The human peripheral B-cell compartment displays a large population of immunoglobulin M–positive, immunoglobulin D–positive CD27+ (IgM+IgD+CD27+) “memory” B cells carrying a mutated immunoglobulin receptor. By means of phenotypic analysis, complementarity-determining region 3 (CDR3) spectratyping during a T-independent response, and gene-expression profiling of the different blood and splenic B-cell subsets, we show here that blood IgM+IgD+CD27+ cells correspond to circulating splenic marginal zone B cells. Furthermore, analysis of this peripheral subset in healthy children younger than 2 years shows that these B cells develop and mutate their immunoglobulin receptor during ontogeny, prior to their differentiation into T-independent antigen-responsive cells. It is therefore proposed that these IgM+IgD+CD27+ B cells provide the splenic marginal zone with a diversified and protective preimmune repertoire in charge of the responses against encapsulated bacteria.
Introduction
The human peripheral B-cell compartment displays, in contrast to the mouse, a large population of CD27+ memory B cells that carry a mutated immunoglobulin receptor and represent up to 40% of circulating B cells. These memory B cells include the classical isotype-switched B cells and a population of immunoglobulin M–positive (IgM+) B cells, which were originally divided into an IgM-only subset, an IgM+IgD+ subset, and a minor IgD-only subpopulation.1-4
The splenic marginal zone (SMZ) is a unique B-cell compartment that contains B cells with a high surface density of IgM and complement receptor 2 (Cr2 or CD21), and which exhibits a rapid activation and immunoglobulin secretion in response to blood-borne T-independent (TI) antigens.5-7 Human SMZ B cells have been shown to carry somatic mutations, and mutated antibodies can be raised after immunization with T-independent polysaccharidic vaccines.8-11
We hypothesized previously that blood IgM+IgD+CD27+ B cells might form a B-cell subset distinct from the classical germinal center–derived memory B cells. This proposition was based on the fact that hyper-IgM (HIGM) patients who carry an invalidating mutation of the CD40L gene and do not possess normally developed germinal centers and switched memory B cells12 still presented a subpopulation of circulating IgM+IgD+CD27+ B cells.13,14 These B cells, moreover, carried a mutated immunoglobulin receptor, which led us to suggest that they could represent a different pathway of diversification that did not require a cognate T-B interaction and could thus be involved in T-independent immune responses.14 By means of phenotypic analysis, complementarity-determining region 3 (CDR3) spectratyping during a T-independent vaccination, and gene-expression profiling of the different blood and splenic B-cell subsets, we show here that the blood IgM+IgD+CD27+ B cells indeed correspond to circulating splenic marginal zone B cells in charge of T-independent responses, which is in accordance with a recent report15 and our previous proposition.
Materials and methods
Biologic samples
Fresh spleen samples were obtained from patients undergoing splenectomy owing to spherocytosis. Blood and spleen samples were obtained after parental or patient's informed consent, according to our institution's ethics committee. The complete diagnosis of asplenic patients is detailed in “Results.”
Antibodies
The following antibodies coupled with biotin, fluorescein isothiocyanate (FITC), R-phycoerythrin (PE), allophycocyanin (APC), Cy-Chrome, or the tandem dye PE-cyanin 5.1 (PC5) were used for flow cytometry or cell sorting: PC5–anti-CD19 (clone J4.119) and PE–anti-CD27 (clone 1A4-CD27) from Beckman Coulter (Fullerton, CA); APC–anti-CD19 (clone HIB19), Cy-Chrome–anti-CD21 (clone B-Ly4), FITC–anti-CD27 (clone M-T271), PE–anti-CD23 (clone M-L233), and biotin anti-IgD (clone IA6-2) from BD Pharmingen (San Jose, CA); goat antihuman IgD–FITC and biotinylated goat F(ab′)2 antihuman IgM from Caltag (Burlingame, CA). Purified anti-CD1c (clone F10/21A3) was provided by Dr B. Moody (Harvard Medical School, Boston, MA). Biotinylated and purified antibodies were revealed, respectively, with streptavidin PE-Cy7 (PC7) and PE-labeled goat antimouse IgG (Caltag). The following antibodies were used for the histologic studies: anti-CD1c (clone F10/21A3), anti-CD20 (clone L26), and polyclonal rabbit anti-IgD (Dako, Glostrup, Denmark); and anti-CD27 monoclonal antibody (mAb) (137B4) (Novocastra Laboratories; Newcastle, United Kingdom).
Immunohistology
All the antibodies were detected by means of the Vectastain ABC elite kit (Vector Laboratories, Burlingame, CA). The procedure has been decribed in detail elsewhere.16 Briefly, serial cryosections of spleen tissue were fixed in cold isopropanol for 10 minutes. After blocking of endogenous peroxidase activity by a glucose oxidase method, the sections were incubated overnight at 4°C with the primary antibodies. Bound antibodies were detected by biotinylated goat antirabbit or antimouse IgG (Dako) incubated for 30 minutes at room temperature. The avidin-biotinylated peroxidase complex was prepared according to the manufacturer's instructions. Sections were incubated with the avidin-biotinylated peroxidase complex for 30 minutes at room temperature. After washing, peroxidase activity was revealed with the use of diaminobenzidine (DAB). The monoclonal anti-CD27 (137B4) was visualized by a tyramide-enhanced ABC method. Sections were analyzed by conventional light microscopy. Photographs were made on film with a Zeiss Axiophot photomicroscope using a Plan-Apochromat 10 ×/0.32 lens. The final magnification before scanning is × 25.
Separation and flow cytometric analysis of IgD+CD27+ B cells
Human B cells from peripheral blood were enriched by negative selection with the RosetteSep B-cell enrichment cocktail (StemCell Technologies, Vancouver, BC, Canada). Splenic B cells were obtained after Ficoll density centrifugation and enrichment to greater than 98% by means of the B-cell–negative isolation kit (Dynal Biotech, Oslo, Norway). Three- and 4-color immunofluorescence analyses were performed on a FACScalibur with CellQuest software (Becton Dickinson, San Jose, CA). For isolation of splenic or peripheric IgD+CD27+, IgD-CD27+, and naive IgD+CD27- cells, purified B cells were stained with anti-IgD–FITC, antihuman CD27-PE, and anti-CD19–PC5 and sorted on a FACSvantage (Becton Dickinson). For microarray analysis, the IgD+CD27+ and IgD-CD27+ fractions were submitted to 2 successive sortings. For isolation of peripheral and splenic naive CD27- B cells, CD27+ B cells were first removed with the use of CD27-magnetic beads and LD depletion columns (Miltenyi Biotec, Gladbach, Germany). Then, enriched naive B cells were stained and sorted as described. Purity of all samples used for microarray analysis was greater than or equal to 99%.
RNA amplification and cDNA microarray analysis
Total RNA was isolated from sorted splenic and peripheral IgD+CD27+, IgD-CD27+, and naive cells by means of the RNeasy isolation Kit (Qiagen, Hilden, Germany). RNA samples were amplified in duplicate by means of a standard 2-round linear amplification protocol (Ambion, Austin, TX) to obtain between 25 and 50 μg cRNA. Gene-expression profiling analysis was performed with the use of Lymphochip microarrays.17 Briefly, amplified cRNA was reverse transcribed, labeled with Cy5, and hybridized to the microarrays together with Cy3-labeled probes generated from a standard pool of RNA derived from 9 lymphoid cell lines.
Amplification and analysis of rearranged V3-23 genes
Genomic DNA was extracted from sorted cells by proteinase K digestion. Rearranged V3-23 gene segments were amplified with Pfu Turbo polymerase (Stratagene, La Jolla, CA), with the use of a seminested polymerase chain reaction (PCR) strategy as previously described.14 PCR products were gel-purified and cloned by means of the TOPO TA Cloning kit (Invitrogen, Carlsbad, CA), and sequences of V3-23–positive colonies were obtained by automated sequencing (ABI310 genetic analyzer; Applera Biosystems, Foster City, CA). The sequences obtained were compared with the germ line V3-23 gene over 288 bp (from Glu1 to Cys92).
H-CDR3 spectratyping
Total RNA from 5 × 104 to 5 × 105 sorted IgD+CD27+ and naive cells was reverse transcribed by random priming, by means of the ProSTAR First Strand RT-PCR Kit (Stratagene). Amplification of V3-15–Cμ transcripts was performed on 2 μL cDNA aliquots with AmpliTaq DNA polymerase (Applera) with the use of a V3-15–specific primer (V3-15 leader: CTGAGCTGGATTTTCCTTGC) and a primer specific of the CH1 region of the μ heavy chain (μCH1: AAAAGGGTTGGGGCGGATGCAC) (45 seconds at 95°C, 60 seconds at 62°C, 90 seconds at 72°C for 25 cycles). V3-15–Cμ products were subjected to a second round of PCR amplification (45 seconds at 95°C, 60 seconds at 58°C, 90 seconds at 72°C for 25 cycles) with the use of the same Cμ primer with an internal V3-15–specific framework region 3 (FR3) primer (V3-15–FR3: CACAGCCGTGTATTACTGTAC). For H-CDR3 spectratyping, one fifth to one tenth of the purified PCR products were size fractionated on a denaturing 6% polyacrylamide gel and visualized by silver staining (Silver Sequence DNA Staining reagents; Promega, Madison, WI). Individual bands of interest (corresponding to a given CDR3 length) were excised from the gel and crushed in 10 μL water. Reamplification was performed with 1 μL excised PCR product for 20 cycles with the use of the primers and PCR conditions described. Several PCR amplifications were performed from 2 independent cDNAs for each cell sample.
Results
The circulating IgM+IgD+CD27+ B-cell subset displays a splenic marginal zone B-cell phenotype
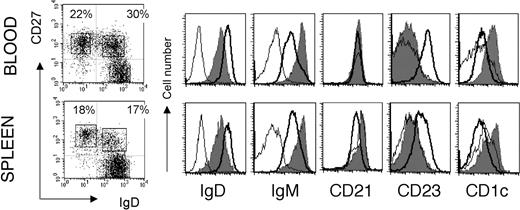
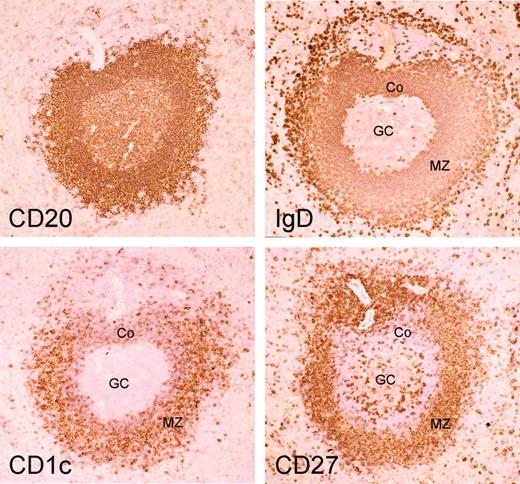
Mouse and human splenic marginal zone B cells can be distinguished from naive and switched B cells as being IgMhiIgDloCD21hiCD23-.6,18 Mouse MZ B cells are CD1d+,6 and it has been proposed that CD1c could be an MZ B-cell marker in humans.19 When human spleen B cells were analyzed with IgD and CD27 surface markers, they displayed a staining profile similar to the blood with a large compartment of CD27+ B cells (about 40%) divided into isotype-switched and IgD+ B cells (Figure 1). The CD27+IgD+ B cells are IgMhiIgDloCD21hiCD23- and CD1chi as compared with naive B cells, thus displaying the phenotype of splenic marginal zone B cells. Staining of serial sections of a human spleen with CD20, CD27, IgD, and CD1c indeed revealed a stronger expression of CD1c in B cells located in the marginal zone and confirmed the marginal zone localization of IgDlowCD27+ B cells, as previously described10 (Figure 2).
Markers specific of marginal zone B cells shared by blood and spleen IgM+IgD+CD27+ subsets. Purified B cells from blood and spleen are analyzed separately for IgM, CD21, CD23, and CD1c surface expression after gating of the 3 different CD19+ lymphocyte subsets distinguished by IgD and CD27 labeling. These data correspond to 1 representative case out of 4 different individuals. Naive B cells (IgD+CD27-) are indicated by the bold line; IgD+CD27+ B cells, the gray shadow; IgD-CD27+ cells, the thin line. Percentages of cells in the 2 CD27+ quadrants are indicated. The absence of IgM+ cells among the IgD-CD27+ subset is noticeable.
Markers specific of marginal zone B cells shared by blood and spleen IgM+IgD+CD27+ subsets. Purified B cells from blood and spleen are analyzed separately for IgM, CD21, CD23, and CD1c surface expression after gating of the 3 different CD19+ lymphocyte subsets distinguished by IgD and CD27 labeling. These data correspond to 1 representative case out of 4 different individuals. Naive B cells (IgD+CD27-) are indicated by the bold line; IgD+CD27+ B cells, the gray shadow; IgD-CD27+ cells, the thin line. Percentages of cells in the 2 CD27+ quadrants are indicated. The absence of IgM+ cells among the IgD-CD27+ subset is noticeable.
CD1c as a strong marker of splenic marginal zone B cells in humans. Serial cryosections of an adult human spleen are stained with anti-CD20, anti-IgD, anti-CD1c, and anti-CD27 antibodies (ABC technique; original magnification, × 25) marginal zone B cells are IgDlowCD27+CD1chigh. Note the more intense staining for IgD of the corona (Co) compared with the MZ B cells while the reverse is true for CD1c. The intense IgD staining of outer marginal zone B cells has been described previously.16 GC indicates germinal center.
CD1c as a strong marker of splenic marginal zone B cells in humans. Serial cryosections of an adult human spleen are stained with anti-CD20, anti-IgD, anti-CD1c, and anti-CD27 antibodies (ABC technique; original magnification, × 25) marginal zone B cells are IgDlowCD27+CD1chigh. Note the more intense staining for IgD of the corona (Co) compared with the MZ B cells while the reverse is true for CD1c. The intense IgD staining of outer marginal zone B cells has been described previously.16 GC indicates germinal center.
Similarly to the spleen, blood CD27+ B cells are divided into isotype-switched and IgD+ B cells. Here again, CD27+IgD+ B cells are IgMhiIgDloCD21+CD23- and CD1chi, although the CD21 marker stains the different B-cell subpopulations in this compartment similarly (Figure 1). In most cases, few cells were characterized as IgM-only within the IgD-CD27+ subset (Figure 1). IgM-only CD27+ B cells represented 1.1% of blood B cells (a mean value estimated from 11 individuals), with the exception of a 2-year-old child who displayed 7% of IgM-only B cells. Slightly higher values were observed for IgM-only CD27+ B cells in the spleen of children younger than 5 years (a mean value of 2.7%, including a 22-month-old child with 7.5% IgM-only cells).
Immunoglobulin gene mutation frequencies also correlate between splenic and blood compartments, being significantly lower in the IgM+IgD+CD27+ subset compared with switched B cells, as previously shown for the blood1 (Table 1). Remarkably enough, in all IgM+IgD+CD27+ B-cell samples analyzed from 1 year on, whether from spleen or blood, more than 90% of V sequences carried somatic mutations, thus indicating that the marginal zone compartment as a whole harbors mutated immunoglobulin genes.
Proportion and V3-23 gene mutation frequency of CD27+ B-cell subsets from blood and spleen of healthy individuals
. | . | IgD+CD27+ . | . | . | . | IgD-CD27+ . | . | . | . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | . | B cells, % . | . | Mutation frequency per 100 bp (total no. mutations; range of mutation per V sequence) . | . | B cells, % . | . | Mutation frequency per 100 bp (total no. mutations; range of mutation per V sequence) . | . | ||||||
| Donors . | Age . | Spleen . | Blood . | Spleen . | Blood . | Spleen . | Blood . | Spleen . | Blood . | ||||||
| C1 | 23 mo | 20 | 7.5 | 2.2 (58 mut; 0-17) | 3.1 (144 mut; 0-28) | 14 | 7 | 4.6 (225 mut; 0-37) | 4.7 (162 mut; 0-29) | ||||||
| C2 | 25 mo | 20 | 9 | 2.0 (82 mut; 0-12) | 3.8 (222 mut; 0-27) | 25 | 13 | 5.3 (216 mut; 8-29) | 4.9 (269 mut; 0-33) | ||||||
| C3 | 46 mo | 28 | 22 | 1.9 (95 mut; 0-25) | 2.8 (124 mut; 0-21) | 22 | 17 | 7.0 (182 mut; 6-31) | 6.6 (267 mut; 4-33) | ||||||
| C4 | 8 y | 31 | 35 | 2.4 (120 mut; 0-16) | 3.9 (183 mut; 2-26) | 10 | 16 | 4.8 (221 mut; 0-26) | 4.5 (90 mut; 8-20) | ||||||
| C5 | 9 y | 17 | 18 | 3.4 (109 mut; 1-17) | 5.2 (226 mut; 5-22) | 18 | 14 | 7.5 (196 mut; 6-32) | 5.2 (166 mut; 5-25) | ||||||
| 2.4 ± 0.6* | 3.8 ± 1* | 5.7 ± 1* | 5.2 ± 0.8* | ||||||||||||
. | . | IgD+CD27+ . | . | . | . | IgD-CD27+ . | . | . | . | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| . | . | B cells, % . | . | Mutation frequency per 100 bp (total no. mutations; range of mutation per V sequence) . | . | B cells, % . | . | Mutation frequency per 100 bp (total no. mutations; range of mutation per V sequence) . | . | ||||||
| Donors . | Age . | Spleen . | Blood . | Spleen . | Blood . | Spleen . | Blood . | Spleen . | Blood . | ||||||
| C1 | 23 mo | 20 | 7.5 | 2.2 (58 mut; 0-17) | 3.1 (144 mut; 0-28) | 14 | 7 | 4.6 (225 mut; 0-37) | 4.7 (162 mut; 0-29) | ||||||
| C2 | 25 mo | 20 | 9 | 2.0 (82 mut; 0-12) | 3.8 (222 mut; 0-27) | 25 | 13 | 5.3 (216 mut; 8-29) | 4.9 (269 mut; 0-33) | ||||||
| C3 | 46 mo | 28 | 22 | 1.9 (95 mut; 0-25) | 2.8 (124 mut; 0-21) | 22 | 17 | 7.0 (182 mut; 6-31) | 6.6 (267 mut; 4-33) | ||||||
| C4 | 8 y | 31 | 35 | 2.4 (120 mut; 0-16) | 3.9 (183 mut; 2-26) | 10 | 16 | 4.8 (221 mut; 0-26) | 4.5 (90 mut; 8-20) | ||||||
| C5 | 9 y | 17 | 18 | 3.4 (109 mut; 1-17) | 5.2 (226 mut; 5-22) | 18 | 14 | 7.5 (196 mut; 6-32) | 5.2 (166 mut; 5-25) | ||||||
| 2.4 ± 0.6* | 3.8 ± 1* | 5.7 ± 1* | 5.2 ± 0.8* | ||||||||||||
Mut indicates mutation.
Average mutation frequency. Mutation frequencies were significantly higher in IgD- compared with IgD+ cells, both in the overall sample (P < 10-9) and after stratification by tissue type (blood, P = .03; spleen, P < 10-9; nonparametric Wilcoxon score test)
Different types of CD27+ B-cell subsets in CD40- and AID-deficient children
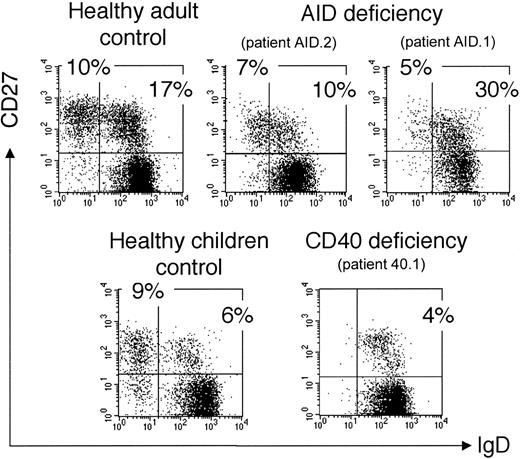
We analyzed 2 other groups of HIGM patients carrying an invalidating mutation either of the CD40 or of the AICDA gene encoding the activation-induced cytidine deaminase (AID). The CD40-deficient patient (40.1) is an 11-year-old child with a homozygous mutation of the CD40 gene described by Ferrari et al.20 This patient shows a peripheral B-cell staining profile similar to CD40 ligand (CD40L)–deficient patients with a large naive compartment and an IgM+IgD+CD27+ B-cell subpopulation (5% of CD19+ cells) as the only “memory” subset (Figure 3). Moreover, these IgM+IgD+CD27+ B cells carry a mutated immunoglobulin receptor with a mutation frequency of 3.8% measured on rearranged V3-23 genes: that is, an average of 11 mutations per VH sequence (Table 2). We also analyzed 2 patients who bear invalidating mutations of the AICDA gene.23,24 One patient (AID.1) presents a homozygous Arg112Cys missense mutation.21 Patient AID.2 presents heterozygous mutations of the AICDA gene (a 1-bp deletion at nucleotide 490 on one allele and an Ala111Thr amino acid missense mutation on the other). In contrast to CD40- and CD40L-deficient patients, AID-deficient patients do have germinal centers but are deficient in the hypermutation and isotype-switch mechanisms.24 Two different CD27+ B-cell subsets are observed in these 2 patients: an IgM+IgD+ and an IgM-only subset, representing, respectively, 10% to 30% and 5% to 7% of the peripheral B-cell pool (Figure 3). The existence of a clear IgM-only CD27+ subset in AID-deficient patients thus appears to be correlated with the presence of germinal centers. It may represent a stage prior to switching during normal B-cell development, accumulating in AID-deficient patients as a consequence of their defect in the molecular mechanism of isotype switch. The absence of somatic mutations was confirmed for both CD27+ B-cell subsets in these patients (Table 2; also data not shown).
Presence of an IgM+IgD+CD27+ subset in hyper-IgM patients. Patients AID.1 and 40.1 have been reported previously.20,21 Control is an 11-year-old child, age-matched with patient 40.1. Since IgD+CD27+cells coexpress IgM, the IgM+IgD+CD27+ subset is analyzed after IgD, CD27, and CD19 labeling of purified B cells. IgD and CD27 expression is shown after gating on CD19+ cells. Percentages of cells in the naive and the 2 CD27+ quadrants are indicated.
Presence of an IgM+IgD+CD27+ subset in hyper-IgM patients. Patients AID.1 and 40.1 have been reported previously.20,21 Control is an 11-year-old child, age-matched with patient 40.1. Since IgD+CD27+cells coexpress IgM, the IgM+IgD+CD27+ subset is analyzed after IgD, CD27, and CD19 labeling of purified B cells. IgD and CD27 expression is shown after gating on CD19+ cells. Percentages of cells in the naive and the 2 CD27+ quadrants are indicated.
Somatic mutations in IgM+IgD+CD27+ peripheral blood B cells of CD40- and AID-deficient patients
. | . | . | Sequences, no. . | . | Mutations . | . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Patient characteristics . | Donor . | Age, y . | Total . | Mutated . | Range . | No. . | Frequency per 100 bp . | |||
| CD40-deficient | 40.1 | 11 | 22 | 21 | 0-29 | 243 | 3.8 | |||
| AID-deficient | AID.1* | 41 | 10 | 0 | 0 | 0 | 0 | |||
| AID-deficient | AID.2* | 25 | 11 | 1 | 0-1 | 1 | 0.02 | |||
. | . | . | Sequences, no. . | . | Mutations . | . | . | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Patient characteristics . | Donor . | Age, y . | Total . | Mutated . | Range . | No. . | Frequency per 100 bp . | |||
| CD40-deficient | 40.1 | 11 | 22 | 21 | 0-29 | 243 | 3.8 | |||
| AID-deficient | AID.1* | 41 | 10 | 0 | 0 | 0 | 0 | |||
| AID-deficient | AID.2* | 25 | 11 | 1 | 0-1 | 1 | 0.02 | |||
Rearranged intronic JH4-JH5 sequences were amplified and sequenced for these 2 patients, as previously.22 The sequences obtained were compared over 334 bp with the germline sequences, from His 102 in the JH4 gene to 298 bp in the JH4-JH5 intron
Blood and splenic IgM+IgD+CD27+ B cells share a common gene-expression profile
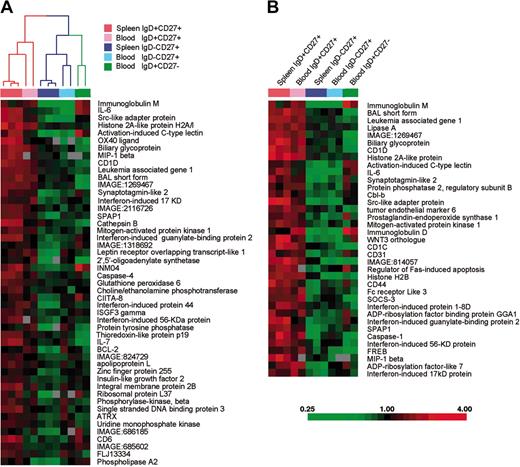
To more extensively compare splenic and blood IgM+IgD+CD27+ B cells with splenic and peripheral naive and switched subsets, we used Lymphochip DNA microarrays17 to profile gene expression of these different subsets after purification by cell sorting. We first searched for genes that were expressed at least 2-fold more highly in splenic IgD+CD27+ populations than in splenic IgD-CD27+ populations with high statistical significance (P < .005; t test). Forty-nine genes satisfied these criteria (Figure 4A). We used the hierarchical clustering algorithm to organize the various B-cell populations on the basis of the expression of these genes, and found that the blood IgD+CD27+ cells clustered with the splenic IgD+CD27+ in one arm of the dendrogram (Figure 4A). In the other arm of the dendrogram were the splenic and blood IgD-CD27+ memory B cells and the blood naive B cells. It is important to emphasize that this set of genes was not deliberately selected for similar expression in blood and splenic IgM+IgD+CD27+ cells. Therefore, the fact that the hierarchical clustering algorithm grouped these 2 B-cell populations together demonstrates that blood IgM+IgD+CD27+ B cells are more related to splenic marginal zone B cells than they are to naive or switched memory B cells. We then searched for genes that were expressed at least 2-fold more highly in the splenic and peripheral IgD+CD27+ populations than in the IgD-CD27+ populations with high statistical significance (P < .005). Figure 4B shows the expression levels in the various B-cell populations of 37 genes that satisfied these selection criteria. This set includes genes that encode the surface markers previously described and associated with marginal zone B cells (IgM, IgD, CD1) as well as a number of genes, like IL-6, not previously associated with this B-cell subpopulation.
A common gene-expression signature for IgM+IgD+CD27+ B cells from blood and spleen. Each column represents microarray data from a sample of the indicated cell subtype, and each row represents the expression of a single gene. The spleen IgD+CD27+ and IgD-CD27+ populations are obtained from 2 separate donors, with 1 of the 2 samples prepared in duplicate. Red squares indicate increased expression and green squares indicate decreased expression relative to the median expression of the gene according to the color bar shown. Gray squares indicate missing or excluded data. (A) The array dendrogram obtained by clustering the 49 genes that differentiated (P < .005, 2-fold higher expression) the splenic IgD+CD27+ samples from the splenic IgD-CD27+ samples. The red branches indicate the coclustering of the blood IgD+CD27+ samples, with the splenic IgD+CD27+ samples, and the blue branches indicate the coclustering of the blood IgD-CD27+ samples with their respective splenic IgD-CD27+ samples. (B) The 37 genes that achieved statistical significance with a 2-fold higher mean expression when the IgD+CD27+ cell populations are compared with the memory IgD-CD27+ cell populations.
A common gene-expression signature for IgM+IgD+CD27+ B cells from blood and spleen. Each column represents microarray data from a sample of the indicated cell subtype, and each row represents the expression of a single gene. The spleen IgD+CD27+ and IgD-CD27+ populations are obtained from 2 separate donors, with 1 of the 2 samples prepared in duplicate. Red squares indicate increased expression and green squares indicate decreased expression relative to the median expression of the gene according to the color bar shown. Gray squares indicate missing or excluded data. (A) The array dendrogram obtained by clustering the 49 genes that differentiated (P < .005, 2-fold higher expression) the splenic IgD+CD27+ samples from the splenic IgD-CD27+ samples. The red branches indicate the coclustering of the blood IgD+CD27+ samples, with the splenic IgD+CD27+ samples, and the blue branches indicate the coclustering of the blood IgD-CD27+ samples with their respective splenic IgD-CD27+ samples. (B) The 37 genes that achieved statistical significance with a 2-fold higher mean expression when the IgD+CD27+ cell populations are compared with the memory IgD-CD27+ cell populations.
Identical clones are shared by blood and splenic IgM+IgD+CD27+ B-cell subsets
To establish a functional link between the splenic and circulating IgM+IgD+CD27+ B cells, we studied the evolution of specific VDJ rearrangements by CDR3 spectratyping25 during a T-independent immunization. A 9-year-old child with a diagnosis of hereditary microspherocytosis, a nonimmunologic disease, was vaccinated against Streptococcus pneumoniae and Neisseria meningitidis 1 week before undergoing a splenectomy. Blood was collected 1 week before vaccination, 8 days after vaccination at the time of surgery together with the splenic sample, and 5 weeks after vaccination. Rearranged V3-15 genes have been shown to be mobilized in response to such an immunization11 and were therefore followed at these different stages. A particular CDR3 length, whose representation was increased in the IgM+IgD+CD27+ blood sample, was chosen for further study (online data supplement Figure 1 on the Blood website; see the supplemental figure link at the top of the online article). A specific V3-15 sequence displaying a unique VDJ join emerged 8 days after vaccination in the splenic and circulating IgM+IgD+CD27+ population, where it was recurrently found in different amplifications and contributed as many as half of the sequences amplified (Figure 5). This clone was found once among 38 sequences in the IgM+IgD+CD27+ subset before vaccination and already harbored 8 mutations in the complete V3-15 sequence at that stage (Figure 5; also data not shown). In contrast, this particular VDJ join could not be found in the naive B-cell compartment, whether from blood or spleen, at any stage of the experiment (no case among 53 different junctions). This clone still represented one third of the amplified sequences in the IgM+IgD+CD27+ cells of the blood 5 weeks after vaccination. This specific CDR3 is present in 9 variant mutated forms after vaccination, with one recurrent amino acid change contributed by 2 different mutations (GAT>GAA or GAT>GAG, leading to the same Asp→Glu replacement) and occurring in the context of various other base substitutions. Some of these mutations are shared by several samples (eg, a GAT>GAG change shared by the spleen and the blood 1 month after splenectomy, or an ACT>TTT double change found in the spleen and the blood at the same time) (Figure 5). From this analysis, we can conclude that a B-cell clone detectable in the blood IgM+IgD+CD27+ B-cell subset and harboring an already mutated V3-15 gene was mobilized and expanded in the splenic marginal zone and in the blood 1 week after immunization with a T-independent vaccination. The same clone was still present, but at a lower degree, 5 weeks later in the same blood subset.
B-cell clones with an identical V3-15 CDR3 shared by IgM+IgD+CD27+ B cells from blood and spleen during a T-independent response. Amplification of V3-15–Cμ mRNA sequences was performed from naive and IgM+IgD+CD27+ B cells of a 9-year-old child undergoing splenectomy and immunized against S pneumoniae and N meningitidis (plain polysaccharidic vaccines). The following samples were analyzed: blood before immunization, blood at the time of splenectomy (ie, 8 days after immunization), spleen, and blood 5 weeks after immunization. The first V3-15–specific PCR products were further amplified with V3-15–specific FR3 and Cμ primers, and the resulting products fractionated by denaturing gel electrophoresis. A specific CDR3 size was excised from the gel after silver staining and reamplified with the same FR3 and Cμ primers, and sequences were determined after cloning. Several PCR amplifications were performed from 2 independent cDNAs for each cell sample. The recurrent CDR3s encompassing the V3-15 and JH3 junctions observed in the various IgM+IgD+CD27+ fractions are shown, with the most frequently occurring sequence taken as reference (CDR3 is defined as amino acids included between the conserved Cys residue of FR3 and Trp residue of JH segments). *Clones found repeatedly in independent PCR amplifications.
B-cell clones with an identical V3-15 CDR3 shared by IgM+IgD+CD27+ B cells from blood and spleen during a T-independent response. Amplification of V3-15–Cμ mRNA sequences was performed from naive and IgM+IgD+CD27+ B cells of a 9-year-old child undergoing splenectomy and immunized against S pneumoniae and N meningitidis (plain polysaccharidic vaccines). The following samples were analyzed: blood before immunization, blood at the time of splenectomy (ie, 8 days after immunization), spleen, and blood 5 weeks after immunization. The first V3-15–specific PCR products were further amplified with V3-15–specific FR3 and Cμ primers, and the resulting products fractionated by denaturing gel electrophoresis. A specific CDR3 size was excised from the gel after silver staining and reamplified with the same FR3 and Cμ primers, and sequences were determined after cloning. Several PCR amplifications were performed from 2 independent cDNAs for each cell sample. The recurrent CDR3s encompassing the V3-15 and JH3 junctions observed in the various IgM+IgD+CD27+ fractions are shown, with the most frequently occurring sequence taken as reference (CDR3 is defined as amino acids included between the conserved Cys residue of FR3 and Trp residue of JH segments). *Clones found repeatedly in independent PCR amplifications.
Healthy and asplenic children younger than 2 years possess a diversified IgM+IgD+CD27+ blood B-cell subset
To ask whether hypermutation occurs in the absence of antigenic stimulation is not a feasible task in humans. Children younger than 2 years do not mount proper antibody responses against T-independent antigens such as bacteria carrying polysaccharidic capsids like Haemophilus influenzae and S pneumoniae. This may be due to the immaturity of the SMZ, which is not yet functional because it lacks the appropriate macrophages and/or the retention of the MZ B cells.26 We therefore focused our study on healthy children younger than 2 years of age and also on young and older patients with congenital asplenia.
We have previously shown that the IgM+IgD+CD27+ subset appears gradually with age in the circulation of the healthy population, starting at around 1% at birth (cord blood samples) and reaching 7% to 19% at 2 years and a mean of 20% in adults, with large individual variations (7% to 55%).27 A parallel increase with age was observed for immunoglobulin mutation frequencies with values close to background in cord blood, values that already reach 2% at 1 year and 3% to 4% in adults.27 For 2 children around 2 years of age, the proportion of IgM+IgD+CD27+ B cells in the spleen (20%) and the mutation frequency (about 2%) were similar to values found in older individuals (Table 1; also data not shown).
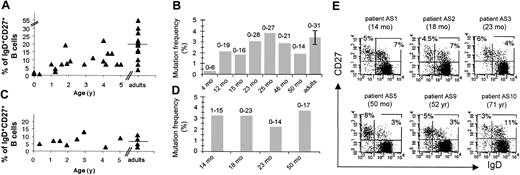
For asplenia syndromes, we restricted our study to patients whose condition was diagnosed according to several criteria, including the presence of Howell-Jolly bodies and the absence of spleen by echography, and in some cases by scintigraphy. Some of these patients suffered from frequent pneumococcal infections, and in 2 cases, death of a brother or sister by bacterial sepsis was reported (a detailed description is given in Table 3). Peripheral B cells were analyzed by flow cytometry for 10 patients, ranging from 4 months to 71 years of age, and by immunoglobulin gene mutation frequency for 4 of them (Figure 6C-E). All children with congenital asplenia displayed a normal peripheral CD27+ B-cell compartment with a normal IgM+IgD+CD27+ B-cell subset. Moreover, in the 4 cases analyzed (14, 18, 23, and 50 months), these IgM+IgD+CD27+ B cells were heavily mutated with a frequency of 2.2% to 3.7% (Figure 6D). For adult asplenic patients, both IgM+IgD+ and isotype-switched CD27+ subsets were on average reduced as compared with age-matched controls (Figure 6C; Table 3). Similarly, a reduction of IgM+IgD+CD27+ B cells has been recently described for an adult asplenic patient.15 In that study, however, a more drastic reduction in both CD27+ memory subsets was reported for young children.
Characteristics of 10 asplenic patients
. | . | . | . | . | % of B cells . | . | Mutation frequency, % . | . | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Patient, age . | Diagnosis . | Ultrasound and/or computerized tomography . | Howell-Jolly bodies . | Severe infections . | IgD+CD27+ . | IgD-CD27+ . | IgD+CD27+ . | IgD-CD27+ . | ||
| AS1 | ||||||||||
| 4 mo | Asplenia syndrome‡ | No spleen | + | None | 5 | 2 | ND | ND | ||
| 14 mo | 7 | 5 | 3.2 | 2.5 | ||||||
| AS2, 18 mo | Congenital asplenia | No spleen | + | Purpura fulminans¶ | 7 | 4.5 | 3.2 | 4 | ||
| AS3 | ||||||||||
| 23 mo | Functional asplenia | Small size§ | + | Meningitis, purpura fulminans, septicemia (S. pneumoniae) | 4 | 6 | 2.2 | 4 | ||
| 30 mo | 8 | 8.5 | ND | ND | ||||||
| AS4, 35 mo | Congenital asplenia | No spleen | + | Purpura fulminans¶ | 13 | 17 | ND | ND | ||
| AS5, 50 mo* | Congenital asplenia | No spleen | + | None | 3 | 8 | 3.7 | 4.7 | ||
| AS6, 5 y† | Congenital asplenia | No spleen | + | None | 9 | 11 | ND | ND | ||
| AS7, 18 y | Congenital asplenia | No spleen∥ | + | Septic shock with otitis¶ | 8 | 11 | ND | ND | ||
| AS8, 43 y | Congenital asplenia | No spleen | + | Septicemia (S. pneumoniae) | 5 | 14 | ND | ND | ||
| AS9, 52 y | Congenital asplenia | No spleen∥ | + | None | 3 | 5 | ND | ND | ||
| AS10, 71 y | Congenital asplenia | No spleen∥ | + | None | 11 | 3 | ND | ND | ||
. | . | . | . | . | % of B cells . | . | Mutation frequency, % . | . | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Patient, age . | Diagnosis . | Ultrasound and/or computerized tomography . | Howell-Jolly bodies . | Severe infections . | IgD+CD27+ . | IgD-CD27+ . | IgD+CD27+ . | IgD-CD27+ . | ||
| AS1 | ||||||||||
| 4 mo | Asplenia syndrome‡ | No spleen | + | None | 5 | 2 | ND | ND | ||
| 14 mo | 7 | 5 | 3.2 | 2.5 | ||||||
| AS2, 18 mo | Congenital asplenia | No spleen | + | Purpura fulminans¶ | 7 | 4.5 | 3.2 | 4 | ||
| AS3 | ||||||||||
| 23 mo | Functional asplenia | Small size§ | + | Meningitis, purpura fulminans, septicemia (S. pneumoniae) | 4 | 6 | 2.2 | 4 | ||
| 30 mo | 8 | 8.5 | ND | ND | ||||||
| AS4, 35 mo | Congenital asplenia | No spleen | + | Purpura fulminans¶ | 13 | 17 | ND | ND | ||
| AS5, 50 mo* | Congenital asplenia | No spleen | + | None | 3 | 8 | 3.7 | 4.7 | ||
| AS6, 5 y† | Congenital asplenia | No spleen | + | None | 9 | 11 | ND | ND | ||
| AS7, 18 y | Congenital asplenia | No spleen∥ | + | Septic shock with otitis¶ | 8 | 11 | ND | ND | ||
| AS8, 43 y | Congenital asplenia | No spleen | + | Septicemia (S. pneumoniae) | 5 | 14 | ND | ND | ||
| AS9, 52 y | Congenital asplenia | No spleen∥ | + | None | 3 | 5 | ND | ND | ||
| AS10, 71 y | Congenital asplenia | No spleen∥ | + | None | 11 | 3 | ND | ND | ||
ND indicates not determined.
One asplenic brother died from invasive infection (Streptococcus spp)
One asplenic brother died from invasive infection (S. pneumoniae)
Or ivemark syndrome (visceral heterotaxia: right atrial isomerism, cardiac malformation and asplenia)
Not visualized by 99mTc sulfur colloid scintigraphy
Confirmed by 99mTc scintigraphy
Germ not identified
Development and diversification of IgM+IgD+CD27+ peripheral B cells from healthy and asplenic children. (A) (B) (C) (D) Percentage of IgM+IgD+CD27+ peripheral B cells from healthy (A) and asplenic children (C) younger than 5 years. Mutation frequency of rearranged V3-23 genes from IgM+IgD+CD27+ peripheral B cells of healthy (B) and asplenic children (D). In panels B and D, each bar represents the mutation frequency of one individual, and the values marked above the bars represent the mutation range over the 288 bp V3-23 sequence analyzed; healthy adult values are pooled from 5 individuals. (E) IgD/CD27 staining profiles of peripheral CD19+ lymphocytes of asplenic individuals from 14 months to 71 years. The percentage of cells in each CD27+ quadrant is indicated. A complete description of asplenic patients is given in Table 3.
Development and diversification of IgM+IgD+CD27+ peripheral B cells from healthy and asplenic children. (A) (B) (C) (D) Percentage of IgM+IgD+CD27+ peripheral B cells from healthy (A) and asplenic children (C) younger than 5 years. Mutation frequency of rearranged V3-23 genes from IgM+IgD+CD27+ peripheral B cells of healthy (B) and asplenic children (D). In panels B and D, each bar represents the mutation frequency of one individual, and the values marked above the bars represent the mutation range over the 288 bp V3-23 sequence analyzed; healthy adult values are pooled from 5 individuals. (E) IgD/CD27 staining profiles of peripheral CD19+ lymphocytes of asplenic individuals from 14 months to 71 years. The percentage of cells in each CD27+ quadrant is indicated. A complete description of asplenic patients is given in Table 3.
Discussion
We have previously observed that the circulating and splenic CD27+ B cells, which represent approximately 40% of the peripheral and splenic B-cell pool in healthy individuals, are divided into isotype-switched and IgM+IgD+ B cells.27 A similar observation was made for peripheral B cells in a recent report by Kruetzmann et al.15 IgM-only CD27+ B cells appear to represent a minor subset (around 1% on average), which is observed only as a distinct subpopulation in some young children and in some pathologic cases (AID-deficiency and specific autoimmune syndromes; Weller S, unpublished data, 2002). Both of these studies, performed on a large cohort of healthy individuals, appear to contrast with the original description of memory B cells by Klein et al,1 who described a subpopulation of 10% IgM-only CD27+ B cells in the blood of 3 healthy subjects. Further analysis should help to clarify this issue.
The IgM+IgD+CD27+ B cells, either in blood or in spleen, display the phenotype of splenic marginal zone B cells, being IgMhiIgDloCD27+CD21+CD23-CD1chi for both organs. Histologic staining of human spleen with CD20, CD27, IgD, and CD1c confirmed that the IgD and CD27 markers indeed identify marginal zone B cells in humans.10
When the different splenic and blood B-cell subpopulations are analyzed by gene-expression profiling, the blood and splenic IgM+IgD+CD27+ B cells appear closer to each other than to any other subset. Some genes seem specifically expressed by these cells such as IL-6, which is also induced after activation of naive B cells, the CD31 receptor that is involved in transendothelial cell migration, and the CD1 surface receptor known to be involved in presentation of lipid antigens. All together, these 2 approaches, using cell-surface markers and gene-expression profiles, identified the blood IgM+IgD+CD27+ B-cell subset as the circulating counterpart of the splenic marginal zone B-cell compartment. In contrast to rodents,28 human SMZ B cells thus appear to recirculate, accounting for 10% to 30% of the B-cell pool in blood and in spleen and harboring mutated immunoglobulin genes.
Analysis of a CD40-deficient child showed, as for CD40L-deficient patients, an IgM+IgD+CD27+ B-cell subpopulation as the only memory subset. These cells carried a mutated immunoglobulin receptor with an average of 11 mutations per sequence. Such a high mutation frequency, with more than 95% of sequences carrying somatic mutations, was previously observed in CD40L-deficient children. It is clear, however, that this CD27+ subset is frequently reduced in HIGM children compared with age-matched controls, as the absence of CD40-CD40L interaction may disturb the cytokine network and alter the development of these cells as previously discussed.14 This study therefore argues in favor of our previous proposition that circulating IgM+IgD+CD27+ B cells belong to a separate B-cell subpopulation that develops and mutates its immunoglobulin receptor outside a germinal center–dependent cognate T-B interaction.
Immunization with a T-independent vaccine followed by splenectomy showed that a specific clone using the V3-15 gene, as followed by its VDJ signature, was strongly and specifically expanded in the splenic and blood marginal zone subsets 1 week after immunization. The same clone could be found 4 weeks after immunization in the blood IgM+IgD+CD27+ cells, although at a lower frequency. These results are consistent with previous reports showing that antibody responses to these bacterial capsular polysaccharides are oligoclonal and mobilize the V3-15 gene.29 Moreover, the potent amplification of the V3-15 clone in the splenic IgM+IgD+CD27+ population strongly suggests that this clone has been stimulated to expand in the splenic marginal zone by the T-independent vaccine. As previously shown for a similar immunization, responding clones remain for a few weeks in the general circulation.29 This V3-15 clone carried 8 mutations on its VH gene prior to vaccination. The results showing selection of some additional mutations on this gene suggest that this process could be reactivated in the spleen during the week following immunization.30 Moreover, they unambiguously establish that IgM+IgD+CD27+ B cells do recirculate between blood and splenic marginal zone in humans.
We have previously reported that the circulating IgM+IgD+CD27+ B-cell population was, surprisingly, already well developed and mutated at 1 year of age in healthy children,27 a stage at which the splenic marginal zone is not yet functional. In children younger than 2 years, the response to TI antigens has been reported to be defective, with a high incidence of infections caused by encapsulated bacteria.26 Our results on young children are thus intriguing since they indicate that circulating IgM+IgD+CD27+ B cells can develop and diversify their immunoglobulin receptors during ontogeny before a functional splenic marginal zone is matured. It is then tempting to propose that these cells could at this early stage have diversified their immunoglobulin receptors by hypermutation as a developmental program rather than following an immune response. Such a possibility has been mentioned in the original work describing these cells.1 A program of diversification involving the hypermutation process to generate a preimmune repertoire has already been reported for species such as sheep and rabbits,31 and humans could have conserved this strategy for one arm of their B-cell system. Hypermutation as a means to enlarge the preimmune repertoire has been recently proposed for the mouse as well, but such a process seems to occur only in specific models harboring a highly restricted V gene usage.32 It is clear that in all these models, a window of tolerance is likely to accompany this prediversification step.
When looking at adult asplenic patients, we could detect a clear diminution of CD27+ B cells, including both the isotype-switched and the IgM+IgD+CD27+ B cells, in accordance with Kruetzmann et al,15 and suggesting, as proposed by these authors, that the spleen could be the main source of these cells. Surprisingly, however, the younger asplenic patients we analyzed did not show, as opposed to their results, any significant alteration in the number and mutational status of their IgM+IgD+CD27+ B cells, implying that there may be large variations among these younger patients. The presence of these cells in several asplenic children would nevertheless suggest that other sites, such as marginal zone–like regions of Peyer patches and tonsils,7 might substitute for the developmental function of the spleen, without being able to sustain a similar B-cell production in adulthood or to replace the role that the splenic environment plays in supporting T-independent responses.
Immune responses to TI antigens are known to generate neither a memory response nor a maturation of affinity and to give protection for approximately 6 months.33 Effectively, SMZ B cells can switch after activation and differentiate rapidly into plasma cells34 that can probably remain in the organism for a few months. In such a sense, in spite of their previous characterization on the basis of their mutated immunoglobulin genes, IgM+IgD+CD27+ B cells would represent an immediate line of defense with none of the functional characteristics of memory. It has in fact been proposed that natural antibodies, which are essential for the immediate protection they provide against an infection, are constantly secreted by bystander stimulation of memory IgM and switched B cells.35 Most of the natural mutated IgM antibodies present in human serum are thus probably produced, as previously proposed,15 by circulating and splenic IgM+IgD+CD27+ B cells.
In conclusion, in this work and the previous ones by our group14 and by Kruetzmann et al,15 a new scheme of human B-cell development seems to emerge in which the IgM+IgD+CD27+ B cells in charge of T-independent responses follow a separate pathway of differentiation as compared with the classical B cells involved in T-dependent responses. We would also suggest that immunoglobulin gene hypermutation may play a double task in the human B-cell repertoire, by allowing the improvement of low-affinity antibodies during classical T-dependent immune responses, but also by generating a protective and diversified marginal zone B-cell repertoire that can be mobilized in rapid T-independent antibacterial responses.
Prepublished online as Blood First Edition Paper, June 10, 2004; DOI 10.1182/blood-2004-01-0346.
C.-A.R. and J.-C.W. share senior authorship.
Supported by the Ligue Nationale contre le Cancer (Equipe Labellisée); the Actions Concertées Incitatives (ACI) “Microbiologie, maladies infectieuses et parasitaires” of the Ministère de la Recherche; the Fourth Programme-Cadre de Recherche (PCRD) of the European Community (contract QLGI-CT-2001 “IMPAD”) (J.-C.W. and A.P.); and the Fondation Princesse Grace. S.W. has been supported during part of this work by the Association pour la Recherche contre le Cancer, the Société de Secours des Amis des Sciences, and the Centre National de la Recherche Scientifique (postdoctoral contract), and M.C.B. by the Deutsche Forschungsgemeinshaft.
The online version of the article contains a data supplement.
An Inside Blood analysis of this article appears in the front of this issue.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
We thank Ninon Lesage and Kun Yang for their contribution. We thank also Jérome Mégret and Frédéric Delbos for cell sorting. We thank Dr B. Moody for the gift of the anti-CD1c antibody, Dr V. Minard for the clinical follow-up of asplenic patients, and Prof C. Rose for providing blood sample. We thank Dr A. Alcais for the statistical analysis. Many thanks to Capucine Picard and Claire Fieschi for their invaluable support. The authors are grateful to Benedita Rocha for critical reading of the manuscript.


This feature is available to Subscribers Only
Sign In or Create an Account Close Modal