Abstract
Abstract 3452
Poster Board III-340
Chronic Lymphocytic Leukemia (CLL) is incurable with current chemotherapy treatments. Epigallocatechin 3 gallate (EGCG), the major catechin in green tea, has been shown to induced caspase-dependent death CLL B-cells and down regulate anti-apoptotic proteins (Mcl-1; XIAP) known to increase the resistance of CLL B-cells to apoptosis (Blood 104:788-94). In Phase I testing for patients with early stage CLL (Rai 0-II), EGCG treatment was well tolerated and induced a decline in absolute lymphocyte count (ALC) and/or lymphadenopathy in the majority of treated patients without myelosuppression (JCO 27:3808-14). Although peak plasma EGCG levels were not measured in this study, trough plasma EGCG levels as high as 4 μg/ML were achieved. To further our understanding of the potential clinical applications of EGCG for patients with CLL, we evaluated the effects of EGCG on the viability of CLL B-cells when combined with fludarabine (F), chlorambucil (C), or fludarabine and chlorambucil in combination (FC).
Primary CLL B-cells were treated with various doses of F (0.25-1 μM), C (10-30 μM), or FC either alone or in combination EGCG (50 to 100 μM). In other experiments, CLL B-cells were treated with various doses of F (0.25-1 μM), C (10-30 μM), or FC with or without a fixed dose of EGCG known to be physiologically achievable in vivo (4 μM). After 48 hours (experiments with C +/− EGCG) or 72 hours (experiments with F +/− EGCG or FC +/− EGCG), cells were harvested, stained with annexin/PI and viability analyzed by flow cytometry. After concentration-effect curves were generated for each agent, data were also analyzed using the CalcuSyn software program (Biosoft, Cambridge, UK) which uses the method of Chou and Talalay to determine whether combination treatment yielded greater effects than expected from summation alone. A combination index (CI) of 0.8 – 1.2 indicates an additive effect, a CI >1.2 indicates an antagonistic effect and a CI <0.8 indicates a synergistic effect.
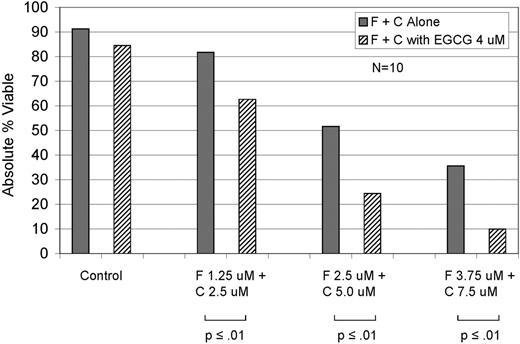
Primary leukemic B-cells from 56 CLL patients were cultured in vitro with various doses of EGCG alone or in combination with F, C, or FC. The median LD50 was approximately 100 μM. Although % apoptotic cells at the 100 μM EGCG dose did not vary based on Rai stage, ZAP-70 status, IGHV gene mutation status, or cytogenetic abnormalities by FISH, CD38 negative patients had greater cell death than CD38 positive patients (67% vs. 49%; p=0.05). In co-titration experiments, EGCG had an additive (CI 0.8 – 1.2) or synergistic (CI <0.8) effect on apoptosis when combined with C in the majority of patients (13/15; additive 11, synergism 2) with rare individuals demonstrating antagonism (CI >1.2; 2/15). The effects of EGCG when combined with F in co-titration experiments were more variable with a relatively even distribution between antagonism (8/18), additive effects (4/18), and synergy (6/18). However, co-titration experiments of FC with or without EGCG demonstrated additive or synergistic effect in most patients (8/10; additive 7, synergism 1). Next we evaluated the effect of F (0.25 - 1 μM), C (10 - 30 μM), or FC with or without a fixed dose of EGCG known to be physiologically achievable in vivo (4 μM). This dose of EGCG had an additive or synergistic effect in the majority of samples across the spectrum of dose levels under all 3 conditions (F, C, and FC). For example, at the 10 μM dose of C the addition of 4 μM EGCG had an additive or synergistic effect in 11/14 patients (synergistic 6; additive 5). With F at the 2.5 μM dose level the addition of 4 μM EGCG had an additive or synergistic effect in 13/16 patients (synergistic 7, additive 6). The addition of 4 μM EGCG appeared particularly beneficial when given in combination with FC (Fig.). For example, at F 2.5 μM in combination with C 5 μM the addition of 4 μM EGCG had an additive or synergistic effect in 10/10 patients (synergism 7; additive 3).
Physiologically achievable doses of EGCG appear to enhance the efficacy of alkylating agents, purine nucleoside analogues, and alkylating agent/purine analogue combination therapy for the majority of CLL patients on in vitro testing. The favorable toxicity profile of EGCG and lack of myelosuppression with this agent in the phase I trial (JCO 27:3808-14) make it an attractive agent to test in combination with purine analogue and alkylator based chemo-immunotherapy for patients with CLL.
Kay:Genentech, Celgene, Hospira, Polyphenon Pharma, Sanofi-Aventis: Research Funding; Biogenc-Idec, Celgene, Genentech, genmab: Membership on an entity's Board of Directors or advisory committees. Shanafelt:Genentech: Research Funding; Hospira: Membership on an entity's Board of Directors or advisory committees, Research Funding; Polyphenon Pharma : Patents & Royalties, Research Funding; Celgene: Research Funding; Cephalon: Research Funding; Bayer Health Care Pharmaceuticals: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal