Abstract
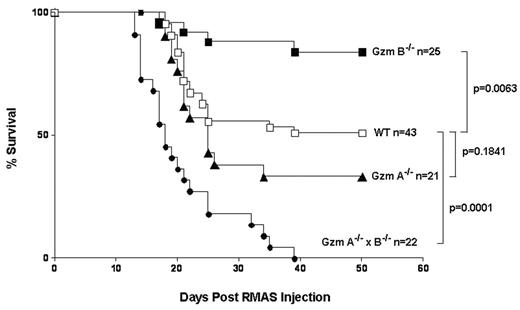
NK-dependent clearance of RMAS lymphoma and B16 melanoma (MHC Ilow) has been demonstrated in NK cell-deficient mice (Kim S et al, PNAS. 2000 Mar; 97(6): 2731–6). We recently investigated the roles of granzyme (Gzm) A and B in NK-dependent clearance of RMAS and B16 cells. The survival curves following intravenous injection of 2x105 RMAS cells are shown in the Figure; similar results were found with B16 cells. All granzyme AxB-deficient mice died within 6 weeks. Survival of granzyme A-deficient mice was similar to that of WT mice. Surprisingly, granzyme B-deficient mice were more resistant than WT mice to these tumor challenges. Previously, our laboratory demonstrated that human regulatory T (Treg) cells can use the granule exocytosis pathway to kill a variety of autologous immune cells in vitro (Grossman WJ et al, Immunity. 2004 Oct; 21(4): 589–601). Based on these results, we hypothesized that these tumor cell lines may induce granzyme B expression in Tregs, which in turn suppress the function of the NK cells responsible for clearing the tumors. Indeed, flow cytometric studies revealed that granzyme B (but not granzyme A) was highly expressed in 10–30% of CD4+/FoxP3+ Tregs found in the tumor environment (i.e. ascites fluid, tumor-infiltrated livers, or lungs). In contrast, very few granzyme B-expressing cells (<1%) were detected in the Tregs found in the peripheral spleens or lymph nodes of tumor-bearing mice or in the spleens, livers and lungs of naive mice. Within the NK cell compartment, granzyme A expression was constitutive, and it was persistently expressed following tumor challenge. While granzyme B was detected in only 1–5% of NK cells in the spleens, livers and lungs of naive mice, 20–40% of NK cells in the tumor environment expressed granzyme B following tumor challenge. To determine whether Tregs require granzyme B to suppress NK cell function in vivo, we purified Tregs (95% pure CD4+/CD25+ cells) from the resting spleens of both WT and granzyme B-deficient mice, and co-injected them intraperitoneally with 2x106 luciferase-tagged RMAS cells into granzyme B-deficient mice. Tumor burden was assessed using in vivo bioluminescence imaging. Add-back of 2x106 WT Tregs into granzyme B-deficient mice restored tumor growth to 40% of that observed at day 7 in WT mice, whose tumor burden was approximately 50-fold higher than that of granzyme B-deficient mice. Add-back of equal doses of granzyme B-deficient Tregs had no effect on tumor burden. Our data suggest that these tumor cell lines induce the expression of granzyme B in Tregs, which subsequently suppress tumor-specific NK cells. These data demonstrate for the first time that granzyme B plays a crucial role in Treg-mediated suppression of NK cell function in vivo. By recruiting and activating Tregs that inhibit NK cell function, these tumors gain a survival advantage that is lost in the setting of granzyme B deficiency in the Tregs. This study therefore suggests that targeted inhibition of granzyme B in Tregs may represent a novel approach to break Treg-mediated tumor tolerance.
Disclosures: Timothy J. Ley holds the license for the anti-mouse granzyme A monoclonal antibod (clone3G8.5) in conjunction with Santa Cruz Bioechnology, Inc.
Author notes
Corresponding author

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal