Abstract
Abstract 2520
Immune thrombocytopenia (ITP) is typically characterized by increased platelet destruction and reduced platelet production. Eltrombopag is a thrombopoietin receptor (TPO-R) agonist that is known to increase platelet counts in patients with ITP by stimulating thrombopoiesis, but it is unknown whether it has any effect on platelet life span. Platelet survival is mediated by two molecular intermediates in an apoptotic pathway, Bcl-xL and Bak. Bcl-xL protein expression in megakaryocytes is thought to be regulated in part by TPO-mediated activation of Akt pathways through Jak2 and Stat5. Although controversial, in a very small number of ITP patients, Eltrombopag may increase platelet counts in 2–5 days. We hypothesized that any increase in platelet count in the first week of treatment might be due to effects of Eltrombopag on platelet survival. Therefore, this study explored whether Eltrombopag treatment has anti-apoptotic effects in patients with ITP.
Following a treatment wash out period, 75 mg of Eltrombopag once daily was initiated for 2 weeks. Blood counts were measured on days 1, 3, 5, 8, 10, 12, and 15. Platelet function and survival was assessed on days 1, 8, and 15 by: immature platelet fraction (IPF); glycocalicin index; Bcl-xL inhibitor (ABT-737) IC50, a novel assay adapted for human platelets; measurement of Bcl-xL by western blot; measurement of several members of the Bcl-xL Akt mediated, apoptotic pathway by flow cytometry (FACS); bleeding score; measurement of thrombin-anti-thrombin complexes (TATs); and quantification of microparticles.
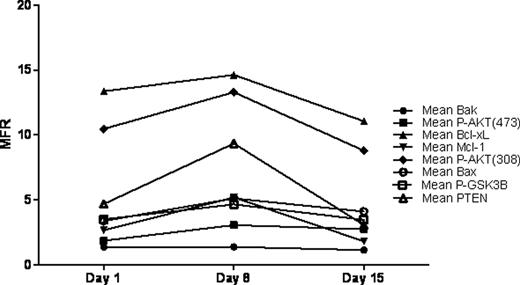
Seven of the 9 patients responded to treatment with Eltrombopag with a platelet count ≥ 50,000/μL, and 6 of the 7 responders at least doubled their counts during the 2 weeks of treatment. There was a significant increase in median platelet count (p<0.001), median large platelet count (p<0.01), and median absolute IPF (A-IPF, p<0.01), while there was a significant decrease in median % IPF (p<0.05). The dose of ABT-737 required to kill half of the platelets in the sample (IC50) did not differ significantly between patients or between patients and controls during the first two weeks of treatment, and remained stable over the 2 weeks of study. However, the relative IC50 values (% of day 1 IC50 value) increased after the first week of treatment but returned to baseline after the second week. While these changes were not significant, their kinetics were similar to those seen in the AKT/ Bcl-xL signal transduction pathway (Figure). There was no significant correlation between the platelet counts and the IC50 values. FACS analysis of members of the AKT signal transduction pathway revealed significant increase in each of the markers between days 1 and 8 (p<0.01), followed by a significant decrease between days 8 and 15 (p<0.05), with no difference between days 1 and 15 (Figure). The other lab tests are pending.
Mean of the median fluorescence ratios (MFR) for components of the AKT/Bcl-xL signal transduction pathway during the first two weeks of Eltrombopag treatment.
Mean of the median fluorescence ratios (MFR) for components of the AKT/Bcl-xL signal transduction pathway during the first two weeks of Eltrombopag treatment.
Because the A-IPF increased by less than the platelet increase and because the life span of the A-IPF is not known, it is unclear if the overall platelet count increase is entirely a result of increased platelet production. Platelet life span may be enhanced by Eltrombopag treatment as there was a parallel albeit transient increase in AKT activation markers and platelet apoptosis resistance. Our data suggest that platelets are more resistant to apoptosis when the levels of anti-apoptotic factors (eg. PTEN, Phospho-GSK3β) involved in the AKT/Bcl-xL pathway are greatest despite a concomitant increase in pro-apoptotic factors (eg. Bak, Bax). Since both increased AKT activation and apoptotic resistance returned to baseline at day 15, megakaryocytes and platelets already present at the start of treatment may respond differently than those generated de novo in the presence of Eltrombopag.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal