Abstract
Abstract 2196
Poster Board II-173
Imatinib (IM) 400 mg daily is the standard treatment for Chronic Myeloid Leukemia (CML) in early chronic phase (ECP). The European LeukemiaNet (ELN) recommendations were designed to help identify ECP CML patients responding poorly to front-line IM, suggesting, at given time points, when the treatment strategy should be changed (”failure”), or when “the long-term outcome of the treatment would not likely be as favourable” (“suboptimal response”). Suboptimal response is a “grey zone”: the patient may still have substantial benefit from continuing IM, but other therapies should be considered.
To assess the outcome of “failure” and “suboptimal responders” Philadelphia-positive (Ph+) CML patients in a large multicentric, nationwide experience.
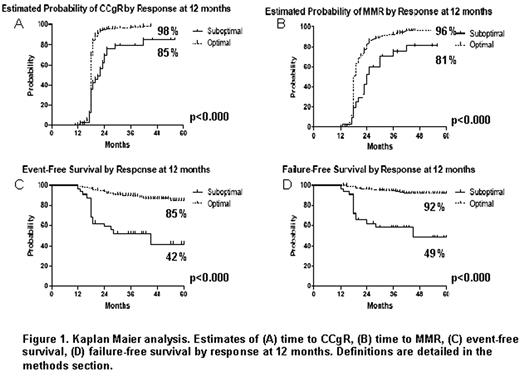
Between January 2004 and April 2007, 559 patients were enrolled in an observational study and in 2 independent intervention studies of the GIMEMA CML WP (Clin Trials Gov. NCT00514488 and NCT00510926). Response monitoring was based on conventional cytogenetic examination of bone marrow cell metaphases every 6 months and RT Q-PCR evaluations of blood cells after 3, 6, 12 months, and every 6 months thereafter. Definitions: major molecular response (MMR): BCR-ABL/ABL ratio < 0,1%IS; failure (according to ELN criteria): no hematologic response (HR) at 3 months, no complete HR (CHR) at 6 months, no cytogenetic response (CgR) at 6 months, no partial CgR (PCgR) at 1 year, no complete CgR (CCgR) at 18 months, loss CHR or CCgR, progression or death; suboptimal response (according to ELN criteria): no CHR at 3 months, no PCgR at 6 months, no CCgR at 12 months, no MMR at 18 months ; optimal response: non-suboptimal and non-failure at each time-point; event: failure or treatment discontinuation for any reason. All the calculations have been made according to the intention-to-treat principle.
The patients who fitted the ELN criteria for failure had a significantly lower probability of subsequently achieving a CCgR and a MMR, and had a significantly lower overall survival (OS), failure-free survival (FFS) and event-free survival (EFS). The patients who fitted the ELN definitions of suboptimal response at 6 months (data not shown) and at 12 months (figure 1) had a significantly lower probability than “optimal” responders of subsequently achieving a CCgR and a MMR, and a significantly poorer FFS and EFS (figure 1), while the OS was not different in the two groups (90% and 95%, p= 0.35). CONCLUSIONS Our data confirms that suboptimal responders at 6 and at 12 months have a poorer outcome with respect to “optimal” responders, comparable to the outcome of failure patients. Acknowledgments: European LeukemiaNet, COFIN, University of Bologna and BolognAIL.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal