Abstract
We describe the toxicity and efficacy of donor lymphocyte infusions (DLIs) given to 81 patients (median age, 50 years) after reduced-intensity conditioning (RIC) transplantations performed at 16 centers in the United Kingdom. The diseases treated included non-Hodgkin lymphoma (NHL; n = 29), chronic myeloid leukemia (CML; n = 12), myeloma (n = 11), acute myeloid leukemia (AML; n = 10), and chronic lymphocytic leukemia (CLL; n = 9). Eighty-eight percent received stem cells from sibling donors. The patients received 130 infusions (median, 1; range, 1-4). Indications for DLI were unsatisfactory response/disease progression in 51 patients, mixed chimerism in 18, preemptive in 10, and other in 2. Graft hypoplasia was uncommon (11%). Grade II to IV graft-versus-host disease (GVHD) occurred in 23 of 81 patients (28%) and limited and extensive chronic GVHD in 5 of 69 and 18 of 69 evaluable patients (total incidence 33%). Conversion from mixed to full donor chimerism occurred in 19 of 55 evaluable patients (35%) at a median of 48 days after the DLI; partial responses occurred in 6 patients (total response rate 45%). Eighteen of 51 (35%) patients with measurable disease after stem cell transplantation had a complete response (2 molecular), and 5 a partial response (total response rate 45%). Eleven of 17 evaluable complete responders had full donor chimerism. Eight of 13 patients with follicular NHL had complete responses as did 4 of 12 patients with CML. Clinical and chimeric responses correlated strongly with acute and chronic GVHD. Forty-seven patients (58%) survive at a median of 508 days after transplantation (range, 155-1171 days) with a median Karnofsky score of 90. Thirty-four patients (42%) died at a median of 211 days after transplantation with the major causes being progressive disease (26%) and GVHD (9%). Further systematic studies are required to determine the efficacy and optimum use of DLI for patients with each disease treated by nonmyeloablative stem cell transplantation.
Introduction
Reduced-intensity conditioning (RIC) allografts, using nonmyeloablative conditioning, have been undertaken recently for a variety of hematologic1-3 and solid tumors.4 Their aim is to reduce short-term transplant-related morbidity and mortality with the intention of providing a platform for a subsequent graft-versus-malignancy (GVM) effect by early withdrawal of posttransplantation immunosuppression or donor lymphocyte infusions (DLIs).
There are a number of indications for DLI after RIC allografts. First, RIC transplantations often result in mixed donor and recipient chimerism. In published data from the 3 most commonly used regimens in the United Kingdom, mixed chimerism (MC) occurred in 42%, 36%, and 73% of cases analyzed, respectively.1,5,6 Sometimes, as the Seattle group3 has shown, a shift to full donor chimerism results spontaneously, but this is not invariably the case. Although graft-versus-host disease (GVHD) can occur in the setting of MC,7 overall there is less chance of a durable GVM effect and there may be a higher chance of graft rejection. Thus conversion to full donor chimerism following administration of DLI is likely to be beneficial in terms of both disease control and survival.
A second important indication for DLI is disease progression or the failure of the transplantation to achieve a complete remission (CR). This may be more common with less intensive conditioning.
Donor lymphocyte infusions, when given for chronic phase chronic myeloid leukemia (CML) that has relapsed after conventional allogeneic stem cell transplantation (SCT), result in a high incidence of durable cytogenetic and molecular remissions.8-10 Their efficacy for other hematologic diseases is much less well documented11,12 and it is only multicenter collaborative studies that have collected sufficient patients in each disease category that enable us to assess the likelihood of their efficacy in diseases such as acute myeloid leukemia (AML)11,12 or multiple myeloma.13 14
Third, DLIs are being increasingly given empirically in the posttransplantation setting, as a preemptive strike against disease relapse or on the assumption that they may eliminate undetectable minimal residual disease.
The RIC allografts confront physicians with a number of novel clinical situations in which decisions must be made. When faced with the need to prescribe DLI, we found that there was virtually no published information defining optimal cell dose and timing of these infusions. As a result, it is not possible to advise individual patients accurately about the chance of success. Similarly, data on the probability of severe toxicities such as graft hypoplasia and severe acute and chronic GVHD are scarce and may become clear only with large-scale studies.
To address these issues we report here the results of a national survey of 81 patients who underwent transplantation at 16 centers and received DLIs following RIC allogeneic transplantation.
Patients, materials, and methods
Patients
Twenty-eight adult allogeneic transplantation centers in the United Kingdom were invited to participate in a study concerning the administration of DLIs after RIC allografts. Five centers had performed no RIC allografts or had not given DLIs after these procedures. Sixteen of the remaining 23 centers (70%) provided data on 81 patients. These data represent all patients given DLIs who received RIC allografts at these centers.
The clinical characteristics of the patients are shown in Table1. The major diseases treated were non-Hodgkin lymphoma (NHL; n = 29), CML (n = 12), myeloma (n = 11), AML (n = 10), and chronic lymphocytic leukemia (CLL; n = 9). Remission status was favorable in 17, intermediate in 45, and unfavorable in 19. The majority of patients received 1 of 2 transplantation protocols: 37 of 81 patients had received fludarabine, melphalan, and Campath-1H1 and 21 received carmustine, etoposide, cytosine arabinoside (ara-C), melphalan, and Campath-1H.5 Seventy-one received stem cells from sibling donors (88%) and 10 from unrelated donors (UDs; only 1 mismatched). Fourteen (17%) experienced GVHD prior to the administration of DLI. Approval was individually obtained from each hospital's review board for these studies. Informed consent was provided according to the Declaration of Helsinki.
Patient and clinical characteristics
| Characteristic . | No. . |
|---|---|
| Median age, y (range) | 50 (25-66) |
| Sex, M/F | 56/25 |
| Sibling donor/unrelated donor | 71/10 |
| Protocol | |
| Fludarabine/melphalan/Campath-1H | 37 |
| BCNU/etoposide/ara-C/melphalan/Campath-1H | 21 |
| Busulphan/fludarabine/T-cell depletion | 8 |
| Fludarabine/cyclophosphamide | 9 |
| Fludarabine/cyclophosphamide/Campath-1H | 4 |
| Fludarabine/melphalan | 2 |
| Disease | |
| AML | 10 |
| CML | 12 |
| CLL | 9 |
| Myeloma | 11 |
| NHL (n = 29) | |
| Follicular | 16 |
| High grade | 6 |
| Mantle cell | 4 |
| Unspecified | 3 |
| Hodgkin disease | 4 |
| Myelodysplastic syndrome | 2 |
| Other* | 4 |
| Remission status/stage of disease at transplantation | |
| CR1 (including 4 CML-CP1) | 15 |
| CR2 | 2 |
| CR3 or later remission | 5 |
| PR (including 2 CML-CP2) | 28 |
| Sensitive relapse | 14 |
| Untested relapse | 6 |
| Primary refractory disease | 3 |
| Resistant relapse | 8 |
| Disease status at transplantation (%) | |
| Favorable | 17 (21) |
| Intermediate | 45 (56) |
| Unfavorable | 19 (23) |
| Characteristic . | No. . |
|---|---|
| Median age, y (range) | 50 (25-66) |
| Sex, M/F | 56/25 |
| Sibling donor/unrelated donor | 71/10 |
| Protocol | |
| Fludarabine/melphalan/Campath-1H | 37 |
| BCNU/etoposide/ara-C/melphalan/Campath-1H | 21 |
| Busulphan/fludarabine/T-cell depletion | 8 |
| Fludarabine/cyclophosphamide | 9 |
| Fludarabine/cyclophosphamide/Campath-1H | 4 |
| Fludarabine/melphalan | 2 |
| Disease | |
| AML | 10 |
| CML | 12 |
| CLL | 9 |
| Myeloma | 11 |
| NHL (n = 29) | |
| Follicular | 16 |
| High grade | 6 |
| Mantle cell | 4 |
| Unspecified | 3 |
| Hodgkin disease | 4 |
| Myelodysplastic syndrome | 2 |
| Other* | 4 |
| Remission status/stage of disease at transplantation | |
| CR1 (including 4 CML-CP1) | 15 |
| CR2 | 2 |
| CR3 or later remission | 5 |
| PR (including 2 CML-CP2) | 28 |
| Sensitive relapse | 14 |
| Untested relapse | 6 |
| Primary refractory disease | 3 |
| Resistant relapse | 8 |
| Disease status at transplantation (%) | |
| Favorable | 17 (21) |
| Intermediate | 45 (56) |
| Unfavorable | 19 (23) |
BCNU indicates 1,3-bis-(2-chloroethyl)-1-nitrosourea; bleomycin and carmustine.
Other diseases treated were renal carcinoma (1), hypereosinophilic syndrome, T-prolymphocytic leukemia, and immunocytoma.
We also collected basic demographic data from patients at the same center who received RIC allografts but did not require DLI. These data were from 264 patients receiving transplants at 15 of 16 of the same centers reporting the 81 study patients (Table2). In comparison to the group who received DLI, their median age was slightly lower (47.5 versus 50 years, P = .084, Mann-Whitney U test), they received stem cells more from donors other than matched siblings (P = .003, continuity corrected χ2 test), and there were more patients with AML (28% versus 12%) and fewer with CML (7% versus 15%). The reasons for these patients not receiving DLI are presented in Table 2. The most common reasons were that the patient was in CR or that the patient's overall condition was poor.
Demographic details of 264 adult patients from 15 centers who had RIC allografts but did not receive DLIs
| Characteristic . | No. . |
|---|---|
| Median age, y (range) | 47 (17-66) |
| Sex, M/F | 165/99 |
| Sibling donor/unrelated donor/other | 186/74/4 |
| Disease | |
| AML and myelodysplastic syndrome | 73 |
| CML | 18 |
| CLL | 16 |
| Myeloma | 37 |
| NHL | 88 |
| Hodgkin disease | 22 |
| Other | 10 |
| Disease status (n = 205)* | |
| Favorable | 65 (32%) |
| Intermediate | 91 (44%) |
| Unfavorable | 49 (24%) |
| Reasons for not receiving DLI (%) | |
| Patient in CR | 112 (42) |
| Poor overall condition of patient | 70 (27) |
| Patient died before DLI could be given | 18 (7) |
| Current GVHD | 16 (6) |
| No DLI available | 7 (3) |
| Past severe GVHD | 4 (2) |
| Other† | 14 (5) |
| Not known | 23 (9) |
| Characteristic . | No. . |
|---|---|
| Median age, y (range) | 47 (17-66) |
| Sex, M/F | 165/99 |
| Sibling donor/unrelated donor/other | 186/74/4 |
| Disease | |
| AML and myelodysplastic syndrome | 73 |
| CML | 18 |
| CLL | 16 |
| Myeloma | 37 |
| NHL | 88 |
| Hodgkin disease | 22 |
| Other | 10 |
| Disease status (n = 205)* | |
| Favorable | 65 (32%) |
| Intermediate | 91 (44%) |
| Unfavorable | 49 (24%) |
| Reasons for not receiving DLI (%) | |
| Patient in CR | 112 (42) |
| Poor overall condition of patient | 70 (27) |
| Patient died before DLI could be given | 18 (7) |
| Current GVHD | 16 (6) |
| No DLI available | 7 (3) |
| Past severe GVHD | 4 (2) |
| Other† | 14 (5) |
| Not known | 23 (9) |
Disease status is not known for 59 patients.
Reasons included declined by patient (3), had second BMT (2), physician choice (2), had second BMT (2), graft failure (2), had imatinib (1), and planned in future (2).
Nonmyeloablative conditioning protocols and GVHD prophylaxis
Four major protocols were used, as follows.
Regimen A.
The most frequently used protocol involves fludarabine (30 mg/m2) day −7 to −3, Campath-1H (10 mg twice daily) from day −7 to −3, and melphalan 140 mg/m2 on day −2 followed by stem cell infusion on day 0.1 Cyclosporin was the sole immunosuppressive agent given after transplantation.
Regimen B.
BEAM-Campath regimen: carmustine (300 mg/m2) day −6, ara-C (200 mg/m2 twice daily) days −5 to −2, etoposide (200 mg/m2) days −5 to −2, and melphalan (140 mg/m2) day −1. Campath-1G (10 mg/d) or Campath-1H (20 mg/d) was given continuously from day −5 to −1 inclusive. Some patients on this protocol received fludarabine (30 mg/m2) days −9 to −7. GVHD prophylaxis was with cyclosporin and 3 doses of methotrexate (10 mg/m2) on days 1, 3, and 6.5
Regimen C.
Busulfan 8 mg/kg given in 2 days, fludarabine 30 mg/m2 for 5 days, and a Miltenyi CD34 selection of the graft with planned DLI at 180, 270, and 360 days.6 Cyclosporin was given after transplantation.
Regimen D.
Fludarabine 30 mg/m2 for 5 days and cyclophosphamide 1 g intravenously for 2 days. The graft was given unmanipulated.
Techniques for assessing chimerism
We asked reporting centers to categorize their method of assessing chimerism. Multilineage chimerism was performed using DNA extracted from granulocytes and T cells with the identification of an informative microsatellite marker (n = 18). Unseparated chimerism was performed on whole blood with no separation of T cells and granulocytes (n = 53). Fluorescent in situ hybridization (FISH) using X- and Y-specific probes was the method of assessing chimerism in some donor-recipient sex mismatched transplants (n = 5). Five patients who were given DLI did not have chimerism studies performed. Where known, peripheral blood (n = 27), marrow (n = 17), or both (n = 29) were analyzed in the specimen prior to the first DLI.
Definitions
A complete hematologic response (CR) means no detectable disease at the morphologic, biochemical, clinical, and radiologic levels. Some patients with CML and follicular NHL who had complete clinical responses were assessed for molecular remission. A partial response (PR) denotes a more than 50% reduction in measurable tumor, a more than 50% reduction in the serum paraprotein level in patients with myeloma, or a response to less than 10% blasts in patients with acute leukemia. Status at transplantation was categorized as CR1, CR2, CR3 or higher, PR, sensitive relapse, untested relapse, resistant relapse, or primary refractory disease.
Favorable remission status comprised CR1 and CR2; intermediate status consisted of PR, sensitive relapse and CR3 or higher, whereas untested or resistant relapse and primary refractory disease were categorized as unfavorable. A complete chimeric response was used to denote conversion of MC to full donor chimerism. A partial chimeric response was used to describe a more than 20% increase in the number of donor cells, whatever method was used. Graft hypoplasia was defined as the development of a neutrophil count of less than 1 × 109/L or a platelet count of less than 20 × 109/L in the presence of a hypocellular marrow. Acute GVHD was graded according to the modified Glucksberg criteria15 and chronic GVHD classified as none, limited, and extensive.
There were various indications for DLIs. The most common were for disease relapse or progression or persistent MC. In many cases it was part of the RIC allograft protocol and was given even if there was no overt disease and full donor chimerism. In other cases the chance of relapse was considered high enough to give DLI preemptively.
Statistics
For the patients who received DLI the outcome measures considered were acute GVHD (any or grade II or higher), any chronic GVHD, complete clinical or chimeric responses, where these were evaluable. The proportions with adverse outcomes for various subgroups were compared using χ2 tests; for 2 × 2 tables a continuity correction was used or 2-tailed Fisher exact test where expected frequencies were small. Two-tailed Mann-Whitney Utests were used to compare good and poor responders with respect to continuous variables such as age and maximum DLI dose. Kaplan-Meier survival estimates were calculated from the last date of patient contact and the survival of subgroups of patients were compared using log-rank tests. The Cox proportional hazards model was used to examine the effect of continuous variables on survival. Minimum follow-up of survivors was 5 months from the date of the transplantation.
Results
DLIs and indications
The patients received a total of 130 infusions (median, 1; range, 1-4). The indications for DLI were unsatisfactory response/disease progression in 51 patients, MC in 18, preemptive in 10, and autoimmune hemolytic anemia16 and progressive cytomegalovirus (CMV) retinitis in 1 each (Table 3). Eight patients received DLI as part of the transplantation protocol. Nine had specific antitumoral therapy prior to DLI. Seven of these 9 patients had lymphoma, 1 had myeloma, and 1 had AML. Two patients received local radiotherapy alone and 7 received chemotherapy. Of the 7 who were administered chemotherapy, 3 had additional rituximab, 1 had local radiotherapy, and 1 had thalidomide. Ten patients were being administered immunosuppressive agents at the time of DLI. The reasons for this are as follows: 3 had ongoing or recent GVHD, 1 was given DLI for CMV retinitis, and 1 received DLI to correct MC. In 5 of 10 patients the reason is not known. The median times of the first, second, and third DLIs were 149 days (range, 61-1008 days), 210 days (range, 119-1029 days), and 303 days (range, 105-1111 days) after SCT and the median CD3+ cell doses were 5 × 106/kg (range, 1 × 105-1 × 108/kg), 1 × 107/kg (3 × 106-1 × 108/kg), and 5 × 107/kg (107-108/kg), respectively. Because the patients came from 16 different centers, there was no consistent approach about the timing of DLI and the “gaps” between DLI and the dose-escalation schemes used were highly variable.
DLIs
| Characteristic . | No. . |
|---|---|
| Disease-specific therapy prior to DLIs | 9 |
| First infusion (n = 81) | |
| Days after transplantation | 149 (61-1008) |
| Median cell dose, per kg recipient weight | 5 × 106 (105-108) |
| Second infusion (n = 34) | |
| Days after transplantation | 210 (90-1029) |
| Median cell dose, per kg recipient weight | 1 × 107 (3 × 106-108) |
| Median days between first and second DLI and range | 45 (8-554) |
| Third infusion (n = 15) | |
| Days after transplantation | 303 (105-1111) |
| Median cell dose, per kg recipient weight | 5 × 107 (107-108) |
| Median days between second and third DLI and range | 42 (14-119) |
| Cytokines given after DLI | 0 |
| Donor reaccessed/stored DLI3-150 | 54/26 |
| GVHD prior to first DLI | 14 |
| On immunosuppression at time of first DLI | 10 |
| Indication | |
| Progressive/persistent disease | 51 |
| MC | 18 |
| Preemptive | 10 |
| Other | 2 |
| Pre-DLI chimerism | |
| All donor | 20 |
| MC | 54 |
| ND/NK | 6 |
| All recipient | 1 |
| Characteristic . | No. . |
|---|---|
| Disease-specific therapy prior to DLIs | 9 |
| First infusion (n = 81) | |
| Days after transplantation | 149 (61-1008) |
| Median cell dose, per kg recipient weight | 5 × 106 (105-108) |
| Second infusion (n = 34) | |
| Days after transplantation | 210 (90-1029) |
| Median cell dose, per kg recipient weight | 1 × 107 (3 × 106-108) |
| Median days between first and second DLI and range | 45 (8-554) |
| Third infusion (n = 15) | |
| Days after transplantation | 303 (105-1111) |
| Median cell dose, per kg recipient weight | 5 × 107 (107-108) |
| Median days between second and third DLI and range | 42 (14-119) |
| Cytokines given after DLI | 0 |
| Donor reaccessed/stored DLI3-150 | 54/26 |
| GVHD prior to first DLI | 14 |
| On immunosuppression at time of first DLI | 10 |
| Indication | |
| Progressive/persistent disease | 51 |
| MC | 18 |
| Preemptive | 10 |
| Other | 2 |
| Pre-DLI chimerism | |
| All donor | 20 |
| MC | 54 |
| ND/NK | 6 |
| All recipient | 1 |
ND/NK indicates not done/not known.
Source of DLI not known in one patient.
Toxicity
All patients were considered evaluable for graft hypoplasia and acute GVHD (Table 4). Graft hypoplasia was uncommon (9 of 81 patients = 11%) and usually rapidly reversible. Five of 9 received hematopoietic growth factors but none required additional stem cells. Eight of 9 patients with post-DLI pancytopenia had MC documented prior to the administration of DLI. Three of the 9 patients with pancytopenia after DLI had other possible contributing causes. Grade II to IV GVHD occurred in 27 of 81 patients (33%) and limited and extensive chronic GVHD in 5 of 69 and 18 of 69 evaluable patients, respectively (total incidence of 33%). In total, 36 of 82 patients (44%) experienced either acute or chronic GVHD. The incidence of acute GVHD was not higher in unrelated donors (data not shown) although numbers were small. Seven patients (9%) died of DLI-related GVHD. An analysis of the possible relationship between GVHD and DLI dose in the 71 recipients of sibling allografts is shown in Table 5. We were unable to demonstrate an association between acute, chronic, or any GVHD and the maximum DLI dose delivered. We also assessed the toxicity of DLI given in the first 6 months after SCT because there are few published data concerning this. When the first DLI infusion was given less than 100 days after SCT (n = 19) the chances of acute, chronic, and any GVHD were 37%, 28%, and 53%, respectively. For DLI given less than 180 days (n = 47) after SCT, the chances were 43%, 34%, and 55%, respectively. These figures are not statistically significantly different from patients who received DLI after day 180. More men developed chronic GVHD than did women (41% versus 15%,P = .075). Of the 14 patients who had GVHD prior to the first DLI, 5 developed acute GVHD and 3 chronic GVHD.
Major outcomes of patients who received DLIs
| Characteristic . | No. . |
|---|---|
| Alive/dead | 47/34 |
| Graft hypoplasia | 9 |
| Range of onset after first DLI, d | 42-294 |
| Acute GVHD | 27 (33%) |
| Grade I | 4 |
| Grade II-IV | 23 (28%) |
| Grade III-IV | 12 (15%) |
| Organs affected | |
| Skin | 21 |
| Gut | 11 |
| Liver | 7 |
| Chronic GVHD | 23/69 (33%) |
| Limited | 5 |
| Extensive | 18 |
| Organs affected | |
| Skin | 13 |
| Gut | 8 |
| Liver | 13 |
| Mouth | 5 |
| Eyes | 3 |
| Antecedent acute GVHD | 14 |
| Clinical response | 23/51 (45%) |
| Complete | 18 |
| Partial | 5 |
| Remained in CR | 13 |
| Chimerism response | 25/55 (45%) |
| Complete | 19 |
| Partial | 6 |
| Remained 100% donor | 8 |
| Characteristic . | No. . |
|---|---|
| Alive/dead | 47/34 |
| Graft hypoplasia | 9 |
| Range of onset after first DLI, d | 42-294 |
| Acute GVHD | 27 (33%) |
| Grade I | 4 |
| Grade II-IV | 23 (28%) |
| Grade III-IV | 12 (15%) |
| Organs affected | |
| Skin | 21 |
| Gut | 11 |
| Liver | 7 |
| Chronic GVHD | 23/69 (33%) |
| Limited | 5 |
| Extensive | 18 |
| Organs affected | |
| Skin | 13 |
| Gut | 8 |
| Liver | 13 |
| Mouth | 5 |
| Eyes | 3 |
| Antecedent acute GVHD | 14 |
| Clinical response | 23/51 (45%) |
| Complete | 18 |
| Partial | 5 |
| Remained in CR | 13 |
| Chimerism response | 25/55 (45%) |
| Complete | 19 |
| Partial | 6 |
| Remained 100% donor | 8 |
Relationship between maximum DLI dose and GVHD in 71 sibling RIC allografts
| Maximum dose CD3+ cells received . | Median day after transplantation (range) . | AGVHD . | AGVHD2+ . | Chronic GVHD5-150 . |
|---|---|---|---|---|
| Less than 5 × 106/kg | 125 (76-412) | 4/13 | 3/13 | 3/11 |
| 1 × 107/kg | 179 (75-1111) | 9/25 | 8/25 | 7/21 |
| 2-6 × 107/kg | 210 (99-494) | 9/25 | 7/25 | 8/24 |
| 0.8-3 × 108/kg | 116 (77-455) | 1/8 | 1/8 | 2/7 |
| Maximum dose CD3+ cells received . | Median day after transplantation (range) . | AGVHD . | AGVHD2+ . | Chronic GVHD5-150 . |
|---|---|---|---|---|
| Less than 5 × 106/kg | 125 (76-412) | 4/13 | 3/13 | 3/11 |
| 1 × 107/kg | 179 (75-1111) | 9/25 | 8/25 | 7/21 |
| 2-6 × 107/kg | 210 (99-494) | 9/25 | 7/25 | 8/24 |
| 0.8-3 × 108/kg | 116 (77-455) | 1/8 | 1/8 | 2/7 |
AGVHD indicates acute GVHD, any grade; AGVHD2+, acute GVHD grades II to IV.
Eight patients did not survive 100 days after the DLI.
Hematologic and chimeric responses
Fifty-one patients received DLI for measurable disease following their RIC allograft. Of these, 18 (35%) patients had a CR, of which 2 were molecular, and 5 had a PR. Thus, the total clinical response rate was 45%, whereas 13 additional patients remained in CR at the time of last patient contact. Details of the 18 complete responders are shown in Table 6. The median follow-up of the 15 surviving complete responders is 700 days from SCT. The median time to a complete response was 132 days and the median duration of complete responses in surviving patients is 220 days. All surviving complete responders remain in CR at the time of analysis. Eleven of the 17 complete responders (65%) who had chimerism studies were shown to have full donor chimerism. Eight of 13 patients with overt follicular NHL after SCT had complete responses and 2 additional patients who had preemptive DLI therapy remained in CR. Two patients with follicular lymphoma had very delayed complete responses to DLI (569 and 660 days, respectively), were given single doses of DLI, and received no other antitumoral therapy. Four of 12 patients with CML had complete responses and 3 stayed in CR. Complete response rates were low in CLL (0 of 7, 1 PR), myeloma (0 of 10, 1 PR), and AML (1 of 7).
Details of 18 complete responders
| . | Diagnosis . | Status SCT . | Age . | Sex . | Maximum DLI dose . | Days after SCT . | GVHD . | Days to CR . | Survival, d . | Other/cause death . |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | CML | CP1 | 46 | F | 107 | 227 | AII, CExt | 144 | 477+ | |
| 2 | HD | PR | 36 | M | 106 | 107 | AIII, CExt | 330 | 474+ | |
| 3 | NHL-F | PR | 47 | M | 5 × 106 | 76 | NIL | 660 | 1170+ | MR |
| 4 | NHL-F | SR | 48 | M | 2 × 107 | 311 | AII, CExt | 67 | 1099+ | MR |
| 5 | NHL-F | SR | 44 | M | 3 × 107 | 124 | NIL | 49 | 920+ | |
| 6 | NHL-F | PR | 49 | M | 107 | 358 | AIII, CExt | 120 | 687 | GVHD |
| 7 | NHL-F | RR | 30 | M | 5 × 107 | 354 | AII, CExt | 110 | 464+ | |
| 8 | NHL-HG | SR | 35 | M | 107 | 115 | AIII, CExt | 270 | 803+ | |
| 9 | CML | CP1 | 50 | F | 106 | 547 | NIL | 43 | 677+ | |
| 10 | MDS | UR | 54 | M | 107 | 75 | AIII | 47 | 205 | Fungus |
| 11 | AML | SR | 57 | F | 107 | 100 | AIV, CNE | 53 | 190 | GVHD |
| 12 | CML | CP1 | 45 | F | 106 | 209 | CLim | 80 | 322+ | |
| 13 | CML | CP2 | 57 | M | 106 | 135 | AII, CExt | 182 | 409+ | |
| 14 | NHL-F | SR | 34 | M | 107 | 120 | AIII | 569 | 1037+ | |
| 15 | NHL-F | SR | 50 | F | 107 | 76 | NIL | 93 | 927+ | |
| 16 | T-PLL | PR | 51 | M | 5 × 107 | 168 | CExt | 267 | 700+ | |
| 17 | NHL-F | CR1 | 53 | M | 2 × 107 | 419 | NIL | 102 | 629+ | |
| 18 | NHL-MC | CR3 | 43 | M | 5 × 107 | 317 | AII, CExt | 203 | 844+ |
| . | Diagnosis . | Status SCT . | Age . | Sex . | Maximum DLI dose . | Days after SCT . | GVHD . | Days to CR . | Survival, d . | Other/cause death . |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | CML | CP1 | 46 | F | 107 | 227 | AII, CExt | 144 | 477+ | |
| 2 | HD | PR | 36 | M | 106 | 107 | AIII, CExt | 330 | 474+ | |
| 3 | NHL-F | PR | 47 | M | 5 × 106 | 76 | NIL | 660 | 1170+ | MR |
| 4 | NHL-F | SR | 48 | M | 2 × 107 | 311 | AII, CExt | 67 | 1099+ | MR |
| 5 | NHL-F | SR | 44 | M | 3 × 107 | 124 | NIL | 49 | 920+ | |
| 6 | NHL-F | PR | 49 | M | 107 | 358 | AIII, CExt | 120 | 687 | GVHD |
| 7 | NHL-F | RR | 30 | M | 5 × 107 | 354 | AII, CExt | 110 | 464+ | |
| 8 | NHL-HG | SR | 35 | M | 107 | 115 | AIII, CExt | 270 | 803+ | |
| 9 | CML | CP1 | 50 | F | 106 | 547 | NIL | 43 | 677+ | |
| 10 | MDS | UR | 54 | M | 107 | 75 | AIII | 47 | 205 | Fungus |
| 11 | AML | SR | 57 | F | 107 | 100 | AIV, CNE | 53 | 190 | GVHD |
| 12 | CML | CP1 | 45 | F | 106 | 209 | CLim | 80 | 322+ | |
| 13 | CML | CP2 | 57 | M | 106 | 135 | AII, CExt | 182 | 409+ | |
| 14 | NHL-F | SR | 34 | M | 107 | 120 | AIII | 569 | 1037+ | |
| 15 | NHL-F | SR | 50 | F | 107 | 76 | NIL | 93 | 927+ | |
| 16 | T-PLL | PR | 51 | M | 5 × 107 | 168 | CExt | 267 | 700+ | |
| 17 | NHL-F | CR1 | 53 | M | 2 × 107 | 419 | NIL | 102 | 629+ | |
| 18 | NHL-MC | CR3 | 43 | M | 5 × 107 | 317 | AII, CExt | 203 | 844+ |
Only patient 13 received stem cells from an unrelated donor.
HD, indicates Hodgkin disease; F, follicular; HG, high grade; MDS, myelodysplastic syndrome; T-PLL, prolymphocytic leukemia; MC, mantle cell; CP1, first chronic phase; AII, acute GVHD grade II; CExt, extensive chronic GVHD; NIL, none; MR, molecular remission; SR, sensitive relapse; AIII, acute GVHD grade III; RR, resistant relapse; UR, untested relapse; AIV, acute GVHD grade IV; CNE, not evaluable for chronic GVHD; CLim, limited chronic GVHD; CP2, second chronic phase; and CR3, third complete remission.
The median DLI dose that resulted in a CR was 1 × 107/kg (range, 1 × 106-3 × 107/kg). Thirteen of the 18 complete responders experienced GVHD and 8 of the 13 both acute and chronic GVHD. A complete response was significantly associated with grade II to IV acute GVHD (P = .005), any acute GVHD (P = .010), chronic GVHD (P = .014), and either acute or chronic GVHD (P =.007). Of all the patients who developed either acute or chronic GVHD, 11 had no response and 4 were not evaluable for assessment of response because they were in CR at the time of receiving DLI. Of the 10 patients given DLI preemptively, 7 survive with 4 being in CR. GVHD prior to DLI was associated with a lower chance of a complete response (0% versus 33%,P = .06). Of the 10 patients on immunosuppressive therapy at the time of DLI administration, 2 had complete responses and 4 remained in CR.
Conversion to full donor chimerism occurred in 19 of 55 patients (35%) with documented at least partial recipient chimerism after RIC allograft at a median of 48 days after the DLI (range, 24-690 days); partial (> 20% increase in donor cells) responses were seen in an additional 6 patients. Thus, the total chimeric response rate was 45%. A complete chimeric response was significantly associated with grade II to IV acute GVHD (P < .001), any acute GVHD (P = .006), chronic GVHD (P = 0007), and either acute or chronic GVHD (P < .001).
Survival
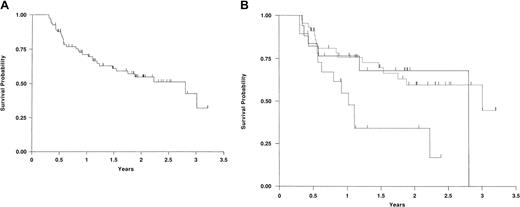
Forty-seven patients (58%) survive at a median follow-up of 508 days after SCT (range, 155-1171 days) with a median Karnofsky score (KS) of 90, but only 5 patients have a KS less than 80. The actuarial survival estimates at 1 and 2 years are 71% (59%-80%, 95% CI) and 55% (42%-66%, 95% CI), respectively (Figure1A). Twenty-six patients (32%) survive in CR. Thirty-two patients (40%) have died at a median of 211 days after transplantation with the major causes being progressive disease (26%) and GVHD (9%).
Kaplan-Meier survival curves.
(A) Kaplan-Meier survival curve of all 81 patients who received DLIs. (B) Kaplan-Meier survival of 81 patients who received DLIs in the favorable, intermediate, and poor remission status groups. Favorable is shown by unbroken line; intermediate, dotted broken line; and poor, dashed broken line. This figure graphically portrays the significantly worse survival of the poor remission status group compared with the other 2 groups.
Kaplan-Meier survival curves.
(A) Kaplan-Meier survival curve of all 81 patients who received DLIs. (B) Kaplan-Meier survival of 81 patients who received DLIs in the favorable, intermediate, and poor remission status groups. Favorable is shown by unbroken line; intermediate, dotted broken line; and poor, dashed broken line. This figure graphically portrays the significantly worse survival of the poor remission status group compared with the other 2 groups.
Disease status is significantly associated with survival (P = .026) with the favorable and intermediate groups showing similar survival curves. This is shown graphically in Figure1B. The only other factor associated with survival is that patients who received therapy prior to DLI had a poorer survival (P = .051). This, of course, may reflect a greater bulk of disease at the time of deciding to give DLI. There was no relationship between disease status and whether treatment was given prior to DLI so a multivariate analysis was not performed.
Discussion
The last 3 years have seen an escalation of RIC transplantations for a variety of diseases in the absence of evidence indicating durable efficacy. Three recent publications from London, Seattle, and Houston, using very different transplantation conditioning protocols, have helped to clarify short-term toxicity and the degree of donor chimeric conversion that can be expected.1-3 Considering the relatively poor prognosis of the patients who received transplants, the reported 1-year survivals of 49% to 67% were encouraging and some documented molecular remissions have indicated that some patients may become long-term survivors but, in 2002, it remains difficult to advise a candidate for an RIC allograft about the chance of being cured.
The RIC SCT has the advantage of being less toxic initially but the potential disadvantages of being less directly tumoricidal and having a higher chance of resulting in incomplete donor chimerism. The use of DLI has been central to the philosophy of many RIC SCT protocols. Some investigators have given DLIs preemptively (as part of the protocol), whereas others have reserved them for disease progression or persistent MC. A major practical difficulty for clinicians was that there were little data concerning the toxicity of early DLIs and certainly no convincing published evidence that giving DLIs is effective.
Before drawing conclusions we should note that this multicenter national study has significant limitations. Although we have sampled more than 50% of the adult transplantation centers in the United Kingdom, we do not know if the data are representative of overall practice. Such limitations are common to much of the information concerning “mini-allografts,” namely, heterogeneity of disease and remission status, short follow-up, and widely varying transplantation protocols. Nonetheless, this study has been performed to generate some large-scale clinical data that may be sufficient to make some broadly based conclusions and recommendations and might enable us to make a subsequent systematic study of DLI.
What can one conclude about toxicity? Grade II to IV acute GVHD was seen in approximately 25% of patients and severe (grade III-IV) acute GVHD in 15%. Chronic GVHD was seen in one third of patients. The incidence of both acute and chronic GVHD, while substantial, appears to be less than that reported after conventional allografts,12 but many patients in previous studies did not receive a program of incremental doses of DLI. DLI given in the first 6 months after transplantation had a more than 50% chance of resulting in acute or chronic GVHD, but we were unable to demonstrate an association between the maximum CD3+ cell dose given and the development of GVHD. However, this latter finding should be interpreted with caution because of the relatively small numbers of patients at each dose level. The only factor predictive of GVHD was male sex. In contrast to the London study reported at the 2001 American Society of Hematology meeting, we did not find that DLIs from unrelated donors were associated with more GVHD.17 Procedural mortality was low (9%)12and the median KS of survivors is acceptable but more than a third have chronic GVHD, most of it extensive. Irreversible graft hypoplasia did not appear to be a major problem in the dose ranges studied and no patient died of related neutropenic sepsis.
It is harder to be definite about efficacy. DLIs after RIC SCT are associated with a significant response rate but the median response duration of complete responders is only 7 months and relapse after DLI-induced remissions is well documented.8,12 The 81 patients comprise 6 main disease groups with small numbers in each group. The results of DLI are certainly very promising for follicular NHL and CML. It is encouraging that 2 patients with follicular lymphoma are in molecular remission,5 but patients with this disease require prolonged follow-up to determine if they are cured. It is of interest that 2 patients with follicular NHL had very delayed responses to DLI. Thirteen additional patients have been in continuous CR since SCT (which also may be evidence of the efficacy of DLI); however, insufficient numbers of patients were given preemptive DLI to analyze the efficacy of this approach. In total, 26 patients are currently candidates to become long-term disease-free survivors, but additional patients in the cohort may remit with subsequent DLIs. Nearly 75% of complete responders experienced GVHD (most of them both acute and chronic) and this study, similar to previous studies,12 indicated a substantial overlap between a GVM effect and GVHD. Subsequent studies should focus on whether it is possible to identify DLI doses that result in a GVM effect without severe GVHD. In addition, DLI may be of lesser importance after certain transplantation protocols (such as the Seattle regimen3); this requires further study.
It is also of interest that a third of evaluable patients were converted from MC to full donor chimerism. However, further studies are required to determine if it is necessary to give DLIs to patients with persistent MC or whether they should be reserved for patients with overt disease. Patients with persistent MC can experience GVHD7 (and therefore may benefit from a GVM effect), but they may also be at risk of graft rejection although stable MC is seen in some preclinical models.18
In summary, RIC allografting has been piloted in a number of centers and a proportion of patients who would not be eligible for conventional intensity allografts may become long-term disease-free survivors. However, the published data describe a very heterogeneous group of patients and do not provide a sound evidence base for future investigations. It is important now to establish who will benefit most from these transplantations and the optimal ways of performing them. The data in this report provide the information necessary to perform a systematic study of the role of DLI following RIC allografts. Although further follow-up is required to determine if the complete responders are cured, the central premise that RIC allografts can provide a platform for subsequent effective cellular immunotherapy appears to have been confirmed. Nonetheless, many important questions need to be answered. Similar to the New York study in full intensity allografts,9 a phase I study that examines the relationship between CD3+ cell dose and toxicity should be performed. Age, source of stem cells, and possibly other factors such as the RIC regimen may be variables to control for. Similar systematic studies need to be performed for each disease because the efficacy of DLI may depend on the disease being treated. In addition, the role of preemptive therapy in patients with diseases at high risk of relapse requires further evaluation. Some of these studies will require multicenter collaboration to generate sufficient numbers.
We wish to acknowledge the assistance of our data manager, Mr Robert Thorne. We also wish to thank a number of other people who contributed to this study including Nigel Russell, Jane Apperley, Charles Craddock, Suparno Chakrabarti, and numerous data managers and statisticians on whom we rely on a daily basis.
Prepublished online as Blood First Edition Paper, July 5, 2002; DOI 10.1182/blood-2002-02-0506.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
David Marks, Bristol Adult BMT Unit, Bristol Children's Hospital, Upper Maudlin St, Bristol BS2 8BJ, United Kingdom; e-mail: dmarks@nildram.co.uk.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal