Abstract
During disease progression in myelodysplastic syndromes (MDS), clonal blasts gain a more aggressive nature, whereas nonclonal immune cells become less efficient via an unknown mechanism. Using MDS cell lines and patient samples, we showed that the expression of an immunoinhibitory molecule, B7-H1 (CD274), was induced by interferon-γ (IFNγ) and tumor necrosis factor-α (TNFα) on MDS blasts. This induction was associated with the activation of nuclear factor-κB (NF-κB) and nearly completely blocked by an NF-κB inhibitor, pyrrolidine dithiocarbamate (PDTC). B7-H1+ MDS blasts had greater intrinsic proliferative capacity than B7-H1− MDS blasts when examined in various assays. Furthermore, B7-H1+ blasts suppressed T-cell proliferation and induced T-cell apoptosis in allogeneic cocultures. When fresh bone marrow samples from patients were examined, blasts from high-risk MDS patients expressed B7-H1 molecules more often compared with those from low-risk MDS patients. Moreover, MDS T cells often overexpressed programmed cell death 1 (PD-1) molecules that transmit an inhibitory signal from B7-H1 molecules. Taken together, these findings provide new insight into MDS pathophysiology. IFNγ and TNFα activate NF-κB that in turn induces B7-H1 expression on MDS blasts. B7-H1+ MDS blasts have an intrinsic proliferative advantage and induce T-cell suppression, which may be associated with disease progression in MDS.
Introduction
B7-H1 (CD274), which was identified by us as a costimulatory molecule, plays a crucial role in T-cell regulation in various immune responses.1,2 B7-H1 molecules deliver a costimulatory signal through an unknown receptor on naive T cells.1-3 They also deliver an inhibitory signal to activated T cells through programmed cell death 1 (PD-1) molecules,4 which are a type I transmembrane protein belonging to the CD28 receptor family and were originally identified in T cells undergoing apoptosis.5 B7-H1 expression is detected not only on antigen-presenting cells but also on activated T cells and some tumor cells (ie, renal cell, colon, breast, and lung carcinoma, and Hodgkin lymphoma).6-10 Rodent data suggest that B7-H1 molecules on tumor cells deliver negative signals through PD-1 and other receptors on tumor-specific cytotoxic T lymphocytes and inhibit antitumor immune responses.11,12 Consistent with those data, it was reported that in patients with renal cell carcinoma and breast cancer, patients whose tumor cells expressed B7-H1 had a poor prognosis.9,13 In a mouse leukemia model in which mice were immunized with irradiated DA1-3b leukemia cells and then challenged with live DA1-3b cells, only leukemia cells expressing high levels of B7-H1 survived for a long period. Moreover, these cells gained tolerance to specific cytotoxic T lymphocyte–mediated killing.14 Therefore, B7-H1 molecules on leukemia cells may be associated with immune evasion in this model.
Myelodysplastic syndromes (MDS) are clonal hematologic stem cell disorders characterized by cytopenias, excessive apoptosis of hematopoietic cells, and a high risk of progression to acute myeloid leukemia (AML). In MDS, various immune abnormalities, including lymphopenia and T-cell dysfunction, have been reported,15-17 although data on B7-related molecules, in particular B7-H1, are lacking. With disease progression, that is, with increases in blast percentages in the bone marrow (BM), clonal MDS blasts become less apoptotic, more proliferative, and phenotypically more immature,18,19 whereas nonclonal immune cells become less efficient with a decrease in T-cell number and increases in T-cell apoptosis and in the number of CD4+ regulatory T cells.16,20,21 However, the mechanism for these observed phenomena linked to disease progression is largely unknown. It is also notable that in a recent study in which MDS patients were treated with the immunomodulatory drug lenalidomide,22 the results suggested that appropriate immunomodulation is effective in suppressing/eradicating MDS clones at least in some cases.23 In the present study, we investigated whether B7-H1 molecules are expressed on MDS blasts, and if so, whether they are associated with the pathophysiology of MDS.
Methods
Cell lines and patients
F-36P, OIH-1 (Riken Cell Bank, Ibaraki, Japan), and SKM-1 (Health Science Research Resources Bank, Osaka, Japan) cells, which are appropriate among the limited number of MDS-related cell lines,24 have a blast morphology, and have been established from AML patients transformed from MDS (AL-MDS), were cultured in RPMI 1640 medium containing 10% fetal bovine serum, and l-glutamine 1mM. Interleukin-3 (IL-3; 5 ng/mL; Peprotech) and granulocyte colony–stimulating factor (5 ng/mL; Peprotech) were added to the F-36P and OIH-1 cultures, respectively, according to the instructions of the suppliers. The RPMI 1640 medium with or without IL-3 or granulocyte colony–stimulating factor is simply referred to as “the medium” in the present experiments. Because OIH-1 cells are often difficult to maintain, we used F-36P and SKM-1 cells for most experiments.
Portions of BM cells aspirated from subjects for diagnostic purposes were used after receiving written informed consent. The study participants included 29 MDS patients, 32 AL-MDS patients in whom a history of MDS was confirmed, and 10 hematologically normal subjects (Table 1). The patients were diagnosed according to the World Health Organization classification.25 Patients with secondary MDS and those who had had infections within 1 month before this study were excluded. Karyotypes were analyzed using the standard G-banding technique. Patients were classified according to the International Prognostic Scoring System (IPSS) as described previously.26 Peripheral blood (PB) was sampled to examine PD-1 expression on T cells. Mononuclear cells (MNCs) separated from BM and PB samples with Histopaque (Sigma-Aldrich) density centrifugation were also used in experiments.
Patient characteristics for study comparing blasts in MDS
| Characteristic . | No. of patients . |
|---|---|
| Sex, M/F | 35/26 |
| Median age, y (range) | 70 (32-88) |
| World Health Organization subtype | |
| RCUD | 9 |
| RCMD | 6 |
| RAEB-1 | 5 |
| RAEB-2 | 9 |
| AL-MDS | 32 |
| IPSS category* | |
| Low | 6 |
| Intermediate-1 | 9 |
| Intermediate-2 | 10 |
| High | 15 |
| Unknown | 1 |
| Characteristic . | No. of patients . |
|---|---|
| Sex, M/F | 35/26 |
| Median age, y (range) | 70 (32-88) |
| World Health Organization subtype | |
| RCUD | 9 |
| RCMD | 6 |
| RAEB-1 | 5 |
| RAEB-2 | 9 |
| AL-MDS | 32 |
| IPSS category* | |
| Low | 6 |
| Intermediate-1 | 9 |
| Intermediate-2 | 10 |
| High | 15 |
| Unknown | 1 |
MDS indicates myelodysplastic syndromes; RCUD, refractory cytopenia with unilineage dysplasia; RCMD, refractory cytopenia with multilineage dysplasia; RAEB, refractory anemia with excess blasts; AL-MDS, acute myeloid leukemia transformed from MDS; and IPSS, International Prognostic Scoring System.
Not applicable for patients whose bone marrow contained 30% or more blasts.
To induce B7-H1 expression, the cells were cultured in the presence of the human cytokines interferon-γ (IFNγ; 1000 U/mL; Peprotech), IL-1β (10 ng/mL; Peprotech), transforming growth factor-β (TGFβ, 10 ng/mL; Peprotech), and tumor necrosis factor-α (TNFα; 500 U/mL; Peprotech) for 2 days. An inhibitor of nuclear factor-κB (NF-κB), pyrrolidine dithiocarbamate (PDTC; Sigma-Aldrich), was added to cultures in some experiments. In some experiments, B7-H1+ and B7-H1− blasts from patient samples or cell lines and CD34+ cells from patient samples were purified using fluorescence-activated cell sorting (FACS) as described previously.27
This study protocol was approved by the Institutional Review Board of Nippon Medical School.
Reverse-transcription polymerase chain reaction
Total cellular RNA was extracted from exponentially growing cell lines and reverse-transcribed with Superscript II Reverse transcriptase (Invitrogen) using random hexamers. The success of cDNA synthesis was monitored by the reverse-transcription polymerase chain reaction (PCR) of β-actin. PCR synthesis using primers for B7-H1 was performed as described previously.28
In some experiments, purified B7-H1+ and B7-H1− F-36P cells were used for the RNA source and their cyclin D1, D2, D3, E1, and E2 gene expressions were examined using real-time quantitative PCR as described previously.27 Their primers are shown in supplemental Table 1 (available on the Blood Web site; see the Supplemental Materials link at the top of the online article).
B7-H1 and PD-1 flow cytometric analyses
All antibodies used in this study were mouse anti–human monoclonal antibodies. B7-H1 expression on MDS cell lines and BM blasts from subjects as well as PD-1 expression on circulating T cells were determined using flow cytometry (FCM), as described previously.16,29 MDS cell lines were stained with anti–B7-H1–phycoerythrin (PE; eBioscience). The BM MNCs were stained with anti-CD45–peridin chlorophyll (PerCP), anti-CD34–fluorescein isothiocyanate (FITC; BD Biosciences), and anti–B7-H1–PE. In MDS patients, blasts were gated using the CD45-gating method and the gating was confirmed by CD34-positivity of the gated blasts. In some cases, the gated blasts were isolated using FACS, and it was confirmed that they were actual blasts. Because blasts from hematologically normal subjects were often too few to gate correctly, B7-H1 expression was examined on CD34+ myeloblasts, which were identified according to the gating method described in detail previously.29 PB MNCs were stained with anti–PD-1–FITC (eBioscience) and either PE-conjugated anti-CD3, -CD4, or -CD8 (BD Biosciences) or, in some cases, with anti–PD-1–FITC, anti–HLA-DR-PE (Beckman Coulter), and anti-CD3–PerCP. Single-labeled cells were used to compensate for the fluorescence emission overlap of each fluorochrome into inappropriate channels. Isotype-matched negative controls were used in all assays.
Immunocytochemistry for NF-κB p65 localization
The nuclear translocation of NF-κB p65 (ie, the activation of NF-κB) was examined using the immunocytochemical method.30 Briefly, cells fixed with paraformaldehyde were plated on a poly-L-lysine–coated glass slide, air-dried, and treated with 0.2% Triton X-100. After washing in phosphate-buffered saline, slides were blocked with 5% normal goat serum for 1 hour and then incubated with rabbit polyclonal anti–human p65 antibody (Cell Signaling Technology) at 1:100 dilution. After overnight incubation at 4°C, the slides were washed and incubated with goat anti–rabbit immunoglobulin G (IgG)–Alexa Fluor 594 at 1:100 dilution for 1 hour. Stained slides were mounted with mounting medium containing 4′-6-diamidino-2-phenylindole for nuclear staining (Vector Laboratories) and analyzed under a fluorescence microscope (IX71; Olympus). Images were acquired and analyzed with Aquacosmos software (Hamamatsu Photonics).
Western blot analysis of NF-κB p65 translocation
Extracts from the cytoplasmic and nuclear fractions of cells were prepared with the Lysate Preparation Module (IMGENEX). Equal amounts of extracts (corresponding to 1 × 105 cells) from each fraction were separated on 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane. The blots were probed with a monoclonal antibody for p65 (Cell Signaling Technology) and detected using an ECL Plus detection kit (GE Healthcare) with horseradish peroxidase-conjugated anti–rabbit IgG (Cell Signaling Technology). Band intensity was quantified with the use of Scion Image software (Scion Corporation).
Cell-cycle, bromodeoxyuridine, and Ki-67 analyses, colony-forming assay, and apoptosis analysis
For cell-cycle analysis, cells were stained with unlabeled anti–B7-H1 antibody (eBioscience) and then with anti–mouse IgG-FITC. After washing, the cells were fixed in 70% cold ethanol, washed, and resuspended in 100 μL of phosphate-buffered saline containing 1 μL of RNase and 0.1 mg/mL propidium iodide (PI; Sigma-Aldrich). The cell cycle of B7-H1+ and B7-H1− cells was determined using FCM.31
For bromodeoxyuridine (BrdU) analysis, cells were pulsed with BrdU (BrdU flow kit; BD Biosciences) at a concentration of 10μM for 1 hour, washed, and stained with anti–B7-H1–PE. Then, cells were permeabilized and labeled with anti-BrdU–FITC according to the manufacturer's instructions. For Ki-67 analysis, cells were stained with anti–B7-H1–PE and anti-CD45–PerCP, washed, permeabilized with 75% ethanol, and stained with anti–Ki-67–FITC (FITC mouse anti–human Ki-67 set; BD Biosciences) according to the manufacturer's instructions.
The colony-forming activity of purified B7-H1+ and B7-H1− F-36P cells was examined after culturing in MethoCult H4230 methylcellulose medium (StemCell Technologies) supplemented with IL-3 (10 ng/mL). Colonies (aggregates of 50 cells or more) were scored on day 7 of culture.
For apoptosis analysis, the cells were stained with one of the PE-conjugated antibodies to CD3, CD4, or CD8 and with annexin V–FITC and PI (Trevigen), according to the manufacturer's instructions, and then analyzed using FCM.16 In some experiments, B7-H1+ and B7-H1− F-36P cells prepared after labeling with anti–B7-H1–PE were cultured with lymphocytes. In this case, the cells were stained with anti-CD3–allophycocyanin (BD Biosciences) and with annexin V–FITC and PI. For caspase-3 analysis, the cells were first stained with FITC-conjugated antibodies to CD3, CD4, or CD8, washed, and then stained for the active form of caspase-3 using an FCM kit (BD Biosciences).
Mixed lymphocyte–blast reaction
CD3+, CD4+, and CD8+ T cells were purified from PB from healthy volunteers using magnetic cell sorting (Miltenyi Biotec). The purity of the isolated cells was more than 95% as determined in FCM. Each of these cell populations (1 × 105 cells/well) was cocultured with irradiated (20 000 rad) MDS blasts, that is, either F-36P cells (whole cells or purified cells according to the status of B7-H1 expression) or patient blasts that were confirmed to express B7-H1, in 96-well U-bottomed plates for 5 days. Antagonistic anti–B7-H1 (eBioscience) or anti–PD-1 (R&D Systems) antibody was added to the culture to block the B7-H1–PD-1 pathway. These mixed lymphocyte–blast reaction (MLR) cultures were subjected to 2 types of assay. To determine T-cell proliferation, 3H-thymidine (1 μCi/well) was added to the wells during the last 18 hours of culture, and its incorporation into T cells was measured. To determine T-cell apoptosis, the cells were subjected to apoptosis analysis after 5-day MLR culture.
Morphometric analysis
Cytospin preparations were prepared using B7-H1+ and B7-H1− blasts isolated by FACS and stained with Wright-Giemsa. Their images were captured with an Olympus AX-80 microscope connected to a digital camera (DP50; Olympus), and diameters of the nucleus and cytoplasm were measured using analysis software (Lumina Vision; Mitani Corportion). One hundred cells were analyzed for each cell fraction. Forward and side light scatter values in FCM for B7-H1+ and B7-H1− blasts, which reflect cell size and cytoplasmic granularity, respectively, were also analyzed. Only samples in which data on more than 100 events were available for both blast populations were used for analysis.
Statistical analysis
Differences between 2 groups of data were determined using Student t test for continuous variables and the χ2 test for categorical variables. A P value less than .05 was considered to represent a statistically significant difference.
Results
B7-H1 expression in MDS cell lines
First, the expression of B7-H1 molecules was examined in 3 MDS cell lines cultured without any specific stimulation of B7-H1 (Figure 1A,B left and middle panels). A high level of B7-H1 expression at the mRNA and protein levels was detected in F-36P cells. OIH-1 and SKM-1 cells expressed less and an undetectable amount of B7-H1 mRNA, respectively, and B7-H1 protein was not detectable on their cell surface. Next, we investigated whether cytokines that may be associated with MDS pathophysiology (ie, IFNγ, IL-1β, TGFβ, and TNFα) induced B7-H1 expression in these cells. IFNγ, which is known to induce B7-H1 expression in antigen-presenting cells from healthy subjects, enhanced or induced B7-H1 expression in all 3 cell lines (right panels of Figure 1B-C). Notably, the combination of IFNγ and TNFα induced a much higher level of B7-H1 expression than IFNγ alone in the F-36P and SKM-1 cell lines.
Constitutive or inducible B7-H1 expression in MDS cell lines.(A) B7-H1 mRNA expression analyzed by reverse-transcription polymerase chain reaction. Equal amounts of cDNA from 3 myelodysplastic syndromes (MDS) cell lines were amplified. Vertical lines have been inserted to indicate a repositioned gel lane. (B) B7-H1 expression analyzed by flow cytometry (FCM). The cell lines were cultured with medium alone or with IFNγ. Thick line histogram represents staining with anti–B7-H1 monoclonal antibodies; dotted line histogram represents staining with isotype-matched control Ig. (C) Effects of cytokines on B7-H1 expression in F-36P and SKM-1 cells. Data (relative fluorescence intensity [MFI]) are mean and SD of 3 experiments. Results without SD error bar indicate small SD.
Constitutive or inducible B7-H1 expression in MDS cell lines.(A) B7-H1 mRNA expression analyzed by reverse-transcription polymerase chain reaction. Equal amounts of cDNA from 3 myelodysplastic syndromes (MDS) cell lines were amplified. Vertical lines have been inserted to indicate a repositioned gel lane. (B) B7-H1 expression analyzed by flow cytometry (FCM). The cell lines were cultured with medium alone or with IFNγ. Thick line histogram represents staining with anti–B7-H1 monoclonal antibodies; dotted line histogram represents staining with isotype-matched control Ig. (C) Effects of cytokines on B7-H1 expression in F-36P and SKM-1 cells. Data (relative fluorescence intensity [MFI]) are mean and SD of 3 experiments. Results without SD error bar indicate small SD.
Involvement of NF-κB activation in B7-H1 induction in MDS cells
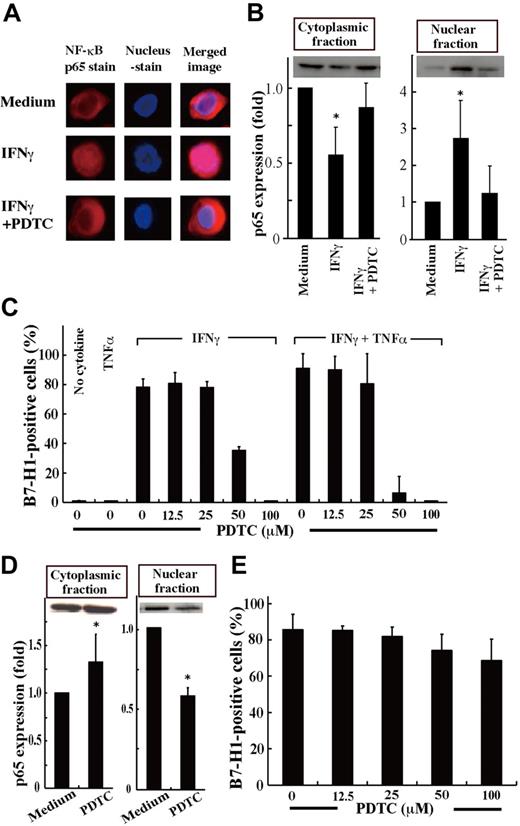
IFNγ and TNFα activate NF-κB in human cells,32-34 and the promoter region of the human B7-H1 gene has an NF-κB motif.35 NF-κB activation was also observed in MDS patients, in particular in patients with advanced disease.36,37 Therefore, we next investigated whether B7-H1 induction by IFNγ and TNFα in MDS cells is mediated by NF-κB activation. When SKM-1 cells were treated with IFNγ, the nuclear translocation of NF-κB p65, indicating NF-κB activation, was observed, and was nearly completely blocked by the NF-κB inhibitor, PDTC (Figure 2A-B). Furthermore, B7-H1 induction by IFNγ and TNFα in SKM-1 cells was completely blocked by PDTC (Figure 2C). When F-36P cells that constitutively express B7-H1 molecules were examined, NF-κB was also constitutively activated (strong Western blot band in the nuclear fraction; Figure 2D). When using PDTC, NF-κB activation and B7-H1 expression were partially inhibited in F-36P cells (Figure 2D-E).
Inhibition of NF-κB activation down-regulated B7-H1 expression.(A) SKM-1 cells cultured for 24 hours with medium alone, IFNγ, or IFNγ and pyrrolidine dithiocarbamate (PDTC; 100μM) were stained for NF-κB p65 (red) and nuclei (blue). The merged image showed that IFNγ induced p65 localization into the nucleus, which was inhibited by PDTC. Images were acquired on an Olympus IX71 inverted microscope using a LCPlanFI 40×/0.60 objective lens. Alexa 594 and 4'-6-diamidino-2-phenylindole were detected using Fluorescence Miler Units of U-MWIG2 and U-MNUA2 (Olympus), respectively. Images were captured using a 3CCD digital color camera (Hamamatsu Photonics) through Aquacosmos Software. (B) After the cultures in A, SKM-1 cells were separated into cytoplasmic and nuclear fractions, and their p65 contents were analyzed by Western blotting to show representative (top panel) and quantified (bottom panel) data (mean + SD) of 3 experiments. In each experiment, the band intensity of cell fractions from SKM-1 cells cultured with medium alone was defined as 1. *P = .017 and .06 (left panel) and *P = .045 and .012 (right panel) compared with “the medium” and IFNγ and PDTC, respectively. (C) SKM-1 cells cultured with or without cytokine(s) and various concentrations of PDTC were analyzed for B7-H1 expression by FCM. (D-E) Using F-36P cells expressing B7-H1 constitutively, the same experiments as in panels B and C were performed, except that no cytokines were used. Data are from 3 experiments. *P = .015 for D (left panel) and *P < .0001 (right panel) compared with the medium.
Inhibition of NF-κB activation down-regulated B7-H1 expression.(A) SKM-1 cells cultured for 24 hours with medium alone, IFNγ, or IFNγ and pyrrolidine dithiocarbamate (PDTC; 100μM) were stained for NF-κB p65 (red) and nuclei (blue). The merged image showed that IFNγ induced p65 localization into the nucleus, which was inhibited by PDTC. Images were acquired on an Olympus IX71 inverted microscope using a LCPlanFI 40×/0.60 objective lens. Alexa 594 and 4'-6-diamidino-2-phenylindole were detected using Fluorescence Miler Units of U-MWIG2 and U-MNUA2 (Olympus), respectively. Images were captured using a 3CCD digital color camera (Hamamatsu Photonics) through Aquacosmos Software. (B) After the cultures in A, SKM-1 cells were separated into cytoplasmic and nuclear fractions, and their p65 contents were analyzed by Western blotting to show representative (top panel) and quantified (bottom panel) data (mean + SD) of 3 experiments. In each experiment, the band intensity of cell fractions from SKM-1 cells cultured with medium alone was defined as 1. *P = .017 and .06 (left panel) and *P = .045 and .012 (right panel) compared with “the medium” and IFNγ and PDTC, respectively. (C) SKM-1 cells cultured with or without cytokine(s) and various concentrations of PDTC were analyzed for B7-H1 expression by FCM. (D-E) Using F-36P cells expressing B7-H1 constitutively, the same experiments as in panels B and C were performed, except that no cytokines were used. Data are from 3 experiments. *P = .015 for D (left panel) and *P < .0001 (right panel) compared with the medium.
Proliferative advantage in B7-H1–expressing MDS cell lines
Based on previous studies,9,31 we speculated that B7-H1+ MDS blasts might have a proliferative advantage over B7-H1− MDS blasts. Therefore, we compared the cell cycle between B7-H1+ and B7-H1− cell fractions in F-36P and SKM-1 cells. The latter cells were cultured with IFNγ for 2 days to induce B7-H1 expression before analysis. In both cell lines, B7-H1+ cells had fewer G0/G1 phase cells and more S and G2/M phase cells compared with B7-H1− cells. All the results showed statistically significant differences between the positive and negative cells, except for S phase–cell data in F-36P cells (Figure 3A-B). Consistent with these data, the B7-H1+ cell population incorporated more BrdU compared with the B7-H1− cell population in F-36P and SKM-1 cells (Figure 3C). Next, when purified B7-H1+ and B7-H1− F-36P cells were subjected to the colony-forming assay using methylcellulose, the former formed more colonies compared with the latter cells (Figure 3D). Furthermore, when the expression of cyclin genes was compared between B7-H1+ and B7-H1− F-36P cells, cyclin D1, D2, and D3 mRNA levels were higher in the former cells compared with the latter (supplemental Figure 1).
Cell proliferative potential as a function of B7-H1 expression in MDS cell lines. (A-B) Cell-cycle analysis of B7-H1+ and B7-H1− cell fractions in F-36P (A) and SKM-1 (B) cells. SKM-1 cells were cultured with IFNγ for 2 days before analyses. (C) BrdU incorporation in B7-H1+ and B7-H1− cell fractions in F-36P and SKM-1 cells. (D) The number of colonies formed by purified B7-H1+ and B7-H1− F-36P cells in the methylcellulose culture. Data in panels A through C are mean (and SD) of 3 experiments. *P < .036 for B7-H1+ versus B7-H1− cell fractions.
Cell proliferative potential as a function of B7-H1 expression in MDS cell lines. (A-B) Cell-cycle analysis of B7-H1+ and B7-H1− cell fractions in F-36P (A) and SKM-1 (B) cells. SKM-1 cells were cultured with IFNγ for 2 days before analyses. (C) BrdU incorporation in B7-H1+ and B7-H1− cell fractions in F-36P and SKM-1 cells. (D) The number of colonies formed by purified B7-H1+ and B7-H1− F-36P cells in the methylcellulose culture. Data in panels A through C are mean (and SD) of 3 experiments. *P < .036 for B7-H1+ versus B7-H1− cell fractions.
Morphometric analysis showed that B7-H1+ cells were larger in cell and nuclear sizes and more granular in their cytoplasm compared with B7-H1− cells in both F-36P and SKM-1 cells (supplemental Table 2).
Effects of B7-H1–expressing MDS cell lines on normal T cells
When normal CD3+ T cells were cultured with or without irradiated F-36P cells, the presence of F-36P cells profoundly increased the percentages of T cells showing apoptosis (Figure 4A). The increase in T-cell apoptosis was accompanied by an increase in T cells with activated caspase-3 (Figure 4B). These findings were observed again when purified CD4+ and CD8+ cells were used instead of the entire CD3+ T-cell population. Furthermore, the apoptosis and caspase-3 activation in T cells induced by F-36P cells were inhibited when the blocking antibody for either B7-H1 or PD-1 was added to the cocultures (analyses of annexin V+PI+ cells showed essentially the same results; Figure 4A-B). Consistent with these findings, the T-cell proliferation in the above allogeneic coculture system was augmented when anti–B7-H1 or anti–PD-1 blocking antibody was added to the culture (Figure 4C). Finally, when purified B7-H1+ or B7-H1− F-36P cells were used instead of whole F-36P cells, the degree of T-cell apoptosis was higher in the B7-H1+ cell cultures than in the B7-H1- cell cultures (Figure 4D). These results thus indicate that B7-H1 molecules on F-36P cells increase T-cell apoptosis and decrease T-cell proliferation.
Effects of B7-H1 molecules expressed by blasts on T cells. (A-C) Purified, normal CD3+, CD4+, and CD8+ T cells were cultured alone or with irradiated F-36P cells in the presence of either control immunoglobulin G (IgG), anti–B7-H1 monoclonal antibody, or anti–PD-1 monoclonal antibody. Data are mean (and SD) of 3 experiments. P < .05 when data in each of the 2 columns on the right were compared with control IgG data. (A) The percentage of annexin V+ cells in each T-cell fraction. (B) The percentage of caspase-3+ cells in T cells. Data labeled control IgG were defined as 100% in each experiment. (C) T-cell proliferation determined in the 3H-thymidine incorporation assay. (D) Normal CD3+ T cells were cultured with irradiated F-36P cells that had been purified into either B7-H1+ or B7-H1− cells.
Effects of B7-H1 molecules expressed by blasts on T cells. (A-C) Purified, normal CD3+, CD4+, and CD8+ T cells were cultured alone or with irradiated F-36P cells in the presence of either control immunoglobulin G (IgG), anti–B7-H1 monoclonal antibody, or anti–PD-1 monoclonal antibody. Data are mean (and SD) of 3 experiments. P < .05 when data in each of the 2 columns on the right were compared with control IgG data. (A) The percentage of annexin V+ cells in each T-cell fraction. (B) The percentage of caspase-3+ cells in T cells. Data labeled control IgG were defined as 100% in each experiment. (C) T-cell proliferation determined in the 3H-thymidine incorporation assay. (D) Normal CD3+ T cells were cultured with irradiated F-36P cells that had been purified into either B7-H1+ or B7-H1− cells.
B7-H1 molecules on blasts from MDS patients
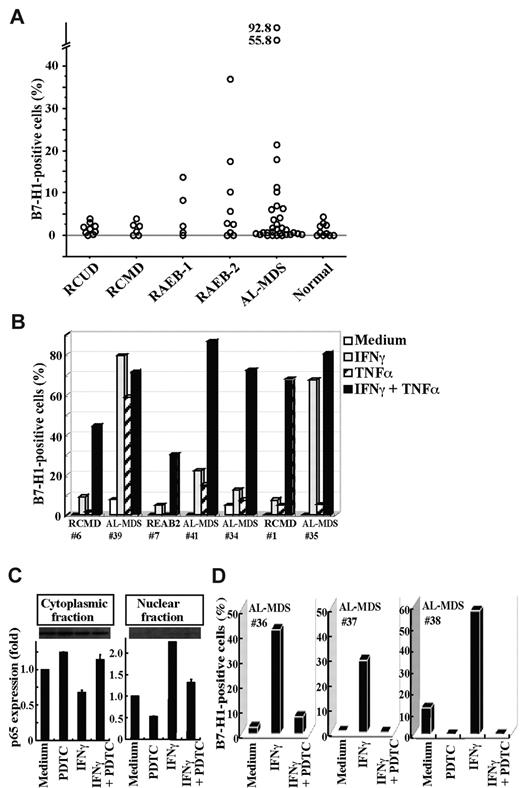
We then examined B7-H1 expression on BM blasts from patients with MDS or AL-MDS and from hematologically normal subjects (Figure 5A). B7-H1 molecules were expressed in more than 5% of blasts in a substantial proportion of patients with refractory anemia with excess blasts (RAEB) and AL-MDS, but not in any patients with refractory cytopenia with unilineage dysplasia (RCUD) and refractory cytopenia with multilineage dysplasia (RCMD) or in normal subjects. When patients were divided into 2 groups according to the IPSS category, a higher proportion of blasts expressed B7-H1 molecules in patients in the intermediate-2 or high category than in patients in the low or intermediate-1 category (mean ± SD, 6.5% ± 8.4% [n = 25] vs 1.4% ± 1.3% [n = 15], P = .0239). Furthermore, when BM cells from patients were cultured with cytokines, B7-H1 expression on blasts was induced or enhanced by stimulation with IFNγ or TNFα, and most strikingly by their combination (Figure 5B). This B7-H1 induction in MDS blasts from patients was mediated by NF-κB activation, as seen in MDS cell lines. The nuclear translocation of NF-κB p65, indicating NF-κB activation, was induced by IFNγ and was nearly completely blocked by an inhibitor for NF-κB, PDTC (Figure 5C). Furthermore, B7-H1 expression induced by IFNγ on patient blasts was nearly completely blocked by PDTC (Figure 5D).
B7-H1 expression on patient blasts. (A) The percentages of B7-H1+ cells in blasts from freshly isolated bone marrow samples. (B) The induction of B7-H1 expression on blasts from 7 patients. The cells had been cultured for 2 days with IFNγ and/or TNFα. (C) Purified CD34+ blasts from a patient (no. 38) were incubated with no additive (Medium), PDTC (100μM), IFNγ, or IFNγ and PDTC. After incubation, the blasts were separated into cytoplasmic and nuclear fractions. The NF-κB p65 contents in each fraction were analyzed using Western blotting to show representative (top panel) and quantified (bottom panel) data (mean + SD) of 2 experiments. In each experiment, the band intensity of cell fractions from blasts that had been cultured with no additive was defined as 1. (E) The inhibitor of NF-κB, PDTC, blocked B7-H1 induction by IFNγ on patient blasts. Purified CD34+ blasts from patients were cultured as indicated, and their B7-H1 expression was analyzed.
B7-H1 expression on patient blasts. (A) The percentages of B7-H1+ cells in blasts from freshly isolated bone marrow samples. (B) The induction of B7-H1 expression on blasts from 7 patients. The cells had been cultured for 2 days with IFNγ and/or TNFα. (C) Purified CD34+ blasts from a patient (no. 38) were incubated with no additive (Medium), PDTC (100μM), IFNγ, or IFNγ and PDTC. After incubation, the blasts were separated into cytoplasmic and nuclear fractions. The NF-κB p65 contents in each fraction were analyzed using Western blotting to show representative (top panel) and quantified (bottom panel) data (mean + SD) of 2 experiments. In each experiment, the band intensity of cell fractions from blasts that had been cultured with no additive was defined as 1. (E) The inhibitor of NF-κB, PDTC, blocked B7-H1 induction by IFNγ on patient blasts. Purified CD34+ blasts from patients were cultured as indicated, and their B7-H1 expression was analyzed.
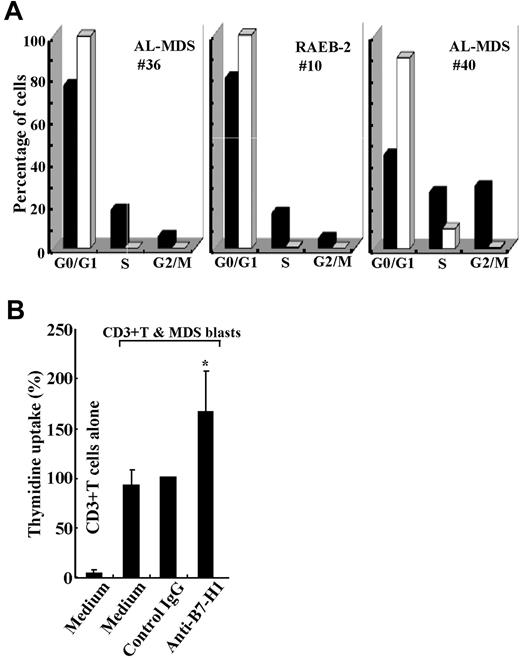
Next, the cell cycles of B7-H1+ and B7-H1− blasts were compared in 3 patients. As observed in the MDS cell lines, the percentages of G0/G1 phase cells were lower and those of S and G2/M phase cells were higher in B7-H1+ blasts than in B7-H1− blasts from all patients examined (Figure 6A). Consistent with these data, B7-H1+ blasts incorporated more BrdU compared with B7-H1− blasts in all 3 patients examined (11.1% vs 5.7%, 4.6% vs 0.5%, and 11.2% vs 0.9% in 1 RAEB-2 and 2 AL-MDS patients, respectively). It was also observed that Ki-67 expression was higher in B7-H1+ blasts than in B7-H1− blasts in all 3 patients examined (15.9% vs 11.5%, 83.4% vs 57.5%, and 39.7% vs 33.5% in 1 RCUD, 1 RAEB-2, and 1 AL-MDS patient, respectively). Contrary to the cell lines, cell and nuclear sizes did not differ between B7-H1+ and B7-H1− blasts, although B7-H1+ blasts were more granular in their cytoplasm (supplemental Table 2). When blasts from 3 patients, in who B7-H1 expression had been induced by IFNγ and TNFα and then irradiated, were cultured with CD3+ T cells from healthy volunteers, T-cell proliferation was significantly augmented by blocking B7-H1 molecules (Figure 6B).
Functional aspects of B7-H1-expressing blasts from patients. (A) Cell cycle of purified CD34+ blasts as a function of B7-H1 expression. Freshly isolated blasts that markedly expressed B7-H1 molecules (from patient no. 36) and blasts cultured with IFNγ and TNFα to induce B7-H1 expression (from patient no. 10 and patient no. 40) were analyzed. B7-H1+ (■) and B7-H1− (□) cells are indicated. (B) B7-H1–expressing patient blasts were cultured with normal T cells in the presence of control IgG or anti–B7-H1 monoclonal antibodies or medium alone. Data are mean (and SD) of 3 experiments; in each experiment using a different patient sample, data labeled control IgG were defined as 100%. *P = .026 compared with control IgG.
Functional aspects of B7-H1-expressing blasts from patients. (A) Cell cycle of purified CD34+ blasts as a function of B7-H1 expression. Freshly isolated blasts that markedly expressed B7-H1 molecules (from patient no. 36) and blasts cultured with IFNγ and TNFα to induce B7-H1 expression (from patient no. 10 and patient no. 40) were analyzed. B7-H1+ (■) and B7-H1− (□) cells are indicated. (B) B7-H1–expressing patient blasts were cultured with normal T cells in the presence of control IgG or anti–B7-H1 monoclonal antibodies or medium alone. Data are mean (and SD) of 3 experiments; in each experiment using a different patient sample, data labeled control IgG were defined as 100%. *P = .026 compared with control IgG.
Finally, we investigated the expression of PD-1, a counter-receptor of B7-H1, on circulating T cells in patients with MDS or AL-MDS. PD-1 expression on CD3+, CD4+, and CD8+ T cells was significantly higher in patients than in hematologically normal subjects. Furthermore, PD-1 expression in patients was increased in T cells expressing HLA-DR, a marker of T-cell activation (supplemental Figure 2).
Discussion
This study showed that blasts from MDS patients in high-risk IPSS categories express B7-H1 molecules more often compared with blasts from other patients. B7-H1 expression on MDS blasts was induced by IFNγ and TNFα via NF-κB activation, and B7-H1+ MDS blasts had greater proliferative capacity than B7-H1− MDS blasts. Furthermore, B7-H1+ blasts suppressed T-cell proliferation and induced T-cell apoptosis. Finally, PD-1 expression levels on circulating T cells were higher in MDS patients, in particular in association with the increased HLA-DR expression on T cells, compared with those in healthy volunteers. The morphometric difference observed between B7-H1+ and B7-H1− MDS blasts may reflect a biologic difference between these 2 cell populations. We believe that B7-H1+ MDS blasts are clonal cells because we confirmed a cytogenetic aberration in B7-H1+ MDS blasts in 2 cases examined (data not shown) and because all patients in whom 5% or more blasts expressed B7-H1+ were in advanced disease stages in which most blasts are clonal.38
When T cells are activated, for example, by tumor-associated antigen, they express PD-1 molecules on their surface and produce IFNγ and TNFα.39 These cytokines suppress colony formation of normal hematopoietic cells and induce apoptosis in hematopoietic cells.40 In MDS patients, IFNγ and TNFα are often overproduced by PB cells41 and by macrophages, one of the stromal components, in the BM.42 These cytokines may also be associated with increased apoptosis of hematopoietic cells. It was also reported that an increased serum TNFα level was associated with leukemic transformation in MDS.43 Based on these findings and in conjunction with the data in the present study, it is speculated that overproduction of IFNγ and TNFα in the BM microenvironment induces B7-H1 expression on MDS blasts, leading to disease progression at least in some cases by inducing T-cell apoptosis as well as by their intrinsic linkage with a high proliferative potential.
It was reported that constitutive activation of NF-κB in MDS blasts was observed in patients in advanced IPSS categories.36 Moreover, this constitutive activation of NF-κB was associated with antiapoptotic features of blasts in those MDS patients.36 The present results suggest a new mechanism of NF-κB in MDS pathophysiology: by inducing B7-H1 expression on MDS blasts, NF-κB confers a growth advantage on the blasts. The finding that B7-H1+ MDS blasts had greater proliferative capacity than B7-H1− MDS blasts is interesting. Similarly, Ghebeh et al44 examined patient breast cancer cells and found that high B7-H1 expression was associated with high Ki-67 expression, a marker of cell proliferation. It has been accepted that B7-H1 acts as a ligand for PD-1 and thus transmits signals to PD-1–expressing cells. However, recent data from one of our groups have shown that the interaction between B7-H1 molecules on murine cancer cells (mastocytoma and renal cell carcinoma) and PD-1 molecules on T cells induces a bidirectional signal: the PD-1–mediated signal inducing T-cell apoptosis and B7-H1–mediated reverse signal inducing cancer cell resistance to apoptosis.45 Thus, one scenario is that there is a B7-H1–mediated reverse signal to MDS blasts that is associated with their growth advantage.
The inhibitory signal to T cells from the interaction between B7-H1 and PD-1 molecules plays a crucial role in regulating various immune responses. For example, appropriate T-cell regulation by this signal is important to suppress harmful autoimmunity as well as the success of allogeneic transplantation.46,47 Meanwhile, this inhibitory signal may be used by some types of cancer cell as one strategy to evade the host anticancer immune response. We conclude that this strategy is seen in at least some MDS patients to evade the anti-MDS immune response. We reported that T-cell apoptosis was increased in MDS and that its degree was particularly extensive in advanced disease stages and correlated with the degree of lymphopenia that is common in MDS.16 We speculate that the signal from B7-H1 molecules on MDS blasts to PD-1 molecules on T cells is one mechanism of the increased T-cell apoptosis observed in MDS patients.
In summary, to the best of our knowledge this is the first report examining the B7-H1 and PD-1 system in MDS in detail. Our results suggest that this system is involved in the pathophysiology in MDS, at least in some patients, by suppressing T-cell immunology and conferring a growth advantage on MDS blasts.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Acknowledgment
This work was supported in part by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (20591157).
Authorship
Contribution: A.K. performed experiments and drafted the manuscript; T.Y. and W.Z. performed experiments; H. Tamura conceived and designed the study, supported clinical aspects, and wrote the manuscript; T.T., M.S., E.S., and H. Takahashi supported technical aspects; K.T. and L.C. supported intellectual aspects; K.D. provided administrative and clinical support; and K.O. designed the study, supported clinical aspects, and wrote the manuscript.
Conflict-of-interest disclosure: The authors declare no competing financial interests.
Correspondence: Hideto Tamura, Division of Hematology, Nippon Medical School, 1-1-5 Sendagi, Bunkyo-ku, Tokyo 113-8603, Japan; e-mail: tam@nms.ac.jp.

![Figure 1. Constitutive or inducible B7-H1 expression in MDS cell lines.(A) B7-H1 mRNA expression analyzed by reverse-transcription polymerase chain reaction. Equal amounts of cDNA from 3 myelodysplastic syndromes (MDS) cell lines were amplified. Vertical lines have been inserted to indicate a repositioned gel lane. (B) B7-H1 expression analyzed by flow cytometry (FCM). The cell lines were cultured with medium alone or with IFNγ. Thick line histogram represents staining with anti–B7-H1 monoclonal antibodies; dotted line histogram represents staining with isotype-matched control Ig. (C) Effects of cytokines on B7-H1 expression in F-36P and SKM-1 cells. Data (relative fluorescence intensity [MFI]) are mean and SD of 3 experiments. Results without SD error bar indicate small SD.](https://ash.silverchair-cdn.com/ash/content_public/journal/blood/116/7/10.1182_blood-2009-12-255125/4/m_zh89991055860001.jpeg?Expires=1767719719&Signature=dTCwbX2xCOUihD~ofYOwdQPdjsCPI0MvGrSnPraW~rEB99KHik5960XLoq6QL7z1MKhbbDAQFULhSI0ODmQFfCsKmnToFJCCkH5euVTLhz4gjZQ5guWRBLJv2opPMmdmz5bsuMOVjpK6I9sRkq~TQBTnF9Uc1tZXptmsmExEeAmy1cis6yKvRa0PH95V~0~EuxBc2sQf-THlAJt9NuNvSRl6Kgf3WhgNgH3dY71D1tpbs3VefPAMJjgyrdDPXrLvzA02HQN6uHuLzu7-i3LwOqn0MbQArqJvksWJ~8mZWFQ6BqoOjZhIshh26kNoJrqiSNW24Tl4BWF3PM-XDGoJjQ__&Key-Pair-Id=APKAIE5G5CRDK6RD3PGA)


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal