predicted grip strength was measured of the dominant hand. A walking velocity in feet/minute was determined by a 2-minute walk test.
NIH cGVHD (0-3) organ-specific scores for 8 organs (skin, eye, mouth, liver, gastrointestinal tract, lungs, joints-fascia, and female genital tract) according to the NIH Consensus Criteria12 were assigned at the conclusion of the comprehensive evaluations by the HSCT clinician who examined the patient. NIH average score represented the sum of each individual organ system divided by the total number of organ systems assessed (8 for female patients [including the genital tract], and 7 for male patients). NIH global scores were graded as “mild” (1-2 organs with scores 1), “moderate” (> 2 organs with score 1, any score 2, or lung score 1), or “severe” (any score of 3 or lung score 2) as described by Filipovich et al.12
Statistical analysis
Preliminary univariate analysis was performed to screen for associations between factors of interest and presence or absence of skin sclerosis. Comparisons were made between the sclerotic and nonsclerotic groups by use of the Wilcoxon rank-sum test for continuous variables. The Fisher exact test was used for dichotomous variables, whereas the Mehta version of the Fisher exact test was used with multiple-level categorical variables.15 The Cochran-Armitage test was used with ordered categorical variables.16
After initial comparisons, factors found to be potentially associated with the presence or absence of skin sclerosis (P < .05) were evaluated with the use of univariate logistic regression analyses (outcome: sclerosis vs no sclerosis). After univariate screening, a multivariable logistic regression model was determined, generally via the use of backward selection modeling.
Factors potentially associated with percent BSA sclerosis were first screened as follows: Spearman rank correlation was used to determine the association between continuous variables and percent BSA sclerosis, interpreted as follows: r > 0.70, strong correlation; 0.5 < r < 0.7, moderately strong correlation; 0.3 < r < 0.5, weak-to-moderately strong correlation; and r < 0.3, weak correlation. The Kruskal-Wallis test was used to test for associations between percent BSA sclerosis and categorical variables, and the Jonckheere-Terpstra trend test was used to test for associations between ordinal variables and percent BSA sclerosis.17 Multiple linear regression analysis included parameters with r > 0.50 for continuous variables and P < .05 for categorical variables in the univariate screening and was performed via the use of backward selection.
Survival was calculated from the date of entry onto the natural history protocol until date of death or last follow-up. This approach was used because the cross-sectional study design did not allow for precise determination of onset of sclerotic symptoms. The probability of survival as a function of time was determined by the Kaplan-Meier method, and the significance of the difference of a pair of Kaplan-Meier curves was determined by the log-rank test. Cox proportional hazards modeling was used to assess the impact of potential prognostic factors jointly on survival. Because there was only a single pediatric death, pediatric patients were excluded from the survival analysis.
Because of the number of tests performed in the univariate analyses, only P values < .01 in the univariate analyses were considered statistically significant. P values between .01 and .05 demonstrated a strong trend toward significance. All P values are 2-tailed and have not been formally adjusted for multiple comparisons.
Results
Patient demographic and transplantation history
Two-hundred six patients (189 adult, 17 children) met criteria for diagnosis of cGVHD according to the NIH cGVHD Consensus Criteria (Table 1).12 Eleven patients were excluded from the study after an evaluation failed to confirm cGVHD (n = 9) or after they were reclassified as having late acute GVHD (n = 2). The mean age at enrollment was 43 years (median, 46 years; range, 3-70 years), and 45.6% of patients were female. The median time from transplantation to protocol entry was 36 months (range, 4-258 months). The majority of patients (77.18%) had undergone peripheral blood stem cell transplantation. The most common indications for transplantation were acute leukemia or myelodysplastic syndrome (n = 89; 41.3%), lymphoma (n = 42; 20.4%), and chronic myelogenous leukemia (n = 30; 14.6%). The majority of patients (n = 123, 63%) had a NIH global cGVHD score defined as “severe. ” One-hundred nine patients (52.9%) manifested ScGVHD. The median BSA involvement among all patients with ScGVHD was 37.1% (37.4% excluding pediatric patients; supplemental Figure 1, available on the Blood Web site; see the Supplemental Materials link at the top of the online article). Percent BSA nonsclerotic skin GVHD was not associated with sclerosis.
Demographic characteristics of NIH cGVHD cohort
| Total number of patients . | n (%) or (range) . |
|---|---|
| All | 206 |
| Adult | 189 (92) |
| Pediatric | 17 (8) |
| Number with ScGVHD of skin | 109 (53) |
| Median BSA sclerosis (adults) | 37.40% |
| Median age, y | 46 (3-70) |
| Sex | |
| Male | 112 (54) |
| Female | 94 (46) |
| Disease | |
| ALL/AML/MDS | 89 (43) |
| CML | 30 (15) |
| CLL | 14 (7) |
| Lymphoma | 42 (20) |
| Multiple myeloma | 15 (7) |
| Aplastic anemia/PNH | 8 (4) |
| Other | 8 (4) |
| Conditioning regimen | |
| Myeloablative | 116 (56) |
| Reduced intensity | 88 (43) |
| Unknown | 2 (1) |
| Stem cell source | |
| BM | 40 (20) |
| Peripheral blood | 159 (77) |
| Cord | 7 (3) |
| Relationship | |
| Related | 136 (66) |
| Unrelated | 70 (34) |
| Sex mismatch | |
| M/M | 50 (25) |
| M/F | 48 (24) |
| F/F | 41 (20) |
| F/M | 45 (22) |
| Unknown | 20 (9) |
| HLA matched | |
| Yes | 167 (81) |
| No | 36 (17) |
| Unknown | 3 (2) |
| Days from transplantation | 1089 (49-1406) |
| cGVHD onset | |
| Progressive | 94 (46) |
| Quiescent | 43 (21) |
| De Novo | 68 (33) |
| Unknown | 1 (0) |
| Median cGVHD treatments | 4 (0-8) |
| GVHD involvement | |
| Skin | 151 (78)* |
| Joints and fascia | 115 (59) |
| Ocular | 157 (81) |
| Mouth | 132 (68) |
| Lung | 145 (75) |
| Liver | 101 (52) |
| GI | 86 (44) |
| Genital (females only) | 44 (47) |
| NIH Global Score | |
| Mild | 4 (2) |
| Moderate | 67 (35) |
| Severe | 123 (63) |
| Total number of patients . | n (%) or (range) . |
|---|---|
| All | 206 |
| Adult | 189 (92) |
| Pediatric | 17 (8) |
| Number with ScGVHD of skin | 109 (53) |
| Median BSA sclerosis (adults) | 37.40% |
| Median age, y | 46 (3-70) |
| Sex | |
| Male | 112 (54) |
| Female | 94 (46) |
| Disease | |
| ALL/AML/MDS | 89 (43) |
| CML | 30 (15) |
| CLL | 14 (7) |
| Lymphoma | 42 (20) |
| Multiple myeloma | 15 (7) |
| Aplastic anemia/PNH | 8 (4) |
| Other | 8 (4) |
| Conditioning regimen | |
| Myeloablative | 116 (56) |
| Reduced intensity | 88 (43) |
| Unknown | 2 (1) |
| Stem cell source | |
| BM | 40 (20) |
| Peripheral blood | 159 (77) |
| Cord | 7 (3) |
| Relationship | |
| Related | 136 (66) |
| Unrelated | 70 (34) |
| Sex mismatch | |
| M/M | 50 (25) |
| M/F | 48 (24) |
| F/F | 41 (20) |
| F/M | 45 (22) |
| Unknown | 20 (9) |
| HLA matched | |
| Yes | 167 (81) |
| No | 36 (17) |
| Unknown | 3 (2) |
| Days from transplantation | 1089 (49-1406) |
| cGVHD onset | |
| Progressive | 94 (46) |
| Quiescent | 43 (21) |
| De Novo | 68 (33) |
| Unknown | 1 (0) |
| Median cGVHD treatments | 4 (0-8) |
| GVHD involvement | |
| Skin | 151 (78)* |
| Joints and fascia | 115 (59) |
| Ocular | 157 (81) |
| Mouth | 132 (68) |
| Lung | 145 (75) |
| Liver | 101 (52) |
| GI | 86 (44) |
| Genital (females only) | 44 (47) |
| NIH Global Score | |
| Mild | 4 (2) |
| Moderate | 67 (35) |
| Severe | 123 (63) |
Values are n (%) unless otherwise specified.
ALL indicates acute lymphoblastic leukemia; AML, acute myelogenous leukemia; BSA, body surface area; cGVHD, chronic GVHD; CLL, chronic lymphocytic leukemia; CML, chronic myelogenous leukemia; F, female; M, male; MDS, myelodysplastic syndrome; NIH, National Institutes of Health; PNH, paroxysmal nocturnal hemoglobinuria; and ScGVHD, sclerotic-type chronic GVHD.
Includes both sclerotic and nonsclerotic dermatologic manifestations of cGVHD.
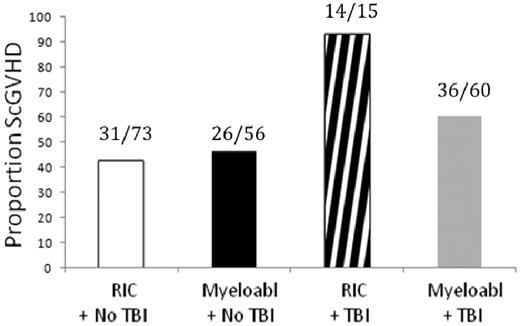
By univariate analysis, lower Karnofsky score (P = .003), greater number of previous immunosuppressive agents (P < .0001), greater intensity of immunosuppressive therapy at time of evaluation (P = .021), longer time from transplantation to diagnosis of cGVHD (P < .0001), longer time from diagnosis of cGVHD to enrollment in the study (P < .0001), and history of total-body irradiation (TBI) before transplantation (P = .0023) were associated with ScGVHD (Table 2). Of 15 patients who received TBI as part of a reduced-intensity regimen, 14 manifested ScGVHD, comprising the greatest risk group for the development of ScGVHD in this cohort (P < .0001; Figure 2).
Variables significantly associated with ScGVHD in univariate analysis
| . | Nonsclerotic GVHD . | ScGVHD . | P . |
|---|---|---|---|
| Patient, transplantation, and cGVHD characteristics | |||
| Karnofsky status | 82.25 (1.40) | 77.45 (1.20) | .003 |
| Number of previous therapies | 2.83 (0.18) | 4.72 (0.18) | < .0001 |
| Days from transplantation to cGVHD diagnosis | 245.2 (25.60) | 350.94 (34.45) | < .0001 |
| Days from cGVHD diagnosis to enrollment | 953.48 (136.83) | 1293.28 (113.37) | < .0001 |
| Total body irradiation | 25/97 (25.77%) | 50/107 (46.73%) | .0023 |
| High intensity of immunosuppression* | 30/97 (30.93%) | 49/108 (45.4%) | .021 |
| Laboratory parameters | |||
| Platelet count, 1000/μL | 224.86 (10.06) | 293.47 (11.37) | < .0001 |
| CRP, mg/dL | 1.16 (0.22) | 1.23 (0.92) | .0012 |
| C3 complement, mg/dL | 120.93 (2.62) | 144.21 (2.97) | < .0001 |
| C4 complement, mg/dL | 24.48 (0.82) | 29.45 (1.00) | < .0001 |
| Total complement, mg/dL | 123.51 (3.21) | 135.43 (3.34) | .017 |
| IgM, mg/dL | 87.6 (7.35) | 71.68 (7.60) | .024 |
| Cardiolipin IgM, mg/dL* | 8/96 (8.33%) | 1/101 (0.99%) | .0019 |
| Percent predicted FVC | 84.73 (1.99) | 74.58 (2.10) | .013 |
| Functional measures | |||
| Severe NIH Joint/Fascia score* | 0/88 | 20/106 (18.87%) | < .0001 |
| Severe NIH global score | 37/88 (42.05%) | 86/106 (81.13%) | < .0001 |
| ROM | 74.72 (2.89) | 48.02 (3.03) | < .0001 |
| Grip strength | 73.41 (3.03) | 56.77 (2.35) | < .0001 |
| Patient-reported measures | |||
| Skin color change: quite a bit or extreme* | 15/79 (18.99%) | 28/88 (31.82%) | .0074 |
| Skin thickness: quite a bit or extreme* | 6/79 (7.59%) | 46/87 (52.87%) | .0001 |
| Sores on skin: quite a bit or extreme * | 9/77 (11.69%) | 16/88 (18.18%) | .0239 |
| Skin itching: quite a bit or extreme* | 11/79 (13.92%) | 29/88 (32.95%) | .0112 |
| Joint stiffness: quite a bit or extreme * | 7/79 (8.86%) | 41/88 (46.59%) | .0001 |
| . | Nonsclerotic GVHD . | ScGVHD . | P . |
|---|---|---|---|
| Patient, transplantation, and cGVHD characteristics | |||
| Karnofsky status | 82.25 (1.40) | 77.45 (1.20) | .003 |
| Number of previous therapies | 2.83 (0.18) | 4.72 (0.18) | < .0001 |
| Days from transplantation to cGVHD diagnosis | 245.2 (25.60) | 350.94 (34.45) | < .0001 |
| Days from cGVHD diagnosis to enrollment | 953.48 (136.83) | 1293.28 (113.37) | < .0001 |
| Total body irradiation | 25/97 (25.77%) | 50/107 (46.73%) | .0023 |
| High intensity of immunosuppression* | 30/97 (30.93%) | 49/108 (45.4%) | .021 |
| Laboratory parameters | |||
| Platelet count, 1000/μL | 224.86 (10.06) | 293.47 (11.37) | < .0001 |
| CRP, mg/dL | 1.16 (0.22) | 1.23 (0.92) | .0012 |
| C3 complement, mg/dL | 120.93 (2.62) | 144.21 (2.97) | < .0001 |
| C4 complement, mg/dL | 24.48 (0.82) | 29.45 (1.00) | < .0001 |
| Total complement, mg/dL | 123.51 (3.21) | 135.43 (3.34) | .017 |
| IgM, mg/dL | 87.6 (7.35) | 71.68 (7.60) | .024 |
| Cardiolipin IgM, mg/dL* | 8/96 (8.33%) | 1/101 (0.99%) | .0019 |
| Percent predicted FVC | 84.73 (1.99) | 74.58 (2.10) | .013 |
| Functional measures | |||
| Severe NIH Joint/Fascia score* | 0/88 | 20/106 (18.87%) | < .0001 |
| Severe NIH global score | 37/88 (42.05%) | 86/106 (81.13%) | < .0001 |
| ROM | 74.72 (2.89) | 48.02 (3.03) | < .0001 |
| Grip strength | 73.41 (3.03) | 56.77 (2.35) | < .0001 |
| Patient-reported measures | |||
| Skin color change: quite a bit or extreme* | 15/79 (18.99%) | 28/88 (31.82%) | .0074 |
| Skin thickness: quite a bit or extreme* | 6/79 (7.59%) | 46/87 (52.87%) | .0001 |
| Sores on skin: quite a bit or extreme * | 9/77 (11.69%) | 16/88 (18.18%) | .0239 |
| Skin itching: quite a bit or extreme* | 11/79 (13.92%) | 29/88 (32.95%) | .0112 |
| Joint stiffness: quite a bit or extreme * | 7/79 (8.86%) | 41/88 (46.59%) | .0001 |
For each of the groups, fibrotic and nonfibrotic, continuous variables are shown as means and SE. Categorical variables are shown as proportions with percentages for each group.
cGVHD indicates chronic GVHD; CRP, C-reactive protein; FVC, forced vital capacity; NIH, National Institutes of Health; ROM, range of motion; and ScGVHD, sclerotic-type chronic GVHD.
Ordered categorical variable, presented as fraction for simplicity. Comparisons were performed with the use of the Cochran-Armitage test for trend.
TBI is associated with increased risk of development of ScGVHD. The association between TBI and ScGVHD was demonstrated most strongly among patients treated with reduced-intensity conditioning (RIC). Of 15 patients who received TBI as part of a RIC conditioning regimen, 14 demonstrated ScGVHD (P = .0114).
TBI is associated with increased risk of development of ScGVHD. The association between TBI and ScGVHD was demonstrated most strongly among patients treated with reduced-intensity conditioning (RIC). Of 15 patients who received TBI as part of a RIC conditioning regimen, 14 demonstrated ScGVHD (P = .0114).
Age, sex, indication for transplantation, current remission status, CMV status at transplantation, and cGVHD onset (quiescent, de novo, or progressive) were not associated with ScGVHD (supplemental Table 1). Donor sex, stem cell source (peripheral blood, BM, or cord blood), relationship of donor to patient (related or unrelated), degree of HLA match, GVHD prophylaxis, history of acute skin GVHD, and receipt of one or more donor lymphocyte infusion(s) were also not associated with ScGVHD.
Laboratory and pulmonary function parameters
Elevated platelet count (P < .0001), C-reactive protein (CRP; P = .0012), C3 (P < .0001) and C4 (P < .0001), total complement (P = .017), and IgM (P = .04) were each associated with ScGVHD in univariate analysis. Elevated cardiolipin IgM was associated with decreased risk of ScGVHD (P = .0019), although only 9 patients demonstrated abnormal cardiolipin IgM titers. Lymphocyte count, absolute neutrophil, eosinophil, and lymphocyte subset counts (CD4 T-cell, CD8 T-cell, B-cell, natural killer cell), IgG, IgA, lactate dehydrogenase, erythrocyte sedimentation rate, albumin, ferritin, and β2 microglobulin had no relationship to ScGVHD. Rheumatoid factor, ANA, cardiolipin IgG, antimitochondrial DNA, anti-DS DNA, and a panel of other autoantibodies (supplemental Table 1) also had no relationship to ScGVHD. Decreased forced vital capacity (FVC) was associated with skin sclerosis (P = .013); however, percent predicted forced expiratory volume in one second (FEV1; P = .16), FEV1/FVC (P = .11), and diffusing capacity for carbon monoxide (P = .12) were not related to ScGVHD. Sclerotic involvement of the anterior torso was detected in 85 of 192 (44.3%) of adult patients. A greater proportion of patients with sclerosis of the anterior torso (48/85, 56.7%) had diminished FVC (< 80% predicted), compared with those without anterior torso sclerosis (42/107, 39.3%; P = .02).
cGVHD involving other organ systems and functional disability
Decreased joint ROM (P < .0001 and grip strength P < .0001) and increased severity of NIH Joint and Fascia score (P < .0001) were strongly associated with the presence of ScGVHD. ScGVHD was not associated with walk velocity during 2-minute walk test. Patients with ScGVHD had NIH global scores graded as more severe (P < .0001); however, individual NIH scores for eyes, lung, gastrointestinal tract, liver, mouth, and genitalia were not associated with ScGVHD.
Patient self-report of symptoms
Patients with ScGVHD reported significantly more change in skin color (P = .0074), increased skin thickness (P < .0001), sores on the skin (P = .024), itchy skin (P = .011), and joint stiffness (P < .0001) than nonsclerotic patients. Patient self-report of rash was not associated with degree of sclerosis (P = .72).
Multivariate predictive models of ScGVHD
Karnofsky performance status, TBI, platelet count, C3 complement, C4 complement, total complement, FVC percent predicted, and TLC percent-predicted were included in an initial multivariable logistic predictive model. Time from cGVHD diagnosis to enrollment, CRP, and IgM were excluded after univariate analysis. The final model, including platelets (P = .0011), C3 complement (P < .0001), and history of TBI as a component of a reduced-intensity conditioning regimen (P = .0114), predicted approximately 72%-73% of patients with and without ScGVHD when tested in the same patients used to develop the model (Table 3).
Factors highly predictive of ScGVHD in a multivariable regression model
| Variable . | Nonsclerotic . | Sclerotic . | P in model . |
|---|---|---|---|
| TBI in reduced-intensity setting | 1 (6.67%) | 14 (93.33%) | .0114 |
| C3, mg/dL | 120.93 (2.62) | 144.21 (2.97) | < .001 |
| Platelets, 1000/μL | 224.86 (10.06) | 293.47 (11.37) | .0011 |
| Variable . | Nonsclerotic . | Sclerotic . | P in model . |
|---|---|---|---|
| TBI in reduced-intensity setting | 1 (6.67%) | 14 (93.33%) | .0114 |
| C3, mg/dL | 120.93 (2.62) | 144.21 (2.97) | < .001 |
| Platelets, 1000/μL | 224.86 (10.06) | 293.47 (11.37) | .0011 |
For each of the groups, fibrotic and nonfibrotic, continuous variables are shown as means and standard errors. Categorical variables are shown as proportions with percentages for each group.
ScGVHD indicates sclerotic-type chronic GVHD; and TBI, total body irradiation.
To develop a multiple linear regression model for percent BSA sclerosis, factors at least moderately associated with severity of ScGVHD in univariate analysis were included. This model initially considered time from transplantation to diagnosis of GVHD, time from GVHD to enrollment in protocol, Karnofsky performance status, platelet count, CRP, C3 and C4 complement levels, FVC % predicted, CRP, history of TBI, and cardiolipin IgM. After backward selection, C3 (P < .0001), platelets (P < .03), and FVC percent predicted (P < .0001) were associated with greater BSA sclerotic skin involvement, although regression modeling demonstrated a moderately strong overall relationship (adjusted r square = 0.51) between the factors and increased BSA of sclerotic skin involvement.
Survival analysis
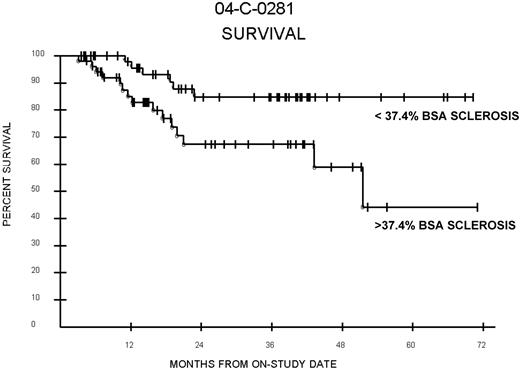
Among patients with ScGVHD, adult patients with greater than median BSA sclerosis (37.4%) had poorer survival than those with less than median BSA sclerosis (P = .015; Figure 3). A total of 67% of patients with greater than median ScGVHD were alive at 3 years follow-up compared with 85% of patients with less than median ScGVHD (median survivor follow-up, 29.8 months). Poorer survival was also observed among patients with greater than median BSA sclerosis compared with all other patients, including those with no sclerosis or values below the median (P = .023). However, in multivariable analysis, after adjusting for NIH lung score, Karnofsky score, and lymphocyte count (factors associated with survival in this cohort on previous analyses), percent-BSA ScGVHD was no longer associated with survival (data not shown). Overall survival of patients with ScGVHD was not significantly different from patients without ScGVHD (P = .58).
Percent BSA and survival. Survival among patients with ScGVHD with percent BSA involvement above and below the median (37.4%) for the entire ScGVHD group. Log-rank (P = .015). In multivariable analysis, after adjusting for NIH lung score, Karnofsky score, and lymphocyte count (factors associated with survival in this cohort on previous analyses), percent-BSA ScGVHD was no longer associated with survival.
Percent BSA and survival. Survival among patients with ScGVHD with percent BSA involvement above and below the median (37.4%) for the entire ScGVHD group. Log-rank (P = .015). In multivariable analysis, after adjusting for NIH lung score, Karnofsky score, and lymphocyte count (factors associated with survival in this cohort on previous analyses), percent-BSA ScGVHD was no longer associated with survival.
Discussion
The NIH cGVHD Natural History cohort comprises a unique population of cGVHD patients with significant disease severity (63% NIH global score of “severe”) and high prevalence of ScGVHD (53%), thereby allowing for detailed analysis of factors associated with established ScGVHD. Although TBI has been associated with elevated risk of acute GVHD,18,19 to our knowledge this is the first study in which an association between TBI conditioning and the later development of ScGVHD has been described. In a series of 7 patients with ScGVHD described by Chosidow et al,3 all had received pretransplantation TBI. Similarly, in a series of 17 ScGVHD patients described by Peñas et al, 13 patients received TBI before transplantation.2 However, these studies did not distinguish between TBI in the setting of myeloablative versus reduced-intensity conditioning.
In our cohort, the association between TBI and ScGVHD was particularly strong in reduced-intensity regimens, which use lower doses of TBI than used in myeloablative regimens. Reduced-intensity conditioning is frequently used to avoid the toxicity associated with myeloablation20 and may also result in less acute GVHD than traditional myeloablation.21 Unfortunately, the use of reduced-intensity regimens has not led to a reduced incidence of cGVHD,22 possibly because of survival of recipient antigen-presenting cells.23 Nonetheless, ScGVHD was detected in both myeloablative and reduced-intensity regimens in greater frequency than in regimens that did not use TBI, suggesting TBI may confer an independent risk of ScGVHD. Because the number of reduced-intensity regimens containing TBI represented a minority of all TBI patients, further investigation is needed to confirm the interrelation between reduced-intensity conditioning, TBI, and risk of ScGVHD.
Localized radiation therapy has been associated with the development of lichenoid cGVHD and ScGVHD limited to the site of radiotherapy, suggesting antecedent subclinical skin injury induces “locus minoris resistentiae,” that is, a site at increased risk for development of skin disease.24-26 In addition, ScGVHD tends be a late manifestation of cGVHD, bearing similarity to the tardive development of chronic radiation fibrosis after localized radiation exposure. Several mechanisms may be relevant for the development of ScGVHD after TBI. In a murine GVHD model, mice grafted with irradiated syngeneic skin and injected with allogeneic lymphoid cells developed GVHD in the irradiated graft, whereas this did not occur in nonirradiated grafts.27 Studies by our group and others have suggested that type I IFN (α/β) plays a key role in cGVHD pathogenesis.28 Irradiation may activate damage-associated molecular patterns through toll-like receptor molecules, leading to type I IFN signaling,29 a cytokine implicated in the development of sclerosis and autoimmunity.30 Radiation treatment may also produce elevated IFN-γ,31 which, in addition to IFN-inducible chemokines, is up-regulated in the skin of a murine model of sclerodermatous GVHD.32 In this model, mice receive TBI before donor cell infusion.32 Local production of IFN and downstream factors may promote and maintain T-cell cytotoxicity. Sclerotic changes in this model are blocked by inhibitors of TGFβ, the critical cytokine driving fibrosis in ScGVHD.33 Other nonspecific effects of radiation, including depletion of local immune regulatory factors, damage to keratinocytes, and stimulation of systemic cytokine production, also may predispose patients to the development of GVHD.27
Elevated serum C3 complement was also associated with ScGVHD. Patients with systemic sclerosis have also been found to have elevated levels of serum complement after production of autoantibodies forming immune complexes.34 Immune complexes have been implicated in GVHD,35 and the success of the B-cell inhibitor rituximab in some patients with cGVHD suggests an important role of B cells in the disease.36 Moreover, IFN may contribute to humoral immunity in cGVHD because patients with cGVHD demonstrate elevated levels of B-cell activating factor, which is stimulated by IFN and TGFβ.37,38 Deposits of C3 and IgM can be found at the dermal-epidermal junction in skin biopsies of patients with GVHD.39 Although tissue deposition of complement would be expected to lead to lower levels of circulating complement, the inflammatory disease state of GVHD may theoretically result in a net increase in the synthesis of complement in response to cytokines such as IL-6.40 Complement has been shown to stimulate other fibrotic processes, such as cirrhosis, by the expression of complement receptors on myofibroblasts in the liver.41
Low platelet count is associated with poor survival in cGVHD42 ; however, in the cohort reported here, elevated platelet counts were significantly associated with ScGVHD. This finding may reflect the role of platelets as acute phase reactants40 ; however, elevated circulating platelet counts and overexpression of the collagen I receptor on platelets has been found in patients with systemic sclerosis.43 Patients with systemic sclerosis demonstrate enhanced platelet aggregation and release of growth factors, such as PDGF, TGF-β, β-fibroblast growth factor, and connective tissue growth factor, which may stimulate the synthesis of collagens and other matrix proteins.44
Acute GVHD is an established risk factor for the development of cGVHD18 ; however, in the current cohort acute GVHD did not predispose patients to a specific increased risk of ScGVHD, in agreement with Skert et al.4 Nonsclerotic “lichenoid” skin lesions are also commonly thought to precede the development of ScGVHD.2,45 We did not identify an association between concurrent erythematous/lichenoid cGVHD skin involvement and sclerosis in our study. Others have described ScGVHD occurring independent of nonsclerotic skin involvement.2,3
cGVHD involvement in other organ systems was approximately equally distributed in the sclerotic and nonsclerotic groups, which is consistent with smaller series,2-4 and likely reflects the heterogeneous nature of the disease.3,8 However, the lack of significant differences in organ involvement in patients with ScGVHD may also reflect a recruitment bias to the extent that patients without ScGVHD frequently manifested other severe organ involvement that led to their enrollment into the study.
In contrast to previous smaller series,4,9 we did not find an association between autoantibody development and ScGVHD. The role of autoantibodies in the pathogenesis of ScGVHD remains uncertain. Antinuclear, antitopoisomerase I (antiScl70), and anticentromere antibodies are strongly associated with systemic sclerosis.46 However, elevated autoimmune serologies, commonly detected in cGVHD,47 are typically of low titer and lack the specificity found in primary autoimmune disease. In our cGVHD cohort, 30% of patients were ANA-positive; however, the distribution was approximately equal among ScGVHD and nonsclerotic patients. Autoimmune testing in our cohort was determined by a single assessment at the time of enrollment and may not be representative of patients' autoimmune status at other times. Autoantibody titers may also change over the natural history of cGVHD, with greater levels corresponding to increased severity and lower levels correlating with response to treatment.9
ScGVHD was highly associated with decreased ROM as assessed by both the NIH Joint and Fascia score and by direct ROM measurement. ScGVHD was also associated with decreased grip strength, providing validation that these objective measures of functional capacity may be useful as surrogate outcome markers of sclerotic disease in clinical trials. A much greater proportion of our ScGVHD cohort experienced functional limitations than has been generally reported in the literature.8,10 This finding likely reflects a high rate of detection because of the diverse assessment tools performed in this study, as well as significant disease severity of our cohort. Nonetheless, these data underscore the importance of careful characterization of the long-term effects of ScGVHD by the use of measures that encompass strength, ROM, and functional capacity to fully demonstrate the impact of the disease. The Lee cGVHD Symptom Scale13 is a validated patient self-report symptom assessment tool but has never been specifically evaluated in ScGVHD patients. Our findings suggest that patient self-report of skin color change, rashes, ulcers, thickness, and joint stiffness by Lee symptom scale correlate well with ScGVHD severity and may be useful as an additional surrogate clinical marker of ScGVHD disease activity and severity. The functional impact of ScGVHD in our study reflects the significant impairment in quality of life domains that has been reported in cGVHD.48
We found that adult patients with extensive skin sclerosis (greater than median BSA, 37.4%) had significantly poorer survival than cGVHD patients with limited skin sclerosis or no sclerosis. Extensive skin involvement in cGVHD is generally thought to portend poor survival; however, the specific impact of sclerosis has not been evaluated. When we controlled for Karnosfksy status and other prognostic factors, however, we found that sclerosis was no longer independently associated with survival. This may, in part, be because of the direct interaction between severe ScGVHD and Karnofksy status, which was significantly reduced in patients with ScGVHD (P = .003). In our analysis, lower FVC was strongly related to both the presence and severity of sclerosis. The correlation of FVC decline with systemic sclerosis has been suggested by others.49 FVC can be used as a marker for restrictive lung disease when FEV1/FVC is normal,50 and in our population, may indicate extrinsic restriction of the chest/abdominal wall because of ScGVHD. Indeed, a greater proportion of patients with ScGVHD of the anterior torso demonstrated diminished FVC compared with those without ScGVHD of this region (P = .02).
There are limitations of this study's cross-sectional design. First, the investigations undertaken reflect ScGVHD during a single period and may not be representative of ScGVHD in its earliest stages. Second, the significant extent of skin involvement observed in this cohort may not be generalizable to patients with mild/limited sclerotic skin disease manifestations or whose manifestations have responded to conventional treatment approaches. By the same token, it is possible that patients with very severe sclerotic disease may have been too unwell to travel to enroll in the NIH study or may have died before enrollment. Finally, the referral pattern for patients without ScGVHD is biased toward recruitment of patients with severe, multisystem and refractory disease; that is, characteristics that prompted referral to the NIH study but that may portend overall poor survival in the non-ScGVHD comparison group. As a result, our analysis may underestimate the impact of ScGVHD on overall survival.
ScGVHD is a late complication of allogeneic HSCT for which no single satisfactory treatment exists. Given the relative rarity of this condition and the lack of existing registry data on this manifestation, prospective analysis studying the risk factors for ScGVHD will require many years of data collection. Our results implicate pretransplantation TBI as an important risk factor for ScGVHD, particularly in the reduced-intensity setting. If confirmed in prospective studies, this finding could impact the use of TBI, especially in at-risk patients (eg, those with a greater degree of mismatch or other significant risk factors for the development of cGVHD). In addition, we propose that elevated C3 and platelet count may be associated with ScGVHD. Future studies are needed to determine whether these common laboratory tests can be used in a predictive manner for the identification or monitoring of ScGVHD. Finally, these data demonstrate that ScGVHD is a physically debilitating manifestation of cGVHD, which also may adversely affect survival. The severity of ScGVHD strongly correlated with ROM limitations and decreased grip strength, as did self-reported measures of skin activity by the Lee symptom scale. These parameters may serve to better characterize sclerotic disease in the clinical trial setting, thereby improving interpretation of clinical outcome data and, as a corollary, improving therapeutic decision making and disease monitoring in patients with ScGVHD.
An Inside Blood analysis of this article appears at the front of this issue.
The online version of the article contains a data supplement.
Presented in abstract form at the 71st annual meeting of the Society of Investigative Dermatology, Phoenix, AZ, May 2-7, 2011.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgments
This study was supported by the Intramural Research Program of the National Institutes of Health, Center for Cancer Research, National Cancer Institute. K.J.M. was supported by the Clinical Research Training Program, a public-private partnership supported jointly by the National Institutes of Health and Pfizer Inc (via a grant to the Foundation for National Institutes of Health from Pfizer Inc).
The publisher or recipient acknowledges right of the U.S. government to retain a nonexclusive, royalty-free license in and to any copyright covering the article.
National Institutes of Health
Authorship
Contribution: K.B., G.O.J., K.M.W., S.A.M., M.D., F.T.H., S.Z.P., and E.W.C. designed the research; K.B., L.G., G.O.J., K.M.W., S.A.M., M.D., F.T.H., S.Z.P., and E.W.C. collected the data; K.J.M., S.M.S., and E.W.C. analyzed results and made the tables and figures; and K.J.M. and E.W.C. wrote the paper.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Edward W. Cowen, MD, MHSc, Dermatology Branch, Center for Cancer Research, National Cancer Institute, National Institutes of Health, 10 Center Drive, MSC 1908, Bethesda, MD 20892; e-mail: cowene@mail.nih.gov.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal