Abstract
Abstract 1906
Lenalidomide is an immunomodulatory agent that interacts with different components of the immune system by altering cytokine production, regulating T cells and increasing NK cell cytotoxicity. In multiple myeloma (MM), lenalidomide is approved for use in combination with dexamethasone in patients who have received at least one prior therapy. Recent observations have shown that dexamethasone enhances the anti-myeloma effect of lenalidomide; however, dexamethasone may also antagonize the immunomodulatory properties of lenalidomide.
In the present study we evaluated by multiparameter flow cytometry (MFC) peripheral blood (PB) T, NK and dendritic (plasmacytoid, myeloid and monocytic) cells (DC) from high-risk smoldering MM (SMM) patients, defined by the presence of at least 2 of the 3 following criteria at diagnosis: bone marrow plasma cell (BMPC) infiltration ≥10%; and/or high M-component (IgG≥30g/L or IgA≥20g/L or B-J Protein>10g/L); and/or ≥95% myelomatous-PC/BMPC and immune paresis. SMM patients were treated according to the QuiReDex trial (NCT 00480363): an induction phase of nine four-week cycles of lenalidomide plus dexamethasone (LenDex) followed by maintenance with lenalidomide until disease progression. In this ongoing study, immunophenotypic data is available in 53 patients at diagnosis (baseline), 30 after 3 cycles of LenDex and 22 at the end of induction therapy (9th cycle). Here we will focus on the 22 cases with information at the 3 time points. For MFC analysis, PB samples were stained using a four-color direct immunofluorescence technique that allowed the quantification and characterization of T, NK and DC cells, including cell cycle analysis.
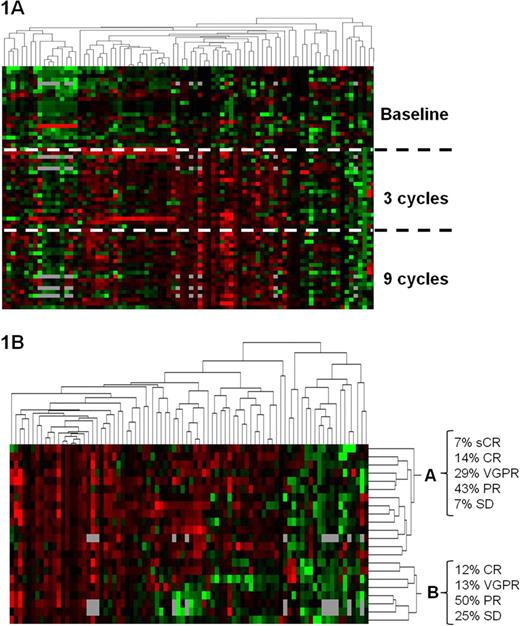
The percentage of PB T cells in total PB cellularity was stable from baseline vs 3 vs 9 cycles of LenDex (22% vs 21% vs 21%; respectively, NS), with similar results also obtained for T CD4 (12% vs 11% vs 9%; respectively, NS) and T CD8 (8% vs 6% vs 8%; respectively, NS) cells. NK cells were slightly increased after 9 cycles of LenDex for both the CD56dim (4.1%, 3.4% and 6%; respectively; NS) and CD56bright (0.05%, 0.04% and 0.15%; respectively; NS) NK cell compartments. Similarly, the percentage of DC slightly increased along treatment, especially for plasmacytoid DC (0.2% at baseline vs 0.4% after 9 cycles; p=0.09). However, when a more detailed immunophenotypic characterization of T and NK cells was carried out significant differences emerged following LenDex treatment (Figure 1A). Accordingly, after 3 and 9 cycles of LenDex both T CD4 and CD8 cells showed increased expression of activation markers such as CD69 (p=.03), CD25 (p=.02 and NS, respectively), CD54 (p<.001), CD28 (p≤.03) and CD120b (p≤.01), together with increased production of IFNγ (p=.03) and IL-2 (p=.1 and p=.008, respectively). Interestingly, after induction therapy an up-regulation of chemokine receptors related to the Th1 (CCR5; p<.001) but also Th2 (CCR4; p≤.002) immune response was detectable in CD4 and CD8 T cells. T CD4 cells displayed a clear maturation into a central memory phenotype following LenDex treatment (38% at baseline vs 50% and 66% at 3 and 9 cycles, respectively; p<.001) while T CD8 cells displayed an increased effector memory phenotype (44% vs 59% vs 62%; p=.004). Further analysis showed increased expression of HLA-DR (p≤.008), the antibody-dependent cell-mediated cytotoxicity associated receptor CD16 (p≤.03), and the adhesion molecules CD11a (p’.006) and CD11b (p≤.004) both on NK (CD56dim and CD56bright) and T cells. No consistent changes were observed in other NK cell receptors, such as CD94 and the immunoglobulin like receptors CD158a, CD161, NKB1 (3DL1) and NKAT2 (2DL3). Concerning cell cycle analysis, the percentage of cells in S-phase was significantly increased from baseline vs 3 vs 9 cycles of LenDex in T CD4 (0.05% vs 0.15% vs 0.16%; p<.001), CD8 (0.05% vs 0.11% vs 0.23%; p<.001) and NK cells (0.09% vs 0.17% vs 0.20%; p=.001). Finally, an unsupervised cluster analysis of the overall immunophenotypic profile obtained after 9 cycles of LenDex (Figure 1B) was able to discriminate two groups of patients (A and B). Interestingly, within the group with higher activation profile (A) 50% of patients achieved ≥VGPR vs 23% in group B (p=.2).
In summary, these preliminary results show that in high risk SMM patients the combination of lenalidomide and dexamethasone modulates PB T and NK cells, with increased activation status that may contribute to disease control.
Off Label Use: Lenalidomide is not approved for the treatment of smoldering multiple myeloma. De La Rubia:Janssen-Cilag: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees. Rosiñol:Celgene: Honoraria. Oriol:Celgene: Consultancy; Janssen-Cilag: Consultancy; Novartis: Consultancy. Hernández:Celgene: Honoraria. de Arriba:Janssen-Cilag: Honoraria; Celgene: Honoraria. Mateos:Celgene: Honoraria. San Miguel:Janssen-Cilag: Honoraria; Celgene: Honoraria, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal