Abstract
Allogeneic hematopoietic stem cell transplantation (HSCT) is the only proven curative therapy for juvenile myelomonocytic leukemia (JMML). We, the European Working Group on Childhood MDS (EWOG-MDS) and the European Blood and Marrow Transplantation (EBMT) Group, report the outcome of 100 children (67 boys and 33 girls) with JMML given unmanipulated HSCT after a preparative regimen including busulfan, cyclophosphamide, and melphalan. Forty-eight and 52 children received transplants from an HLA-identical relative or an unrelated donor (UD), respectively. The source of hematopoietic stem cells was bone marrow, peripheral blood, and cord blood in 79, 14, and 7 children, respectively. Splenectomy had been performed before HSCT in 24 children. The 5-year cumulative incidence of transplantation-related mortality and leukemia recurrence was 13% and 35%, respectively. Age older than 4 years predicted an increased risk of disease recurrence. The 5-year probability of event-free survival for children given HSCT from either a relative or a UD was 55% and 49%, respectively (P = NS), with median observation time of patients alive being 40 months (range, 6 to 144). In multivariate analysis, age older than 4 years and female sex predicted poorer outcome. Results of this study compare favorably with previously published reports. Disease recurrence remains the major cause of treatment failure. Outcome of UD-HSCT recipients is comparable to that of children receiving transplants from an HLA-identical sibling. (Blood. 2005;105:410-419)
Introduction
Juvenile myelomonocytic leukemia (JMML) is a rare hematopoietic malignancy of early childhood, representing 2% to 3% of all pediatric leukemias.1-3 JMML usually runs an aggressive clinical course, with median duration of survival for children left untreated being less than 12 months from diagnosis.1-5 Some young children with JMML (mainly those diagnosed before 2 years of age) may experience a longer course, sometimes characterized by temporary clinical improvement in the absence of therapy.2,6 Death usually occurs as the result of tumor cell infiltration of organs, leading to organ dysfunction, infection, and bleeding.
Neither intensive nor moderate chemotherapy approaches have been demonstrated to consistently improve the outcome of children with JMML,4-9 and allogeneic hematopoietic stem cell transplantation (HSCT) is presently the only curative treatment for this disorder. Different studies have reported that a significant proportion of children with JMML are cured by an allograft.10-15 Leukemia recurrence represents the main cause of treatment failure in children with JMML given HSCT, with the relapse rate being as high as 50%.13,14 Relapse occurs early, at a median of 4 months from transplantation and generally within the first year after the allograft.10,11,15
All studies published so far on the results of HSCT in children with JMML enrolled a limited number of patients treated with heterogeneous approaches. This fact significantly precluded the possibility of identifying patient-, disease-, and transplantation-related variables predicting the clinical course of a child with JMML treated with HSCT.
In this paper we analyze the outcome of 100 children with JMML given unmanipulated HSCT after an homogeneous preparative regimen consisting of 3 alkylating agents: busulfan, cyclophosphamide, and melphalan.
Patients and methods
Approval for the study was obtained from the institutional review board of each single institution. Written informed consent was provided by the patients' parents, according to the Declaration of Helsinki. The patients included in this study were diagnosed as affected by JMML according to previously published criteria.16,17 A total of 100 children who underwent transplantation from January 1993 through December 2002 in 29 centers from 7 countries and reported to the European Working Group on Myelodysplastic Syndrome in Childhood (EWOG-MDS) were evaluated. Data concerning patient and disease characteristics and transplantation outcome were collected by standardized questionnaires for each child enrolled into this study. Submitted data were reviewed by 1 physician (C.M.N.), and computerized error checks were performed to ensure data quality.
Patient characteristics, preparative regimen, GVHD prophylaxis, and supportive therapy
The characteristics of the 100 children (67 boys and 33 girls) and of the transplantation procedure are detailed in Tables 1 and 2, respectively. Fourteen children (7 boys and 7 girls) had clinical evidence of neurofibromatosis type 1 (NF1) in association with JMML.
Patient characteristics
No. of patients enrolled | 100 |
| Median patient sex, M/F | 67/33 |
| Median patient age at diagnosis, y (range) | 1.4 (0.1-14) |
| Median patient age at HSCT, y (range) | 2.5 (0.3-15) |
| Median interval between diagnosis and HSCT, mo (range) | 6 (0.3-49) |
| Median WBC count at diagnosis,* × 109/L (range) | 34 (3-240) |
| Median monocyte count at diagnosis,† × 109/L (range) | 5.5 (1-50) |
| Median platelet count at diagnosis,‡ × 109/L (range) | 65 (9-530) |
| Percentage of HbF at diagnosis§ (range) | 9 (0-80) |
| Karyotype | |
| Normal karyotype | 66 |
| Monosomy 7 | 20 |
| Trisomy 8 | 9 |
| Other abnormalities | 4 |
| Missing/unknown | 1 |
| No. of patients with clinical evidence of NF1∥ | 14 |
| Median WBC count at HSCT,¶ × 109/L (range) | 9.6 (0.7-320) |
| Percentage of marrow blasts at HSCT** (range) | 6 (0-85) |
| Splenectomy before HSCT | 24 |
| Median spleen size at HSCT,†† cm (range) | 5 (0-16) |
| Patient HCMV serology | |
| Negative | 57 |
| Positive | 39 |
| Unknown | 4 |
No. of patients enrolled | 100 |
| Median patient sex, M/F | 67/33 |
| Median patient age at diagnosis, y (range) | 1.4 (0.1-14) |
| Median patient age at HSCT, y (range) | 2.5 (0.3-15) |
| Median interval between diagnosis and HSCT, mo (range) | 6 (0.3-49) |
| Median WBC count at diagnosis,* × 109/L (range) | 34 (3-240) |
| Median monocyte count at diagnosis,† × 109/L (range) | 5.5 (1-50) |
| Median platelet count at diagnosis,‡ × 109/L (range) | 65 (9-530) |
| Percentage of HbF at diagnosis§ (range) | 9 (0-80) |
| Karyotype | |
| Normal karyotype | 66 |
| Monosomy 7 | 20 |
| Trisomy 8 | 9 |
| Other abnormalities | 4 |
| Missing/unknown | 1 |
| No. of patients with clinical evidence of NF1∥ | 14 |
| Median WBC count at HSCT,¶ × 109/L (range) | 9.6 (0.7-320) |
| Percentage of marrow blasts at HSCT** (range) | 6 (0-85) |
| Splenectomy before HSCT | 24 |
| Median spleen size at HSCT,†† cm (range) | 5 (0-16) |
| Patient HCMV serology | |
| Negative | 57 |
| Positive | 39 |
| Unknown | 4 |
WBC indicates white blood cell.
*Three unknown; †6 unknown; ‡3 unknown; §23 unknown; ∥8 unknown; ¶8 unknown; **13 unknown; ††6 unknown.
Transplantation procedure
| Donor | |
| HLA-identical family donor | 48 |
| Matched unrelated donor | 52 |
| Donor sex, M/F* | 50/46 |
| Median donor age, y† (range) | 19 (1-54) |
| Donor HCMV serology | |
| Negative | 55 |
| Positive | 38 |
| Unknown | 7 |
| Stem cell source | |
| Bone marrow | 79 |
| Peripheral blood | 14 |
| Cord blood | 7 |
| Median mononuclear cell dose infused | |
| Bone marrow, × 108/kg (range) | 6.1 (0.6-18.9) |
| Peripheral blood, CD34+ × 106/kg (range) | 20 (10-30) |
| Cord blood, × 107/kg (range) | 5 (4-14) |
| GVHD prophylaxis: matched family donor; matched unrelated donor | |
| None | 1; 0 |
| Cs-A | 32; 0 |
| MTX | 2; 0 |
| Cs-A + MTX | 7; 3 |
| Cs-A + ALG or MoAbs | 3; 3 |
| Cs-A + MTX + ALG or MoAbs | 1; 41 |
| Cs-A ± PDN ± MTX ± MoAbs | 2; 5 |
| Donor | |
| HLA-identical family donor | 48 |
| Matched unrelated donor | 52 |
| Donor sex, M/F* | 50/46 |
| Median donor age, y† (range) | 19 (1-54) |
| Donor HCMV serology | |
| Negative | 55 |
| Positive | 38 |
| Unknown | 7 |
| Stem cell source | |
| Bone marrow | 79 |
| Peripheral blood | 14 |
| Cord blood | 7 |
| Median mononuclear cell dose infused | |
| Bone marrow, × 108/kg (range) | 6.1 (0.6-18.9) |
| Peripheral blood, CD34+ × 106/kg (range) | 20 (10-30) |
| Cord blood, × 107/kg (range) | 5 (4-14) |
| GVHD prophylaxis: matched family donor; matched unrelated donor | |
| None | 1; 0 |
| Cs-A | 32; 0 |
| MTX | 2; 0 |
| Cs-A + MTX | 7; 3 |
| Cs-A + ALG or MoAbs | 3; 3 |
| Cs-A + MTX + ALG or MoAbs | 1; 41 |
| Cs-A ± PDN ± MTX ± MoAbs | 2; 5 |
Cl-A indicates cyclosporin-A; MTX, short-term methotrexate; ALG, antilymphocyte globulin; MoAbs, monoclonal antibodies.
*Four unknown; †21 unknown.
The median age at presentation was 1.4 years, with a range between 0.1 and 14 years. Forty-eight children received transplants from an HLA-identical relative (sibling in 46 cases and phenotypically identical aunt or mother in 1 case each); the remaining 52 children were given the allograft from an unrelated donor (UD).
Successful cytogenetic analysis of malignant cells was available in all cases but one. For 81 patients, karyotype taken within 6 weeks prior to HSCT was available; for the remaining 18 children the karyotype obtained at time of diagnosis was used. Monosomy of chromosome 7 was the most frequent cytogenetic abnormality, being observed in 20 of the 33 patients with an abnormal karyotype.
Splenectomy before transplantation had been performed in 24 children.
To evaluate the impact of therapy before transplantation on posttransplantation outcome, patients were subdivided into 2 groups according to the different kind of therapy received before transplantation: Group 1 comprised 84 children given either no treatment (43 patients, 12 of whom were splenectomized), differentiative therapy (ie, cis-retinoic acid; 1 patient who subsequently received splenectomy), or low-intensity chemotherapy (ie, 6-thioguanine, 6-mercaptopurine, low-dose cytarabine, others; 40 patients, 9 of whom were splenectomized); group 2 consisted of 16 children (2 of whom were splenectomized) receiving schemes of chemotherapy similar to those adopted for children with acute myelogenous leukemia (AML).
The preparative regimen was based on the use of busulfan (16 to 20 mg/kg given orally over 4 consecutive days), cyclophosphamide (60 mg/kg/d for 2 consecutive days), and melphalan (140 mg/m2 in single dose).18 In 25 children who underwent transplantation in centers in Italy, a pharmacokinetic (PK) study of busulfan was performed. In detail, the PK study was performed on the first administration of busulfan and the dosage adjusted starting from the fifth administration to maintain plasmatic levels between 500 and 800 ng/mL.19
Most children (35 of 48; 73%) given the allograft from an HLA-identical relative received cyclosporine-A (Cs-A) (1-3 mg/kg/d) alone to prevent occurrence of graft-versus-host disease (GVHD). The combination of Cs-A, short-term methotrexate (MTX) (15 mg/m2 on day +1 and 10 mg/m2 on days +3, +6, and +11), and either antithymocyte globulin (ATG) or, in a few cases, the monoclonal antibody Campath1-G was used in most patients receiving transplants from an unrelated volunteer.
Supportive therapy as well as prophylaxis and treatment of infections were similar among centers participating in this study. Human cytomegalovirus (HCMV) serologic status was studied before transplantation in donor-recipient pairs (see Tables 1 and 2). In all patients, HCMV infection was monitored and treated according to previously reported strategies.20
HLA typing
HLA-A and -B antigen serologic typing and a low-resolution generic DRB1 oligotyping were available for all donor-recipient pairs. Unrelated donors were located through networks of national and international bone marrow and cord blood donor registries. In all unrelated donor-recipient pairs, as well as when the donor was a relative other than an HLA-identical sibling, histocompatibility was determined by serology for HLA-A and -B antigens and by high-resolution DNA typing for DRB1 antigen. All children receiving transplants from an unrelated volunteer were either identical or had one antigenic/allelic disparity with their donor.
Definitions
Acute GVHD was diagnosed and graded by investigators at each transplantation center according to previously reported criteria.21 All patients surviving more than 10 days after transplantation were considered at risk for developing acute GVHD. Children alive at day +100 after transplantation with sustained donor engraftment were considered to be evaluable for chronic GVHD, which was classified according to previously reported criteria.22 Tissue biopsy samples were obtained to confirm diagnosis of GVHD whenever clinically indicated and feasible. Treatment of both acute and chronic GVHD was administered according to the protocols in use at each single institution.
Myeloid engraftment was defined as the first of 3 consecutive days when the neutrophil count was higher than 0.5 × 109/L and platelet engraftment as the first of 7 consecutive days with an unsupported platelet count higher than 50 × 109/L. Patients who did not engraft, as well as those with transient engraftment of donor cells, were considered to have graft failure.
Statistical analysis
Analysis used July 1, 2003, as the reference date (ie, the day at which all centers locked data on patient outcomes).
Overall survival (OS) was defined as the probability of survival, irrespective of disease state, at any point in time; patients alive at their last follow-up were censored, while only death was considered as an event. Event-free survival (EFS) was defined as the probability of being alive, disease-free, and with complete donor chimerism at any time point; death, relapse, rejection, and graft failure were considered as events, while patients alive and disease free with donor engraftment at their last follow-up were censored. Both these probabilities were analyzed by the Kaplan-Meier method, and comparisons between probabilities in different patient groups were performed using the log-rank test.23
Relapse incidence (RI) was defined as the probability of having a relapse before time t; death without experiencing a relapse was considered a competing event. On the contrary, transplantation-related mortality (TRM) was defined as the probability of dying without previous occurrence of a relapse, which was the competing event. Both these probabilities were estimated as cumulative incidence curves, as previously described.24-26
Also, the probabilities of acute and chronic GVHD and those of neutrophil and platelet engraftment were estimated as cumulative incidence. For acute GVHD analysis, relapse, death, and either rejection or graft failure were treated as competing events, while patients alive and relapse free at day +100 without having experienced acute GVHD were censored. For chronic GVHD, only patients surviving in remission and with donor engraftment for at least 100 days were evaluated. Also in this case, relapse, death, and either rejection or graft failure were considered competing events, data being censored at time of last follow-up for patients who did not experience chronic GVHD.
Finally, for neutrophil and platelet engraftment, competing events were relapse, death, or rejection before engraftment.
All results were expressed as 5-year probability or 5-year cumulative incidence (%) and 95% confidence interval (95% CI).
A univariate analysis of EFS, RI, and TRM was performed for the whole study population considering the following variables: patient and donor age; sex mismatch; interval between diagnosis and HSCT; leukocyte, monocyte, and platelet count at diagnosis; hemoglobin F (HbF) percentage corrected for patient age at diagnosis; karyotype, leukocyte count, and bone marrow blast percentage at HSCT; NF1; spleen size or splenectomy prior to HSCT; pretransplantation treatment; type of donor; stem cell source; infused cell dose; recipient and donor HCMV serology; type of GVHD prophylaxis; busulfan PK study; development of grades II-IV acute GVHD; and development of chronic GVHD.
For this purpose, continuous variables were categorized as follows: Each variable was first divided into 4 categories at approximately the 25th, 50th, and 75th percentiles. If the relative event rates (ratio of the observed number of events to the expected number of events in a category, assuming no variation across categories) in 2 or more adjacent categories (and the mean times to event) were not substantially different, these categories were grouped. If no clear pattern was observed for the primary outcome, the median was taken as cut point.27
For multivariate analyses, the Cox proportional hazard regression model was used, including in the models all the variables with P less than .1 in univariate analysis.28,29
The χ2 test was used to compare differences in percentages.
All P values were 2-sided, and values less than .05 were considered statistically significant. P values above .1 were reported as nonsignificant (NS), whereas those between .05 and .1 were reported in detail.
The SAS package (SAS Institute, Cary, NC) and NCSS 2001 (Number Cruncher Statistical Systems, Kaysville, UT) were used for the analysis of the data.
Results
Engraftment and GVHD occurrence
Information on kinetics of myeloid recovery was available for all children included in this study. Three patients failed to engraft, 2 of whom had received a UD HSCT. Two more patients, both receiving transplants from a UD, presented a secondary marrow failure 27 and 39 days after HSCT. No other factor was associated with the occurrence of either primary or secondary graft failure. In children with sustained engraftment of donor cells, the median time to achieve neutrophil recovery was 18 days (range, 8 to 44). In the Cox analysis on the whole population, the use of cord blood as stem cell source and the absence of grades II-IV acute GVHD were factors associated with a delayed neutrophil engraftment (P = .0015, relative risk [RR] = 0.16, 95% CI: 0.051-0.49; and P = .017, RR = 0.58, 95% CI: 0.38-0.91; respectively).
The median time to obtain a self-sustained platelet count higher than 50 × 109/L was 30 days (range, 11 to 148). From the Cox model, we found that the most adverse factors for platelet recovery in the overall population were a platelet count at diagnosis less than 100 × 109/L and the use of cord blood as stem cell source (P = .02, RR = 0.54, 95% CI: 0.32-0.91; and P = .0065, RR = 0.21, 95% CI: 0.07-0.65; respectively).
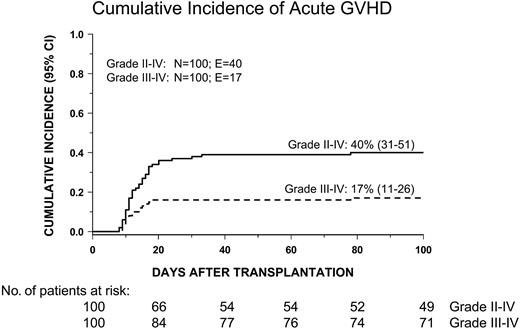
Grades II-IV acute GVHD developed in 40 patients. The cumulative incidence at day 100 of grades II-IV acute GVHD was 40% (31% to 51%) whereas that of grades III-IV acute GVHD was 17% (11% to 26%) (Figure 1). Children given HSCT from an HLA-compatible family donor had a cumulative incidence of grades II-IV acute GVHD comparable to that of patients receiving transplants from an unrelated volunteer (46% versus 35%, respectively; P = NS). No patient- or transplantation-related variable was significantly associated with the development of grades II-IV acute GVHD in a multivariate model.
Cumulative incidence of grades II-IV and grades III-IV acute GVHD in the overall cohort of patients studied.
Cumulative incidence of grades II-IV and grades III-IV acute GVHD in the overall cohort of patients studied.
Thirteen (15%) of the 86 patients at risk developed chronic GVHD, which was limited in 6 cases and extensive in the remaining 7 patients. The overall cumulative incidence of chronic GVHD was 17% (10% to 28%). Children given HSCT from an HLA-compatible family donor had a cumulative incidence of chronic GVHD similar to that of patients receiving transplants from an unrelated volunteer (17% versus 16%, respectively; P = NS). From the multivariate analysis performed using the Cox model, we found that previous grades II-IV acute GVHD was the only statistically significant risk factor for the occurrence of chronic GVHD (P = .016, RR = 4.96, 95% CI: 1.35-18.2).
Transplantation-related mortality
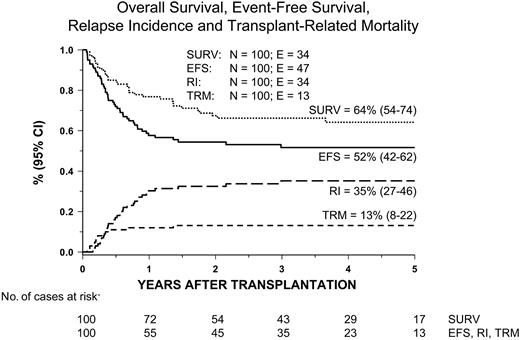
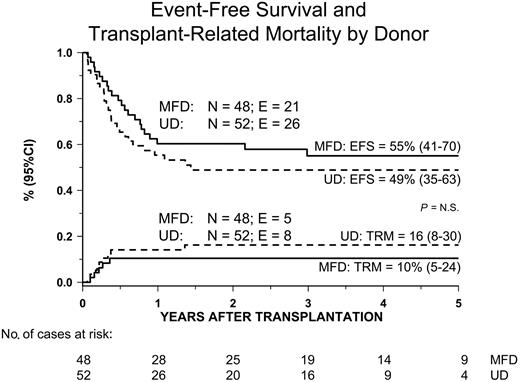
Thirteen patients died of transplantation-related causes, with the 5-year cumulative incidence of TRM being 13% (8% to 22%) (Figure 2). The 5-year cumulative incidence of TRM for patients receiving transplants from either an HLA-identical sibling or an unrelated volunteer was 10% (5% to 24%) and 16% (8% to 30%), respectively (P = NS; Figure 3). The median time to treatment-related death was 2.7 months (range, 1 to 16). Table 3 lists probabilities of TRM, RI, and EFS not adjusted for differences in factors that influence transplantation outcome. In univariate analysis, we found that patients receiving transplants from a female donor had a statistically higher probability of dying of transplantation-related causes; all other variables did not have any impact on the probability of death due to transplantation complications, possibly because of the limited number of events. None of the variables considered influenced TRM in multivariate analysis (Table 4).
Kaplan-Meier estimate of overall survival (surv) and event-free survival (EFS) and cumulative incidence of relapse (RI) and transplantation-related mortality (TRM) in the overall population.
Kaplan-Meier estimate of overall survival (surv) and event-free survival (EFS) and cumulative incidence of relapse (RI) and transplantation-related mortality (TRM) in the overall population.
Kaplan-Meier estimate of event-free survival (EFS) and transplantation-related mortality (TRM) according to the type of donor used.
Kaplan-Meier estimate of event-free survival (EFS) and transplantation-related mortality (TRM) according to the type of donor used.
Univariate analysis of 5-year event-free survival probability (EFS), cumulative incidence of relapse (RI), and cumulative incidence of transplantation-related mortality (TRM)
. | . | EFS . | . | RI . | . | TRM . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
. | No. of patients . | Probability . | 95% CI . | Cumulative incidence . | 95% CI . | Cumulative incidence . | 95% CI . | |||
| Overall probability or incidence | 100 | 52% | 42-62 | 35% | 27-46 | 13 | 8-22 | |||
| Patient sex | ||||||||||
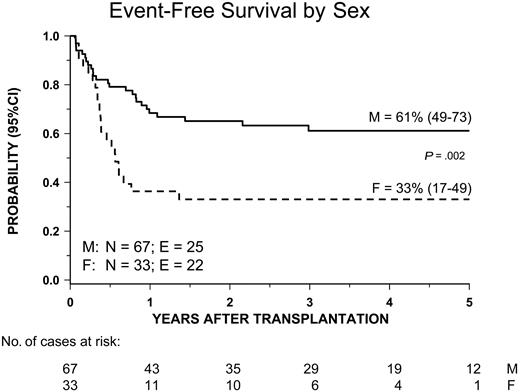
| Male | 67 | 61% | 49-73 | 30% | 20-44 | 9% | 4-19 | |||
| Female | 33 | 33% | 17-49 | 45% | 31-66 | 21% | 11-41 | |||
| p | — | .0021 | — | .012 | — | .065 | — | |||
| Donor sex | ||||||||||
| Male | 50 | 53% | 38-67 | 41% | 30-59 | 6% | 2-18 | |||
| Female | 46 | 53% | 39-68 | 25% | 15-42 | 22% | 13-38 | |||
| Missing | 4 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | .035 | — | |||
| Age at diagnosis | ||||||||||
| Less than 1 y | 38 | 65% | 46-78 | 17% | 8-35 | 21% | 12-40 | |||
| 1-2 y | 18 | 67% | 45-88 | 22% | 9-53 | 11% | 3-41 | |||
| 2-3 y | 9 | 47% | 10-84 | 53% | 27-100 | 0% | — | |||
| 3-4 y | 18 | 50% | 27-73 | 44% | 27-75 | 6% | 1-37 | |||
| 4 y or more | 17 | 16% | 0-34 | 73% | 53-98 | 12% | 3-43 | |||
| p | — | .013 | — | .0003 | — | NS | — | |||
| Less than 2 y | 56 | 64% | 51-76 | 18% | 10-32 | 18% | 10-32 | |||
| 2-4 y | 27 | 50% | 31-70 | 46% | 30-70 | 4% | 1-25 | |||
| 4 y | 17 | 16% | 0-34 | 73% | 53-98 | 12% | 3-43 | |||
| p | — | .0020 | — | .0001 | — | NS | — | |||
| Interval between diagnosis and HSCT | ||||||||||
| Less than 3 mo | 24 | 56% | 35-77 | 31% | 17-59 | 13% | 4-36 | |||
| 3-6 mo | 29 | 54% | 35-73 | 39% | 24-62 | 7% | 2-28 | |||
| 6-9 mo | 25 | 47% | 27-67 | 37% | 22-62 | 16% | 7-40 | |||
| 9 mo or more | 22 | 50% | 29-71 | 32% | 17-59 | 18% | 7-44 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Age at HSCT | ||||||||||
| Less than 1 y | 17 | 64% | 41-87 | 12% | 3-45 | 24% | 10-55 | |||
| 1-2 y | 27 | 66% | 48-84 | 19% | 9-42 | 15% | 6-37 | |||
| 2-3 y | 13 | 62% | 35-88 | 23% | 9-62 | 15% | 4-55 | |||
| 3-4 y | 18 | 55% | 32-78 | 45% | 27-75 | 0% | — | |||
| 4 y or more | 25 | 23% | 6-40 | 65% | 48-87 | 12% | 4-35 | |||
| p | — | .014 | — | .0008 | — | NS | — | |||
| Less than 2 y | 44 | 65% | 51-79 | 16% | 8-32 | 18% | 10-34 | |||
| 2-4 y | 31 | 57% | 39-75 | 37% | 23-59 | 6% | 2-25 | |||
| 4 y or more | 25 | 23% | 6-40 | 65% | 48-87 | 12% | 4-35 | |||
| p | — | .0022 | — | .0008 | — | NS | — | |||
| Donor age | ||||||||||
| Less than 10 y | 31 | 60% | 42-78 | 34% | 20-56 | 6% | 2-25 | |||
| 10-20 y | 9 | 33% | 3-64 | 33% | 13-84 | 33% | 13-84 | |||
| 20 y or more | 39 | 47% | 31-63 | 37% | 25-57 | 16% | 8-33 | |||
| Missing | 21 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Leukocyte count at diagnosis, × 109/L | ||||||||||
| Less than 20 | 25 | 51% | 31-71 | 45% | 29-70 | 4% | 1-27 | |||
| 20-39 | 35 | 49% | 32-67 | 30% | 18-51 | 20% | 10-39 | |||
| 40-59 | 17 | 40% | 17-64 | 36% | 19-69 | 24% | 10-55 | |||
| 60-79 | 11 | 64% | 35-92 | 36% | 17-79 | 0% | — | |||
| 80 or more | 9 | 63% | 30-97 | 37% | 15-91 | 0% | — | |||
| Missing | 3 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | .09 | — | |||
| Monocyte count at diagnosis, × 109/L | ||||||||||
| 1-2 | 13 | 54% | 27-80 | 31% | 14-70 | 15% | 4-55 | |||
| 2-3 | 15 | 51% | 25-78 | 42% | 23-78 | 7% | 1-44 | |||
| 3-5 | 16 | 56% | 32-81 | 31% | 15-65 | 13% | 3-46 | |||
| 5-10 | 25 | 38% | 19-58 | 37% | 22-62 | 25% | 12-50 | |||
| 10 or more | 25 | 62% | 43-82 | 34% | 19-59 | 4% | 1-27 | |||
| Missing | 6 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Platelet count at diagnosis, × 109/L | ||||||||||
| Less than 50 | 38 | 44% | 28-60 | 42% | 29-62 | 13% | 6-30 | |||
| 50-100 | 27 | 48% | 28-69 | 37% | 22-63 | 15% | 6-37 | |||
| 100 or more | 32 | 62% | 45-79 | 29% | 17-50 | 9% | 3-28 | |||
| Missing | 3 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
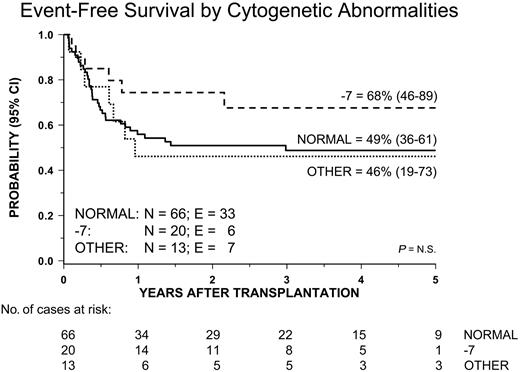
| Karyotype | ||||||||||
| Normal | 66 | 49% | 36-61 | 37% | 27-52 | 14% | 8-25 | |||
| Monosomy 7 | 20 | 68% | 46-89 | 22% | 9-54 | 10% | 3-37 | |||
| Other | 13 | 46% | 19-73 | 46% | 26-83 | 8% | 1-51 | |||
| Missing | 1 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| HbF % | ||||||||||
| Less than 5% | 26 | 61% | 42-80 | 20% | 9-43 | 20% | 9-43 | |||
| 5%-9% | 14 | 43% | 17-69 | 57% | 36-90 | 0% | — | |||
| 10%-39% | 23 | 51% | 29-72 | 32% | 17-59 | 17% | 7-42 | |||
| 40% or more | 14 | 19% | 0-41 | 81% | 62-100 | 0% | — | |||
| Missing | 23 | — | — | — | — | — | — | |||
| p | — | NS | — | .0083 | — | NS | — | |||
| Less than 40% | 63 | 53% | 41-66 | 33% | 23-47 | 14 | 8-26 | |||
| 40% or more | 14 | 19% | 0-41 | 81% | 62-100 | 0 | — | |||
| Missing | 23 | — | — | — | — | — | — | |||
| p | — | .07 | — | .004 | — | NS | — | |||
| Clinical evidence of NF1 | ||||||||||
| No | 78 | 55% | 43-66 | 34% | 24-47 | 12% | 6-22 | |||
| Yes | 14 | 36% | 11-61 | 50% | 30-84 | 14% | 4-52 | |||
| Missing | 8 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Leukocyte count at HSCT, × 109/L | ||||||||||
| Less than 10 | 46 | 53% | 38-68 | 32% | 20-49 | 15% | 8-30 | |||
| 10-19 | 20 | 47% | 25-70 | 37% | 20-67 | 16% | 6-45 | |||
| 20-39 | 17 | 45% | 20-69 | 43% | 25-77 | 12% | 3-43 | |||
| 40 or more | 9 | 44% | 12-77 | 56% | 31-100 | 0% | — | |||
| Missing | 8 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | — | — | |||
| Bone marrow blast percentage at HSCT | ||||||||||
| Less than 5% | 30 | 63% | 46-81 | 20% | 10-41 | 17% | 7-37 | |||
| 5%-19% | 47 | 52% | 37-67 | 35% | 24-52 | 13% | 6-27 | |||
| 20% or more | 10 | 0% | — | 90% | 73-100 | 10% | 2-64 | |||
| Missing | 13 | — | — | — | — | — | — | |||
| p | — | .10 | — | .017 | — | NS | — | |||
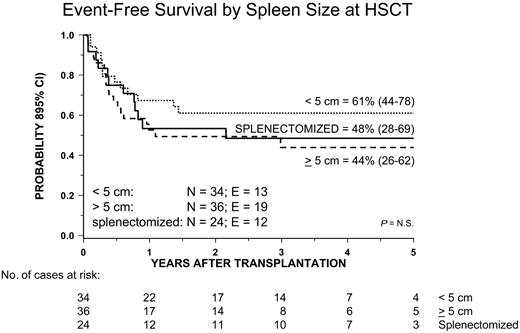
| Spleen size at HSCT | ||||||||||
| Less than 5 cm | 34 | 61% | 44-78 | 24% | 13-44 | 15% | 7-33 | |||
| 5 cm or more | 36 | 44% | 26-62 | 45% | 30-67 | 11% | 4-28 | |||
| Splenectomized | 24 | 48% | 28-69 | 39% | 23-65 | 13% | 4-36 | |||
| Missing | 6 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Pre-HSCT treatment | ||||||||||
| None or low dose | 84 | 52% | 41-63 | 35% | 26-47 | 13% | 8-23 | |||
| AML-like | 16 | 50% | 26-75 | 38% | 20-71 | 13% | 3-46 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Donor | ||||||||||
| Matched family donor | 48 | 55% | 41-70 | 35% | 23-52 | 10% | 5-24 | |||
| Unrelated donor | 52 | 49% | 35-63 | 36% | 24-52 | 16% | 8-30 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Stem cell source | ||||||||||
| Bone marrow | 79 | 51% | 40-62 | 35% | 26-47 | 14% | 8-24 | |||
| Peripheral blood | 14 | 55% | 28-82 | 36% | 18-72 | 9% | 1-59 | |||
| Cord blood | 7 | 54% | 14-93 | 32% | 10-100 | 14% | 2-88 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Stem cell dose, BM only | ||||||||||
| Less than 5 × 108/kg | 22 | 58% | 37-79 | 33% | 18-61 | 9% | 2-34 | |||
| 5 × 108/kg or more | 46 | 54% | 39-68 | 31% | 20-48 | 15% | 8-30 | |||
| Missing | 11 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| HCMV serology | ||||||||||
| Negative/Negative | 41 | 49% | 33-65 | 39% | 26-58 | 13% | 6-29 | |||
| Other | 53 | 54% | 40-67 | 33% | 22-49 | 13% | 7-26 | |||
| Missing | 6 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Busulfan PK study | ||||||||||
| Yes | 25 | 60% | 40-79 | 32% | 18-57 | 8% | 2-30 | |||
| No | 75 | 49% | 37-61 | 36% | 27-59 | 15% | 9-269 | |||
| p | — | NS | — | NS | — | NS | — | |||
| GVHD prophylaxis | ||||||||||
| All transplantations | ||||||||||
| Monotherapy | 35 | 59% | 42-75 | 33% | 20-54 | 9% | 3-25 | |||
| Combination treatment | 14 | 36% | 11-61 | 36% | 18-72 | 29% | 12-65 | |||
| Serotherapy | 51 | 52% | 37-66 | 36% | 25-53 | 12% | 6-25 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Sibling donor transplantations | ||||||||||
| Monotherapy | 35 | 59% | 42-75 | 33% | 20-54 | 9% | 3-25 | |||
| Combination treatment | 8 | 38% | 4-71 | 38% | 11-92 | 25% | 8-83 | |||
| Serotherapy | 5 | 60% | 17-100 | 40% | 14-100 | 0% | — | |||
| p | — | NS | — | NS | — | — | — | |||
| Unrelated donor transplantations | ||||||||||
| Monotherapy | 0 | — | — | — | — | — | — | |||
| Combination treatment | 6 | 33% | 0-71 | 33% | 11-100 | 33% | 11-100 | |||
| Serotherapy | 46 | 51% | 36-66 | 36% | 24-53 | 13% | 6-28 | |||
| p | — | NS | — | NS | — | 0.08 | — | |||
| Acute GVHD occurrence | ||||||||||
| Grades 0-I | 60 | 48% | 35-61 | 37% | 26-52 | 15% | 8-28 | |||
| Grades II-IV | 40 | 57% | 42-73 | 33% | 21-51 | 10% | 4-25 | |||
| p | — | NS | — | NS | — | .083 | — | |||
| Chronic GVHD | ||||||||||
| Absent | 73 | 59% | 48-71 | 35% | 26-48 | 6% | 2-15 | |||
| Present | 13 | 67% | 41-94 | 25% | 9-67 | 8% | 1-51 | |||
| Not evaluable | 14 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
. | . | EFS . | . | RI . | . | TRM . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
. | No. of patients . | Probability . | 95% CI . | Cumulative incidence . | 95% CI . | Cumulative incidence . | 95% CI . | |||
| Overall probability or incidence | 100 | 52% | 42-62 | 35% | 27-46 | 13 | 8-22 | |||
| Patient sex | ||||||||||
| Male | 67 | 61% | 49-73 | 30% | 20-44 | 9% | 4-19 | |||
| Female | 33 | 33% | 17-49 | 45% | 31-66 | 21% | 11-41 | |||
| p | — | .0021 | — | .012 | — | .065 | — | |||
| Donor sex | ||||||||||
| Male | 50 | 53% | 38-67 | 41% | 30-59 | 6% | 2-18 | |||
| Female | 46 | 53% | 39-68 | 25% | 15-42 | 22% | 13-38 | |||
| Missing | 4 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | .035 | — | |||
| Age at diagnosis | ||||||||||
| Less than 1 y | 38 | 65% | 46-78 | 17% | 8-35 | 21% | 12-40 | |||
| 1-2 y | 18 | 67% | 45-88 | 22% | 9-53 | 11% | 3-41 | |||
| 2-3 y | 9 | 47% | 10-84 | 53% | 27-100 | 0% | — | |||
| 3-4 y | 18 | 50% | 27-73 | 44% | 27-75 | 6% | 1-37 | |||
| 4 y or more | 17 | 16% | 0-34 | 73% | 53-98 | 12% | 3-43 | |||
| p | — | .013 | — | .0003 | — | NS | — | |||
| Less than 2 y | 56 | 64% | 51-76 | 18% | 10-32 | 18% | 10-32 | |||
| 2-4 y | 27 | 50% | 31-70 | 46% | 30-70 | 4% | 1-25 | |||
| 4 y | 17 | 16% | 0-34 | 73% | 53-98 | 12% | 3-43 | |||
| p | — | .0020 | — | .0001 | — | NS | — | |||
| Interval between diagnosis and HSCT | ||||||||||
| Less than 3 mo | 24 | 56% | 35-77 | 31% | 17-59 | 13% | 4-36 | |||
| 3-6 mo | 29 | 54% | 35-73 | 39% | 24-62 | 7% | 2-28 | |||
| 6-9 mo | 25 | 47% | 27-67 | 37% | 22-62 | 16% | 7-40 | |||
| 9 mo or more | 22 | 50% | 29-71 | 32% | 17-59 | 18% | 7-44 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Age at HSCT | ||||||||||
| Less than 1 y | 17 | 64% | 41-87 | 12% | 3-45 | 24% | 10-55 | |||
| 1-2 y | 27 | 66% | 48-84 | 19% | 9-42 | 15% | 6-37 | |||
| 2-3 y | 13 | 62% | 35-88 | 23% | 9-62 | 15% | 4-55 | |||
| 3-4 y | 18 | 55% | 32-78 | 45% | 27-75 | 0% | — | |||
| 4 y or more | 25 | 23% | 6-40 | 65% | 48-87 | 12% | 4-35 | |||
| p | — | .014 | — | .0008 | — | NS | — | |||
| Less than 2 y | 44 | 65% | 51-79 | 16% | 8-32 | 18% | 10-34 | |||
| 2-4 y | 31 | 57% | 39-75 | 37% | 23-59 | 6% | 2-25 | |||
| 4 y or more | 25 | 23% | 6-40 | 65% | 48-87 | 12% | 4-35 | |||
| p | — | .0022 | — | .0008 | — | NS | — | |||
| Donor age | ||||||||||
| Less than 10 y | 31 | 60% | 42-78 | 34% | 20-56 | 6% | 2-25 | |||
| 10-20 y | 9 | 33% | 3-64 | 33% | 13-84 | 33% | 13-84 | |||
| 20 y or more | 39 | 47% | 31-63 | 37% | 25-57 | 16% | 8-33 | |||
| Missing | 21 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Leukocyte count at diagnosis, × 109/L | ||||||||||
| Less than 20 | 25 | 51% | 31-71 | 45% | 29-70 | 4% | 1-27 | |||
| 20-39 | 35 | 49% | 32-67 | 30% | 18-51 | 20% | 10-39 | |||
| 40-59 | 17 | 40% | 17-64 | 36% | 19-69 | 24% | 10-55 | |||
| 60-79 | 11 | 64% | 35-92 | 36% | 17-79 | 0% | — | |||
| 80 or more | 9 | 63% | 30-97 | 37% | 15-91 | 0% | — | |||
| Missing | 3 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | .09 | — | |||
| Monocyte count at diagnosis, × 109/L | ||||||||||
| 1-2 | 13 | 54% | 27-80 | 31% | 14-70 | 15% | 4-55 | |||
| 2-3 | 15 | 51% | 25-78 | 42% | 23-78 | 7% | 1-44 | |||
| 3-5 | 16 | 56% | 32-81 | 31% | 15-65 | 13% | 3-46 | |||
| 5-10 | 25 | 38% | 19-58 | 37% | 22-62 | 25% | 12-50 | |||
| 10 or more | 25 | 62% | 43-82 | 34% | 19-59 | 4% | 1-27 | |||
| Missing | 6 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Platelet count at diagnosis, × 109/L | ||||||||||
| Less than 50 | 38 | 44% | 28-60 | 42% | 29-62 | 13% | 6-30 | |||
| 50-100 | 27 | 48% | 28-69 | 37% | 22-63 | 15% | 6-37 | |||
| 100 or more | 32 | 62% | 45-79 | 29% | 17-50 | 9% | 3-28 | |||
| Missing | 3 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Karyotype | ||||||||||
| Normal | 66 | 49% | 36-61 | 37% | 27-52 | 14% | 8-25 | |||
| Monosomy 7 | 20 | 68% | 46-89 | 22% | 9-54 | 10% | 3-37 | |||
| Other | 13 | 46% | 19-73 | 46% | 26-83 | 8% | 1-51 | |||
| Missing | 1 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| HbF % | ||||||||||
| Less than 5% | 26 | 61% | 42-80 | 20% | 9-43 | 20% | 9-43 | |||
| 5%-9% | 14 | 43% | 17-69 | 57% | 36-90 | 0% | — | |||
| 10%-39% | 23 | 51% | 29-72 | 32% | 17-59 | 17% | 7-42 | |||
| 40% or more | 14 | 19% | 0-41 | 81% | 62-100 | 0% | — | |||
| Missing | 23 | — | — | — | — | — | — | |||
| p | — | NS | — | .0083 | — | NS | — | |||
| Less than 40% | 63 | 53% | 41-66 | 33% | 23-47 | 14 | 8-26 | |||
| 40% or more | 14 | 19% | 0-41 | 81% | 62-100 | 0 | — | |||
| Missing | 23 | — | — | — | — | — | — | |||
| p | — | .07 | — | .004 | — | NS | — | |||
| Clinical evidence of NF1 | ||||||||||
| No | 78 | 55% | 43-66 | 34% | 24-47 | 12% | 6-22 | |||
| Yes | 14 | 36% | 11-61 | 50% | 30-84 | 14% | 4-52 | |||
| Missing | 8 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Leukocyte count at HSCT, × 109/L | ||||||||||
| Less than 10 | 46 | 53% | 38-68 | 32% | 20-49 | 15% | 8-30 | |||
| 10-19 | 20 | 47% | 25-70 | 37% | 20-67 | 16% | 6-45 | |||
| 20-39 | 17 | 45% | 20-69 | 43% | 25-77 | 12% | 3-43 | |||
| 40 or more | 9 | 44% | 12-77 | 56% | 31-100 | 0% | — | |||
| Missing | 8 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | — | — | |||
| Bone marrow blast percentage at HSCT | ||||||||||
| Less than 5% | 30 | 63% | 46-81 | 20% | 10-41 | 17% | 7-37 | |||
| 5%-19% | 47 | 52% | 37-67 | 35% | 24-52 | 13% | 6-27 | |||
| 20% or more | 10 | 0% | — | 90% | 73-100 | 10% | 2-64 | |||
| Missing | 13 | — | — | — | — | — | — | |||
| p | — | .10 | — | .017 | — | NS | — | |||
| Spleen size at HSCT | ||||||||||
| Less than 5 cm | 34 | 61% | 44-78 | 24% | 13-44 | 15% | 7-33 | |||
| 5 cm or more | 36 | 44% | 26-62 | 45% | 30-67 | 11% | 4-28 | |||
| Splenectomized | 24 | 48% | 28-69 | 39% | 23-65 | 13% | 4-36 | |||
| Missing | 6 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Pre-HSCT treatment | ||||||||||
| None or low dose | 84 | 52% | 41-63 | 35% | 26-47 | 13% | 8-23 | |||
| AML-like | 16 | 50% | 26-75 | 38% | 20-71 | 13% | 3-46 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Donor | ||||||||||
| Matched family donor | 48 | 55% | 41-70 | 35% | 23-52 | 10% | 5-24 | |||
| Unrelated donor | 52 | 49% | 35-63 | 36% | 24-52 | 16% | 8-30 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Stem cell source | ||||||||||
| Bone marrow | 79 | 51% | 40-62 | 35% | 26-47 | 14% | 8-24 | |||
| Peripheral blood | 14 | 55% | 28-82 | 36% | 18-72 | 9% | 1-59 | |||
| Cord blood | 7 | 54% | 14-93 | 32% | 10-100 | 14% | 2-88 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Stem cell dose, BM only | ||||||||||
| Less than 5 × 108/kg | 22 | 58% | 37-79 | 33% | 18-61 | 9% | 2-34 | |||
| 5 × 108/kg or more | 46 | 54% | 39-68 | 31% | 20-48 | 15% | 8-30 | |||
| Missing | 11 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| HCMV serology | ||||||||||
| Negative/Negative | 41 | 49% | 33-65 | 39% | 26-58 | 13% | 6-29 | |||
| Other | 53 | 54% | 40-67 | 33% | 22-49 | 13% | 7-26 | |||
| Missing | 6 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
| Busulfan PK study | ||||||||||
| Yes | 25 | 60% | 40-79 | 32% | 18-57 | 8% | 2-30 | |||
| No | 75 | 49% | 37-61 | 36% | 27-59 | 15% | 9-269 | |||
| p | — | NS | — | NS | — | NS | — | |||
| GVHD prophylaxis | ||||||||||
| All transplantations | ||||||||||
| Monotherapy | 35 | 59% | 42-75 | 33% | 20-54 | 9% | 3-25 | |||
| Combination treatment | 14 | 36% | 11-61 | 36% | 18-72 | 29% | 12-65 | |||
| Serotherapy | 51 | 52% | 37-66 | 36% | 25-53 | 12% | 6-25 | |||
| p | — | NS | — | NS | — | NS | — | |||
| Sibling donor transplantations | ||||||||||
| Monotherapy | 35 | 59% | 42-75 | 33% | 20-54 | 9% | 3-25 | |||
| Combination treatment | 8 | 38% | 4-71 | 38% | 11-92 | 25% | 8-83 | |||
| Serotherapy | 5 | 60% | 17-100 | 40% | 14-100 | 0% | — | |||
| p | — | NS | — | NS | — | — | — | |||
| Unrelated donor transplantations | ||||||||||
| Monotherapy | 0 | — | — | — | — | — | — | |||
| Combination treatment | 6 | 33% | 0-71 | 33% | 11-100 | 33% | 11-100 | |||
| Serotherapy | 46 | 51% | 36-66 | 36% | 24-53 | 13% | 6-28 | |||
| p | — | NS | — | NS | — | 0.08 | — | |||
| Acute GVHD occurrence | ||||||||||
| Grades 0-I | 60 | 48% | 35-61 | 37% | 26-52 | 15% | 8-28 | |||
| Grades II-IV | 40 | 57% | 42-73 | 33% | 21-51 | 10% | 4-25 | |||
| p | — | NS | — | NS | — | .083 | — | |||
| Chronic GVHD | ||||||||||
| Absent | 73 | 59% | 48-71 | 35% | 26-48 | 6% | 2-15 | |||
| Present | 13 | 67% | 41-94 | 25% | 9-67 | 8% | 1-51 | |||
| Not evaluable | 14 | — | — | — | — | — | — | |||
| p | — | NS | — | NS | — | NS | — | |||
GVHD prophylaxis was defined as follows: monotherapy, a single drug used (cyclosporin-A or methotrexate; the single patient who did not receive any GVHD prophylaxis was included in this group); combination treatment: more than one drug used (cyclosporin-A + methotrexate or steroids) without the addition of antilymphocyte globulin or monoclonal antibodies; serotherapy: any drug combination plus the addition of antilymphocyte globulin or monoclonal antibodies (Campath-1G).
—indicates that no value is available.
Multivariate analysis of variables influencing the probability of event-free survival (EFS), relapse incidence (RI), and transplantation-related mortality (TRM)
. | Relative risk . | 95% CI . | P . |
|---|---|---|---|
| EFS | |||
| Patient age at HSCT, 4 y or more vs less than 4 y | 2.24 | 1.07-4.69 | .032 |
| Patient sex, female vs male | 2.22 | 1.09-4.50 | .028 |
| HbF %, 40% or more vs less than 40% | 1.20 | 0.52-2.72 | NS |
| Bone marrow blast % at HSCT | |||
| 5%-19% vs less than 5% | 1.70 | 0.76-3.79 | NS |
| 20% or more vs less than 5% | 1.82 | 0.64-5.15 | NS |
| RI | |||
| Patient age at HSCT, 4 y or more vs less than 4 y | 2.96 | 1.26-6.92 | .012 |
| Patient sex, female vs male | 1.80 | 0.77-4.20 | NS |
| HbF %, 40% or more vs less than 40% | 1.90 | 0.79-4.54 | NS |
| Bone marrow blast % at HSCT | |||
| 5%-19% vs less than 5% | 2.08 | 0.78-5.55 | NS |
| 20% or more vs less than 5% | 2.06 | 0.60-7.06 | NS |
| TRM | |||
| Patient sex, female vs male | 2.18 | 0.66-7.22 | NS |
| Donor sex, female vs male | 2.87 | 0.76-10.87 | NS |
| WBC count at diagnosis, × 109/L; 30 or more vs less than 30 | 3.16 | 0.68-14.69 | NS |
| GVHD prophylaxis | |||
| Cs-A + MTX vs monotherapy | 3.95 | 0.77-20.35 | NS |
| Cs-A + MTX + ALG vs monotherapy | 1.43 | 0.35-5.92 | NS |
. | Relative risk . | 95% CI . | P . |
|---|---|---|---|
| EFS | |||
| Patient age at HSCT, 4 y or more vs less than 4 y | 2.24 | 1.07-4.69 | .032 |
| Patient sex, female vs male | 2.22 | 1.09-4.50 | .028 |
| HbF %, 40% or more vs less than 40% | 1.20 | 0.52-2.72 | NS |
| Bone marrow blast % at HSCT | |||
| 5%-19% vs less than 5% | 1.70 | 0.76-3.79 | NS |
| 20% or more vs less than 5% | 1.82 | 0.64-5.15 | NS |
| RI | |||
| Patient age at HSCT, 4 y or more vs less than 4 y | 2.96 | 1.26-6.92 | .012 |
| Patient sex, female vs male | 1.80 | 0.77-4.20 | NS |
| HbF %, 40% or more vs less than 40% | 1.90 | 0.79-4.54 | NS |
| Bone marrow blast % at HSCT | |||
| 5%-19% vs less than 5% | 2.08 | 0.78-5.55 | NS |
| 20% or more vs less than 5% | 2.06 | 0.60-7.06 | NS |
| TRM | |||
| Patient sex, female vs male | 2.18 | 0.66-7.22 | NS |
| Donor sex, female vs male | 2.87 | 0.76-10.87 | NS |
| WBC count at diagnosis, × 109/L; 30 or more vs less than 30 | 3.16 | 0.68-14.69 | NS |
| GVHD prophylaxis | |||
| Cs-A + MTX vs monotherapy | 3.95 | 0.77-20.35 | NS |
| Cs-A + MTX + ALG vs monotherapy | 1.43 | 0.35-5.92 | NS |
All variables with a P ≤ .1 in univariate analysis were considered as covariates and included in the Cox proportional hazard regression model.
Hepatic venoocclusive disease occurred in 11 patients (5 of whom received transplants from an unrelated volunteer) but, fortunately, it was not fatal in any of them. Three of these 11 patients relapsed, with the remaining 8 still being alive and in complete remission.
Relapse incidence
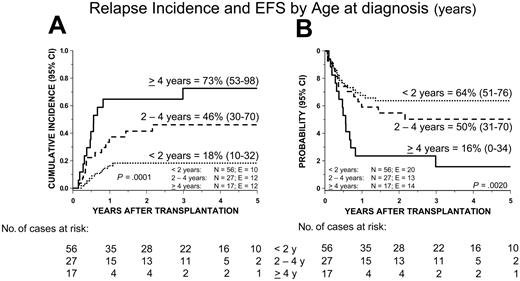
Thirty-four patients had hematologic relapse after transplantation, at a median time of 6 months (range, 2 to 36) after the allograft. Twenty-one children died due to disease progression at a median of 11 months after transplantation (range, 2 to 65). The 5-year cumulative incidence of relapse was 35% (Figure 2), with no significant difference between patients receiving transplants from either a relative or an unrelated donor (Table 3).
In univariable analysis (Table 3), the following features were associated with increased RI: female sex, age at diagnosis older than 4 years, high percentage of HbF, and blast percentage in the bone marrow at time of transplantation above 20%. From the Cox model, we found that only age at diagnosis older than 4 years remained a predictive variable for an increased risk of relapse (Table 4 and Figure 4A).
Cumulative incidence of relapse (RI) and Kaplan-Meier estimate of event-free survival (EFS) according to age at diagnosis.
Cumulative incidence of relapse (RI) and Kaplan-Meier estimate of event-free survival (EFS) according to age at diagnosis.
Survival and leukemia-free survival
Overall, 66 children remain alive after HSCT, the 5-year Kaplan-Meier estimate of survival being 64% (54% to 74%) (Figure 2).
Fifty-three patients are alive in first complete remission after HSCT, with a median observation time of 40 months (range, 6 to 144). The 5-year cumulative probability of EFS after the first allograft is 52% (42% to 62%) (Figure 2) for the whole cohort of patients studied, being 55% (41% to 70%) and 49% (35% to 63%) for patients given HSCT from either a relative or an unrelated donor, respectively (P = NS; Figure 3). Six patients are alive with disease, and 7 patients are alive in hematologic remission after a second allograft, which was performed in a total of 15 cases. Five of these 15 patients given a second transplantation died because of further disease recurrence, and 3 died due to transplantation-related complications. In 10 of the 15 children who received a second transplantation, the same donor used in the first HSCT was used and total body irradiation was added as part of the preparative regimen in 8 of these 15 patients. Moreover, less intensive GVHD prophylaxis was adopted to exploit a graft-versus-leukemia (GVL) effect; this choice resulted in the occurrence of grades II-IV acute GVHD in 8 of the 15 patients given a second allograft. The median follow-up of the 7 patients who are alive and disease free after the second HSCT is 2.3 years (range, 0.4 to 5.4).
Univariate analysis of factors related to the patient, disease, and transplantation influencing EFS showed that male sex and both age at diagnosis and age at transplantation younger than 4 years were associated with a better outcome. Both age at diagnosis and patient sex remained significant in multivariate analysis (Table 4 and Figures 4B and 5). All other variables did not have any impact on the probability of EFS. In particular, no significant differences in terms of EFS, RI, and TRM were observed between children given AML-type chemotherapy or less intensive treatment (Table 3). Also, neither splenectomy prior to HSCT nor spleen size at time of transplantation influenced the outcome (Figure 6). Of particular interest are the data on cytogenetics: Patients with monosomy 7 had an outcome comparable to that of children with either a normal karyotype or other cytogenetics abnormalities (Table 3 and Figure 7).
Kaplan-Meier estimate of event-free survival (EFS) according to the patient's sex.
Kaplan-Meier estimate of event-free survival (EFS) according to the patient's sex.
Kaplan-Meier estimate of event-free survival (EFS) in patients splenectomized before transplantation and according to spleen size at time of allograft.
Kaplan-Meier estimate of event-free survival (EFS) in patients splenectomized before transplantation and according to spleen size at time of allograft.
Kaplan-Meier estimate of event-free survival (EFS) according to the patient's karyotype.
Kaplan-Meier estimate of event-free survival (EFS) according to the patient's karyotype.
Finally, the probability of EFS of the 14 patients with NF1 was lower, although not statistically significantly, than that of children who did not have NF1 (Table 3).
Discussion
This is the largest study reported so far on children with JMML treated in the context of a prospective clinical trial with the same preparative regimen. With the follow-up now available, our data support the conclusion that allogeneic HSCT may cure approximately 50% of patients with JMML, with disease recurrence being the major cause of treatment failure. The probability of children enrolled in this study being alive and disease free compares favorably with that of many previously published reports on HSCT in children with JMML.10-14 In particular, the overall probability of EFS of the 48 children receiving transplants from an HLA-identical relative is 55%, a value better than that (38%) reported in the retrospective analysis published by our EWOG-MDS group on 24 children given the allograft from a family donor.11 Likewise, the EFS probability of 49% at 5 years after the allograft we have obtained in children receiving transplants from an unrelated volunteer is higher than that reported by the recent retrospective analysis of the National Marrow Donor Program in 46 children (24% at 2 years after transplantation).14
The choice of adopting a preparative regimen consisting of 3 alkylating agents was based on a preliminary study demonstrating the safety of this therapy18 and on the fact that a retrospective analysis of the EWOG-MDS group showed that a myeloablative therapy including busulfan was associated with a better EFS and a lower relapse incidence in comparison with regimens employing total body irradiation.11 Furthermore, we reasoned that avoiding radiotherapy could have the advantage of reducing the risk of severe radiation-induced growth retardation,30 endocrine and neuropsychological sequelae,31-33 and secondary malignancies.34,35 The results of this study confirm that the preparative regimen is safe, as the cumulative incidence of TRM was only 13%, with no significant difference between recipients of either HLA-identical sibling or UD transplants.
Also, in terms of ultimate outcome, our results seem to indicate that using UDs offers minimal or possibly no significant disadvantage as compared with employing an HLA-identical sibling. These data are in agreement with previously published studies in children with acute leukemia, where the probability of EFS in recipients of sibling HSCT was reported to be comparable to that of children given a UD allograft in the most recent years, thus suggesting the possibility of applying the same indications for HSCT independently of the type of donor available (ie, an HLA-identical sibling or an HLA-matched UD).36,37 Several factors may have contributed to the favorable outcome of our children receiving transplants from a UD. The possibility of selecting the donor using high-resolution molecular typing of HLA loci has been suggested to be potentially able to decrease the risk of graft failure, GVHD, and TRM.38,39 A learning and experience effect in handling recipients of UD HSCT, as well as optimization of the strategies of both prevention and treatment of GVHD, are also variables that could have contributed to the improved outcome.
Our results confirm the conclusion, reported in previously published studies,11-15 that relapse is the major cause of treatment failure in patients with JMML undergoing allogeneic HSCT. Relapse occurred in one third of our patients after a relatively short time from the allograft, with the median time from HSCT being 6 months with only 2 patients having relapsed later than 18 months after transplantation. Disease progression was also the most frequent cause of death. Previously published studies have found that older age,11,15 increased HbF,14 and abnormal karyotype15 are patient-specific risk factors for relapse after transplantation and that occurrence of chronic GVHD protects from the risk of disease relapse.14 Age above 2 years at diagnosis and a high percentage of HbF at diagnosis have been found to predict short survival also in studies analyzing the natural history of the disease in patients with JMML not given HSCT.2,5,6 In univariate analysis, we found that older age, female sex, increased percentage of HbF, and blast percentage in the bone marrow above 20% predicted the occurrence of leukemia relapse. However, only the former of these 4 variables remained significant in multivariate analysis.
Despite the usually aggressive reemergence of the malignant clone and the short time interval between first and second HSCT, a substantial number of our children (7 of the 15 who were given a second allograft) have achieved a second sustained hematologic remission thanks to a second transplantation. It is reasonable to hypothesize that less intensive GVHD prophylaxis could have contributed to the sustained remission after the second allograft in these patients by better preserving GVL effect. This finding is in agreement with previously published reports40,41 and indicates that leukemia relapse does not necessarily means a desperate prognosis and that a second transplantation should be considered as an option to be offered to every patient in good clinical condition.
Despite the delayed hematologic recovery, the ultimate outcome of cord blood transplant recipients was comparable to that of children given HSCT using either bone marrow or peripheral blood progenitors, thus providing further support to previously published studies that reported similar probabilities of EFS in children with malignancies undergoing transplantation with either placental blood or bone marrow-derived hematopoietic stem cells.42,43 The advantages of using cord blood are mainly represented by the prompt availability of this source of hematopoietic progenitors, which shortens the time needed to locate a suitable donor, and by the possibility of performing transplantations in the presence of 1 or 2 HLA disparities in the donor-recipient pairs.43,44 Both these factors can be relevant for treating children with JMML, whose disease, often running an aggressive clinical course, might not allow an extended time for finding a suitable unrelated bone marrow donor.
Two thirds of our patients had a normal karyotype, monosomy 7 being the most frequent cytogenetic anomaly. In contrast with a previous report documenting a negative impact of abnormal karyotype on the probability of OS after HSCT,15 we found that neither monosomy 7 nor other cytogenetic abnormalities confer a worse prognosis. This finding supports a recently published study from a United Kingdom study group on childhood MDS, which reported that in children with JMML monosomy 7 was associated with an outcome comparable to or even better than that of patients with normal karytotype.45
Splenectomy before HSCT, as well as spleen size at time of the allograft, did not appear to have an impact on posttransplantation outcome. One could argue that patients given splenectomy before transplantation were those with the largest spleen and, thus, with the greatest tumor burden, this possibly being associated with a higher risk of treatment failure. However, the fact that spleen size at time of transplantation influenced neither the risk of relapse nor the probability of survival in patients who did not undergo splenectomy does not support this hypothesis. The results of this study, as well those of previously published reports,11,14,46 are not in favor of an indiscriminate use of splenectomy before transplantation, the potential advantages having to be weighed against the risks related to the procedure or to postsplenectomy infections. The indication of performing splenectomy has to be carefully evaluated for each single child, the presence of massive splenomegaly with evidence of hypersplenism and/or refractoriness to platelet transfusions being an argument for considering this procedure in order to promote engraftment, to hasten hematologic recovery, and to lower the risk of hemorrhagic complications.
Clinical remissions and long-term survival after AML-type combination therapy have been reported in small series of children with JMML.8,47,48 Other investigators, however, pointed out that intensive chemotherapy is notably unsuccessful, especially in patients with aggressive disease.6,9,49 Neither EFS was improved; nor was relapse incidence reduced in our patients who had received intensive chemotherapy before the allograft. Thus, in view of these results, intensive chemotherapy prior to allogeneic HSCT cannot be recommended.
The worse outcome of female patients, also confirmed in multivariate analysis, is a finding never reported in previously published studies on children with JMML given HSCT.10-15 There is no immediate explanation for this finding, although it is noteworthy that among females there was a relatively higher percentage of patients with both NF1 and a low platelet count at time of diagnosis.
In conclusion, this study indicates that HSCT, after a preparative regimen consisting of busulfan, cyclophosphamide, and melphalan, may cure approximately 50% of patients with JMML and that nowadays results achievable using UD are comparable to those obtained employing an HLA-compatible related donor. Identification of factors influencing relapse rate and EFS can be of help in counseling patients. Disease recurrence remains the major cause of treatment failure, and novel strategies to lower the risk of relapse are warranted. In this regard, a reduction in both intensity and duration of GVHD prophylaxis might favor the emergence of a GVL effect displayed by donor lymphocytes, thus contributing to better leukemia control.
Appendix
The following transplantation teams enrolled patients in the present study.
Transplant center . | Country . | Principal investigator . |
|---|---|---|
| Berlin | Germany | Wolfram Ebell |
| Bologna | Italy | Andrea Pession |
| Cagliari | Italy | Franca Argiolu |
| Copenhagen | Denmark | Carsten Heilmann |
| Dublin | Ireland | Angus O'Marcaigh |
| Düsseldorf | Germany | Dagmar Dilloo |
| Erlangen | Germany | Wolfgang Holter |
| Essen | Germany | Bernhard Kremens |
| Frankfurt | Germany | Thomas Klingebiel |
| Freiburg | Germany | Charlotte Niemeyer |
| Geneva | Switzerland | Pierre Wacker |
| Genova | Italy | Giorgio Dini |
| Giessen | Germany | Alfred Reiter |
| Greifswald | Germany | James Beck |
| Hamburg | Germany | Hartmut Kabisch |
| Hannover | Germany | Karl Sykora |
| Jena | Germany | Felix Zintl |
| Kiel | Germany | Alexander Claviez |
| Leiden | The Netherlands | Elisabeth Korthof |
| Lund | Sweden | Albert Bekassy |
| Monza | Italy | Cornelio Uderzo |
| München | Germany | Monika Führer |
| Padova | Italy | Chiara Messina |
| Paraná | Brazil | Carmen S. Bonfim |
| Pavia | Italy | Franco Locatelli |
| Pisa | Italy | Claudio Favre |
| Prague | Czech Republic | Jan Stary |
| Tübingen | Germany | Peter Bader |
| Uppsala | Sweden | Johan Arvidson |
| Utrecht | The Netherlands | Tom Révész |
| Wien | Austria | Christina Peters |
Transplant center . | Country . | Principal investigator . |
|---|---|---|
| Berlin | Germany | Wolfram Ebell |
| Bologna | Italy | Andrea Pession |
| Cagliari | Italy | Franca Argiolu |
| Copenhagen | Denmark | Carsten Heilmann |
| Dublin | Ireland | Angus O'Marcaigh |
| Düsseldorf | Germany | Dagmar Dilloo |
| Erlangen | Germany | Wolfgang Holter |
| Essen | Germany | Bernhard Kremens |
| Frankfurt | Germany | Thomas Klingebiel |
| Freiburg | Germany | Charlotte Niemeyer |
| Geneva | Switzerland | Pierre Wacker |
| Genova | Italy | Giorgio Dini |
| Giessen | Germany | Alfred Reiter |
| Greifswald | Germany | James Beck |
| Hamburg | Germany | Hartmut Kabisch |
| Hannover | Germany | Karl Sykora |
| Jena | Germany | Felix Zintl |
| Kiel | Germany | Alexander Claviez |
| Leiden | The Netherlands | Elisabeth Korthof |
| Lund | Sweden | Albert Bekassy |
| Monza | Italy | Cornelio Uderzo |
| München | Germany | Monika Führer |
| Padova | Italy | Chiara Messina |
| Paraná | Brazil | Carmen S. Bonfim |
| Pavia | Italy | Franco Locatelli |
| Pisa | Italy | Claudio Favre |
| Prague | Czech Republic | Jan Stary |
| Tübingen | Germany | Peter Bader |
| Uppsala | Sweden | Johan Arvidson |
| Utrecht | The Netherlands | Tom Révész |
| Wien | Austria | Christina Peters |
Prepublished online as Blood First Edition Paper, September 7, 2004; DOI 10.1182/blood-2004-05-1944.
A complete list of the transplantation teams that enrolled patients in the present study appears in the “Appendix.”
Partly supported by grants from AIRC (Associazione Italiana Ricerca sul Cancro), CNR (Consiglio Nazionale delle Ricerche), 6th Framework Programme European Community “ALLOSTEM,” and IRCCS (Istituto di Ricovero e Cura a Carattere Scientifico) Policlinico S. Matteo to F.L.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal