Abstract
Abstract 2911
AZA gives 50–60% responses and improves OS in higher-risk MDS but its role in t MDS/AML, characterized by a high frequency of unfavorable karyotypes and poor response to available treatments, remains unknown.
An AZA compassionate program (ATU) was opened in France between Dec 2004 and Jan 2009 for higher risk MDS, and for AML not candidates to or refractory to intensive chemotherapy (IC). We analyzed t MDS/AML included in this program and having received at least 1 cycle of AZA, excluding patients (pts) previously treated with chemotherapy (CT) for their tMDS/AML.
60 tMDS/AML were included: M/F:24/36, median age 69 (range 20–87). Primary disease was Breast Carcinoma (n=17), CLL (n=6), Hodgkin's disease (n=4), NHL (n=7), ALL (n=2), ovarian (n=2), liver(n=2), prostate carcinoma (n=2), other cancers (n=16), immunosuppressive therapy for lupus or Lung transplant in the last 2 pts. Treatment of primary neoplasm consisted in C×T in 86% and R×T in 61%.
Diagnosis (WHO) was RCMD+/− RS in 4 pts, RAEB-1 in 10 pts, RAEB-2 in 28 pts MDS with myelofibrosis in 1 pt, and AML in 17 pts (including 12 FAB RAEBt). IPSS cytogenetics were fav in 4 (6.7%), int in 11 (18.3%), and unfav in 45 (75%, including 71% Complex karyotypes).
Patients received a median of 4 cycles (range 1–24) of AZA, at FDA/EMEA-approved schedule (75 mg/m2/d ×7d/4 week) in 73% patients and a less intensive schedule (5d/4w, or <75mg/m2/d) in 27% patients. 25 (41.6%) pts received < 4 cycles, mainly due to early death (32%), failure (28%) or hematological toxicity (16%). Best response according to IWG 2006 criteria was CR in 9 pts (15%), marrow CR (mCR) in 8 (13%), PR in 1, stable disease (SD) with HI in 5 (8%) ie an overall response rate (ORR) of 38%. In pts who received >4 cycles, ORR was 21/35 (60%). Age, WBC count, baseline platelet count, % marrow blasts, karyotype (whatever the categorization) and ECOG status had no impact on ORR.
1, 2 and 3 year survival was 35%, 15% and 7%. Female gender (p=0.02), achievement of response (p=0.003) and ECOG status (0-1 vs 2–4) (p=0.04) had significant impact on OS while karyotype and % marrow blasts had no impact. However, there was a trend for lower OS in patients with chrom 5 abn (2y 0S 7.7% vs 23 %, p=0.08).
9 (15%) patients, all females, median age 55 (range 35–66) were allografted after a median number of 6 AZA cycles (range 2–17), including 3 pts, 4 pts and 2 pts who had achieved CR, mCR and no response to AZA, respectively. 4 (44%) of them were alive after 5+,17+,32+ and 42+ months.
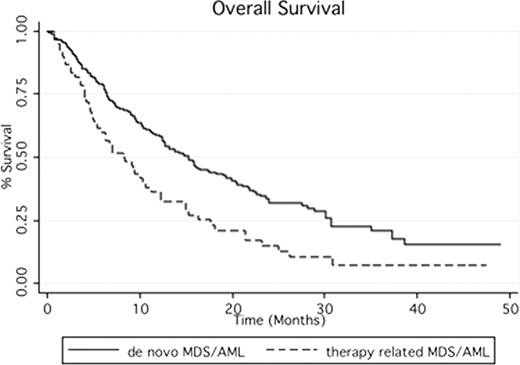
By comparison to the 232 pts with de novo MDS/AML included in the same program, tMDS/AML had a higher frequency of complex karyotype (71 vs 29.5, p<0.01), received fewer cycles of AZA (4 vs 6, p=0.0014) due to more frequent discontinuation of AZA before 4 cycles (41.6% vs 25%, p=0.025) mainly related to a higher incidence of early deaths. tMDS/AML had a similar response rate (38% vs 45% in de novo MDS/AML, p=0.67), but significantly shorter OS (2yOS 15% vs 33.3, p=0.003, figure 1).
In this cohort of tMDS/AML, survival with AZA was shorter than in de novo MDS/AML, probably largely due to their more severe baseline characteristics. Only 15% of tMDS/AML could be allografted, half of them with so far a favorable outcome.
Fenaux:CELGENE, JANSSEN CILAG, AMGEN, ROCHE, GSK, NOVARTIS, MERCK, CEPHALON: Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal