Abstract
Hematopoietic progenitor cells in 2 myeloproliferative disorders, juvenile chronic myelomonocytic leukemia and polycythemia vera, are known to be hypersensitive to cytokines that control normal progenitor cell proliferation, differentiation, and survival in their respective granulocyte/macrophage and erythroid lineages. Because thrombopoietin controls these functions in the normal megakaryocytic lineage, we asked the question: Are megakaryocytic progenitor cells in the myeloproliferative disorder essential thrombocythemia (ET) hypersensitive to thrombopoietin? Peripheral blood mononuclear cells from patients with ET, or secondary (reactive) thrombocytosis (2°T), or healthy volunteers were grown in strictly serum-free agarose culture containing interleukin 3 (IL-3) and all-trans-retinoic acid, with various concentrations of PEG-rHu megakaryocyte growth and development factor (MGDF). The concentration of cytokine at half-maximum colony number served as a measure of progenitor cell sensitivity. Hypersensitivity to PEG-rHu MGDF was found in circulating progenitors from 18 of 20 (90%) informative patients with presumptive diagnosis ET, 1 of 8 (12.5%) 2 °T patients, and none of the 22 healthy volunteers. Median MGDF sensitivity ratio in ET patients was approximately 53 times greater than in the controls. This hypersensitivity, which was also directed to rHu thrombopoietin, was highly specific with respect to cytokine, disease, and cell lineage. We propose that, despite their single pluripotential cell origin, the different clinicopathologic phenotypes in different chronic myeloproliferative disorders are determined by lineage-restricted hypersensitivities of hematopoietic progenitor cells to endogenous cytokines. This work emphasizes the importance of stringent serum-free conditions for revealing true sensitivities to cytokines. The findings also offer a basis for evolving a positive test for ET, a diagnosis now made essentially by exclusion.
Introduction
Essential (primary) thrombocythemia (ET), also known as idiopathic thrombocytosis, is a member of the family of chronic myeloproliferative disorders generally believed to originate as a regulatory defect in a single pluripotential myeloid stem cell.1-5 These disorders are characterized by trilineal bone marrow hyperplasia in which the major emphasis is usually on a single lineage only,6,7 in this case, the megakaryocyte/platelet lineage. A pronounced and sustained increase in the number of megakaryocytes and platelets in this condition often leads to thrombosis or hemorrhage.8 The basis for the selective clinicopathologic emphasis on one lineage is at present unknown.
In the clonal myeloproliferative disorders juvenile chronic myelogenous (myelomonocytic) leukemia (JCML/JMML) and polycythemia vera (PV), the granulocyte/macrophage (GM) progenitor cells on the one hand and the erythroid progenitor cells on the other are known to be hypersensitive to specific and different cytokines that control normal proliferation, differentiation, and survival in their respective lineages.9,10 Thrombopoietin (TPO), the ligand for the receptor c-Mpl,11 is considered responsible for controlling proliferation and differentiation of progenitor cells in the normal megakaryocytic lineage.12 Accordingly, we asked the question, are megakaryocytic progenitor cells in ET hypersensitive to TPO?
We began by determining the relative sensitivities of circulating megakaryocytic progenitor cells to PEGylated-rHu megakaryocyte growth and development factor (MGDF) with the help of a strictly serum-free cell culture system for the production of megakaryocytic colonies, based on the use of our basal serum-free medium,13,10,14together with all-trans-retinoic acid (ATRA) and interleukin-3 (IL-3). PEG-rHu MGDF is a truncated and PEGylated human recombinantEscherichia coli-derived c-Mpl ligand related to the thrombopoietin (TPO) group in that it encompasses the erythropoietin (Epo)-like receptor-binding N-terminal domain of TPO, modified by conjugation with polyethylene glycol.15 It is approximately 10 to 20 times more potent in vivo than the unconjugated polypeptide (mainly because of its prolonged half-life), but both have similar activities in vitro.
In a series of preliminary experiments, a systematic investigation of the serum-free system was carried out to define the optimal cell numbers, culture wells, cytokine combinations, matrix characteristics, and duration of culture. These conditions were then used in the subsequent studies on patients with the presumptive diagnosis of ET and appropriate controls to determine the relative sensitivities to PEG-rHu MGDF of their circulating megakaryocytic progenitor cells in culture.
Patients, materials, and methods
Patients
Cell preparation
After informed written consent as approved by the University of Toronto Research Ethics Board, 20 to 50 mL peripheral blood was obtained from healthy donors and patients, and mixed with preservative-free sodium heparin (final concentration 10-20 IU/mL) (No. 820 5077 MF; GIBCO BRL, Burlington, ON). Usually within 2 to 4 hours at room temperature, the heparinized blood was diluted with an equal volume of α-minimum essential medium (α-MEM), and was layered onto Ficoll-Paque (Catalog No. 17-0840-02 Pharmacia Biotech, Ste-Anne-de Bellevue QC, Canada). After centrifugation at 400g, 30 minutes at room temperature, the interface containing the light density peripheral blood mononuclear cells (PBMNCs) was collected in α-MEM containing 0.1% bovine serum albumin (BSA) (see below), the cells were washed twice in the same medium, counted, diluted to the appropriate concentrations, and plated.
Serum-free cell culture
Referred to locally as “serozero stem cell medium” (SCM), the composition of the basal serum-free medium13,10 14 is as follows (all are final concentrations): fatty acid-free and globulin-free (FAF) crystallized BSA 0.7% (A-0281 BSA, Sigma, St Louis, MO), deionized with analytical-grade ion exchange resin (AG501-X8D, BioRad Labs, Richmond, CA); human apo-transferrin 200 μg/mL (T2252 Sigma) fully iron-saturated with FeCl3 × 6 H2O); Lipids: lecithin (L-α-phosphatidylcholine, dipalmitoyl synthetic) 5.3 μg/mL (P6267 Sigma); porcine liver cholesterol, grade 1 5.2 μg/mL (C3137 Sigma); oleic acid (cis-9-octadecenoic acid) 3.7 μg/mL (O1383 Sigma); the 4 deoxy and ribo nucleosides 7.0 μg/mL of each (2′-deoxyadenosine D8558, thymidine T1895, 2′ deoxyguanosine D0901, 2′ deoxycytidine HCI D0776, uridine U3003, adenosine A4036, guanosine G5254, cytidine C4654, Sigma); Iscove's Modified Dulbecco's Medium (IMDM) (17633, Sigma); L-glutamine 311.5 μg/mL in IMDM (2 mmol/L) or α-MEM; α thioglycerol 4.7 mg/L (M2172, Sigma); penicillin G sodium 100 U/mL and streptomycin sulfate 50 μg/mL (15070-089, GIBCO); 2-mercaptoethanol 6.7 × 10−5 mol/L (B444143-70, BDH Inc, Toronto, Canada); calcium chloride dihydrate (CaCl2 × 2H2O) 0.028 mg/mL (C7902, Sigma); α-MEM (TCMP, Faculty of Medicine, University of Toronto); and low gel agarose 0.3% (type VII A-9045, Sigma). For routine megakaryocytic colony culture to the basal medium, all trans-retinoic acid (ATRA) 3 × 10−8 mol/L (R2625, Sigma) and the recombinant cytokines rHu IL-3 and PEG-rHu MGDF were added at the indicated final concentrations. PBMNCs were seeded in triplicate or quadruplicate at 1.5 × 105 cells in 0.7 mL of this medium in flat-bottomed (1.5 × 1.0 cm) plastic mini-macro wells (No. 7600004, Flow Laboratories, McLean, VA, now discontinued), or in quintuplicate at 4 × 104 cells in 0.1 mL of this medium in round-bottomed plastic microwells (Linbro Chemical Co, Hamden, CT, Disposo trays, model MRC 96, now discontinued). We have found Falcon flexible polyvinyl chloride plates cut into trays of round- or-flat-bottomed microwells (Falcon catalog no. 3911 or 3912, Fisher Scientific Ltd, Nepean, ON) satisfactory for this purpose. Experiments numbered 155 to 197 were carried out in the mini-macro wells; those numbered from 198 onward were in microwells. Petri dishes containing the wells were incubated at 37°C in a fully humidified atmosphere and 5% CO2 for 14 to 15 days in mini-macro wells. Harvesting and staining were performed on day 18 for best results with microwells.
Recombinant cytokines
Unless otherwise stated, a concentration of 10 ng/mL rHuIL-3 (R & D Systems, Minneapolis, MN) was used throughout. Cytokines in the specificity experiments were rHu IL-3, granulocyte-macrophage colony-stimulating factor (GM-CSF), granulocyte colony-stimulating factor (G-CSF), stem cell factor (SCF), interleukin-6 (IL-6), interleukin-11 (IL-11), erythropoietin (Epo), and insulinlike growth factor-I (IGF-I), all from R & D. Recombinant human thrombopoietin (rHu TPO) was from Peprotech Inc, Rocky Hill, NJ. PEG-rHu MGDF was a generous gift from AMGEN, Thousand Oaks, CA, through Ms Janet Lee Nichol. All cytokines were carrier-free and diluted in α-MEM + 0.1% FAF BSA.
Collecting and staining agarose gels
Gel cultures were released onto slides, fixed in 5% glutaraldehyde (B28682-74 BDH), washed, air-dried, stained with Giemsa (B35086-74 improved R66 “Gurr,” BDH), dried and mounted in Permount (No. SP15 Fisher Scientific). Megakaryocytic colonies were scored at 80 × magnification, a minimum number of 3 megakaryocytes being considered as a colony, and reaching more than 100 cells per colony. The identity of colonies as being megakaryocytic was confirmed by positive staining with an antibody against gpIIb/IIIa, specific for megakaryocytes and platelets, with the technique of Teramura et al,17 slightly modified. Once the validity of megakaryocytic colony identification was established on the basis of these molecular markers, routine scoring of megakaryocytic colonies was carried out on Giemsa-stained preparations.
Statistical analysis
Comparison of the numbers of ET patients and controls whose progenitor cells exhibited hypersensitivity to PEG-rHu MGDF was made with the Chi-square test. In the experiments carried out in the absence of added PEG-rHu MGDF, the mean numbers of megakaryocytic colonies in ET and normal cultures were compared by Student t test and by the paired t test.
Results
Relative sensitivities to PEG-rHu MGDF of circulating megakaryocytic progenitor cells from patients with presumptive diagnosis of essential thrombocythemia and controls (ie, healthy volunteers and secondary thrombocytosis patients)
Figure 1 shows the results of 2 representative experiments in which PBMNCs from an ET patient (upper part of the figure) and a 2°T patient (lower part of the figure) were compared with those of healthy volunteers, with respect to megakaryocytic colony formation. PBMNCs were seeded in serum-free agarose culture in the presence of fixed concentrations of IL-3 (10 ng/mL) and ATRA (3 × 10−8 mol/L) and various concentrations of PEG-rHu MGDF. In each group, the number of colonies that developed (mean ± SEM) was plotted against the log10 concentration of the cytokine (Figure 1A).
Megakaryocytic colony formation in culture by ET, 2°T, and healthy circulating progenitor cells as a function of PEG-rHu MGDF concentration.
PBMNCs from an ET patient (upper panels) and a 2°T patient (lower panels) (▴) are compared with those of healthy volunteers (▪) with respect to the numbers of megakaryocytic colonies (mean ± SEM) of quintuplicate samples (A) and the percentage maximum of megakaryocytic colonies (B) produced at various concentrations of PEG-rHu MGDF. The cell number plated in microwells (upper panels) was 4 × 104 PBMNCs per 0.1 mL culture (experiment 218). The cell number plated in mini-macro wells (lower panels) was 1.5 × 105 cells per 0.7 mL culture (experiment 188).
Megakaryocytic colony formation in culture by ET, 2°T, and healthy circulating progenitor cells as a function of PEG-rHu MGDF concentration.
PBMNCs from an ET patient (upper panels) and a 2°T patient (lower panels) (▴) are compared with those of healthy volunteers (▪) with respect to the numbers of megakaryocytic colonies (mean ± SEM) of quintuplicate samples (A) and the percentage maximum of megakaryocytic colonies (B) produced at various concentrations of PEG-rHu MGDF. The cell number plated in microwells (upper panels) was 4 × 104 PBMNCs per 0.1 mL culture (experiment 218). The cell number plated in mini-macro wells (lower panels) was 1.5 × 105 cells per 0.7 mL culture (experiment 188).
To deal with the problem of heterogeneity of colony-forming efficiencies among different cell populations being compared, the data were normalized by conversion of the actual numbers of colonies observed to percentages of the maximum number of colonies that each of the cell populations achieved under the conditions of the same experiment (Figure 1B). The measure of PEG-rHu MGDF sensitivity of progenitor cells used was the concentration of PEG-rHu MGDF at which megakaryocytic colony formation was at half-maximum. The ratio of this value for the normal to the value for the ET or 2°T patient (expressed as the number of times normal) was taken as the PEG-rHu MGDF sensitivity ratio.
In the upper part of Figure 1, it can be seen that there was a shift to the left of the ET curve, indicating an increased sensitivity of the megakaryocytic progenitors to PEG-rHu MGDF relative to that of the normal. Concentrations of cytokine at half-maximum colony number were less than 0.16 and 73 ng/mL for the ET patient and the normal, respectively, a PEG-rHu sensitivity ratio of over 456 × normal (experiment 218). In the lower part of the figure, the half-maximum cytokine concentrations were 58 and 66 ng/mL for the 2°T patient and the normal, respectively, a PEG-rHu MGDF sensitivity ratio of 1.1 × normal, indicating that the progenitor cells of the 2°T patient and those of the normal had similar sensitivities to this ligand (experiment 188).
Figure 2 shows the data from 11 ET patients and 11 healthy volunteers, in which the determinations of PEG-rHu MGDF sensitivity for each pair were performed on the same day. The curve for ET is shifted to the left, indicating a pronounced increase over the normal in sensitivity of the progenitor cells to PEG-rHu MGDF.
Relative sensitivities to PEG-rHu MGDF of circulating megakaryocytic progenitor cells from patients with ET and healthy subjects, determined in strictly serum-free culture.
Cells from 11 ET patients (▴) and 11 healthy subjects (▪) were simultaneously compared with respect to numbers of megakaryocytic colonies produced as a function of PEG-rHu MGDF concentration and the data were normalized as percentage of maximum colony number (mean ± SEM ); see Table 2 for details. The first 6 samples were performed in quadruplicate mini-macro wells at 1.5 × 105 cells per 0.7 mL medium; the last 5 were performed in quintuplicate microwells at 4 × 104 cells per 0.1 mL medium.
Relative sensitivities to PEG-rHu MGDF of circulating megakaryocytic progenitor cells from patients with ET and healthy subjects, determined in strictly serum-free culture.
Cells from 11 ET patients (▴) and 11 healthy subjects (▪) were simultaneously compared with respect to numbers of megakaryocytic colonies produced as a function of PEG-rHu MGDF concentration and the data were normalized as percentage of maximum colony number (mean ± SEM ); see Table 2 for details. The first 6 samples were performed in quadruplicate mini-macro wells at 1.5 × 105 cells per 0.7 mL medium; the last 5 were performed in quintuplicate microwells at 4 × 104 cells per 0.1 mL medium.
We have arbitrarily defined hypersensitivity to PEG-rHu MGDF as greater than or equal to a 10-fold reduction in the concentration of this cytokine at half-maximum colony number compared with that of healthy controls. The median concentration of PEG-rHu MGDF at half-maximum colony number for 21 quantitatively informative healthy subjects was found to be 61.5 ng/mL; for 20 ET patients, it was 0.80 ng/mL; and for 8 2°T patients, it was 70.0 ng/mL. That is, the circulating megakaryocytic progenitor cells from ET patients had a median PEG-rHu MGDF sensitivity ratio that was approximately 53 times greater than that of healthy volunteers or 2°T patients. Table3 gives the overall experience; it shows that of 26 patients with a presumptive diagnosis of ET, 3 had low and flat responses, ie, they had low colony-forming efficiencies and their colony numbers did not significantly increase with increasing concentration of cytokine. Three showed high and flat responses, ie, in the absence of added PEG-rHu MGDF, they developed significant numbers of megakaryocytic colonies, and with increasing concentrations of PEG-rHu MGDF, they did not show increased numbers of colonies. Their cells thus appeared to be independent of added PEG-rHu MGDF and to show reduced or absent sensitivity to the cytokine. No cells from healthy subjects exhibited low and flat responses; one healthy subject showed a high and flat response. The basis for these responses is still unknown. Thus, 20 patients with a presumptive diagnosis of ET were quantitatively informative. Of these, 18 (90.0%) patients had circulating megakaryocytic progenitor cells that were hypersensitive to PEG-rHu MGDF in culture (Χ2 ET compared with healthy volunteers = 30.76, P ≪ .001; Χ2 ET compared with 2°T = 13.78, P < .001). Of 9 patients with a presumptive diagnosis of 2°T, one had a high and flat response, leaving 8 patients who were quantitatively informative. Of these, one showed hypersensitivity to PEG-rHu MGDF. On review, this patient was found to have had gastric lymphoma and had undergone gastrectomy and splenectomy, which prevented knowing whether his spleen had been enlarged; he had a high white blood cell count and a very high platelet count. There were no instances of PEG-rHu MGDF hypersensitivity among the 22 healthy subjects.
The frequency distribution of PEG-rHu MGDF concentrations at half-maximal colony number among megakaryocytic progenitor cells from patients with ET, 2°T, or healthy volunteers is given in Figure3. It is evident that the values for ET are clustered far to the left, whereas those of 2°T patients, which are in the same range as those of the healthy volunteers, are far to the right of the ET distribution. Nevertheless, there are outliers, ie, those that appear not to belong to the same population as the majority. Whether these represent true subgroups or incorrect diagnoses remains to be determined.
Frequency distribution of sensitivities to PEG-rHu MGDF.
Histogram showing frequency distribution of sensitivities to PEG-rHu MGDF (concentration in ng/mL at 1/2 max colony number) of megakaryocytic progenitor cells in 3 conditions: normal, secondary (reactive) thrombocytosis (2°T), and ET. (Note that the point on the abscissa designated as 0.16 ng/mL actually represents a cytokine concentration of ≤ 0.16 ng/mL).
Frequency distribution of sensitivities to PEG-rHu MGDF.
Histogram showing frequency distribution of sensitivities to PEG-rHu MGDF (concentration in ng/mL at 1/2 max colony number) of megakaryocytic progenitor cells in 3 conditions: normal, secondary (reactive) thrombocytosis (2°T), and ET. (Note that the point on the abscissa designated as 0.16 ng/mL actually represents a cytokine concentration of ≤ 0.16 ng/mL).
We conclude from all these findings that megakaryocytic progenitor cells in ET have a highly significantly increased sensitivity to PEG-rHu MGDF compared with those of patients with 2°T or healthy volunteers.
Nature of the cell populations responding to PEG-rHu MGDF
The possibility existed that PEG-rHu MGDF acted indirectly on megakaryocytic progenitor cells by stimulating accessory cells among the PBMNCs to release a factor(s) that could directly influence the progenitor cells to produce megakaryocytic colonies. To deal with this possibility, we compared the sensitivities to PEG-rHu MGDF of nonadherent with total PBMNCs from an ET patient. Figure 6shows that the responding cells in the 2 preparations had identical sensitivities to PEG-rHu MGDF. This finding is consistent with the hypothesis that the action of PEG-rHu MGDF on megakaryocytic progenitor cells is direct and not via accessory cells.
Megakaryocytic colony sizes and morphology
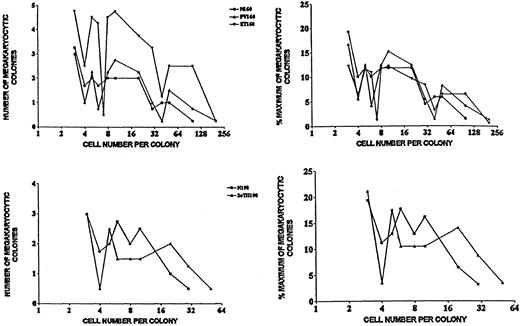
As mentioned in “Patients, materials, and methods,” a cluster of a minimum of 3 megakaryocytes was considered as a megakaryocytic colony. To determine the frequency distribution of megakaryocytic colony sizes in cultures of circulating healthy, ET, and PV progenitor cells, the numbers of cells per colony were counted in all colonies of each of 4 cultures per group for the 3 groups on Giemsa-stained preparations. The results are presented in Figure4. Each group shows a multimodal distribution of cell numbers per colony, a wide range of colony sizes, and a similarity of size distributions among the different groups. Absolute numbers of cells per colony differ among groups, but their distributions within groups are remarkably similar (Figure 4). Similar data were obtained in a separate comparison of cells from healthy subjects and 2°T patients. Microscopic appearances were not appreciably different in the various groups either (Figure 5), although a round or oval cytoplasmic region was frequently prominent in the ET, 2°T, and PV megakaryocytes, but not in the healthy ones.18
Frequency distributions of megakaryocytic colony sizes in cultures of healthy, ET and PV (experiment 160) and healthy and 2°T (experiment 198) progenitor cells.
Abscissa: cell number per colony; ordinate: number of megakaryocytic colonies (per 1.5 × 105 PBMNCs per 0.7 mL) with a given number of cells per colony.
Frequency distributions of megakaryocytic colony sizes in cultures of healthy, ET and PV (experiment 160) and healthy and 2°T (experiment 198) progenitor cells.
Abscissa: cell number per colony; ordinate: number of megakaryocytic colonies (per 1.5 × 105 PBMNCs per 0.7 mL) with a given number of cells per colony.
Individual megakaryocytic colonies.
Photomicrographs from healthy volunteers (A, 30 ×; B, 480 ×); 2°T (C, 30 ×; D, 480 ×), and ET (E, 30 ×; F, 480 ×) patients. (G) A healthy megakaryocytic colony composed of 3 megakaryocytes (480 ×).
Individual megakaryocytic colonies.
Photomicrographs from healthy volunteers (A, 30 ×; B, 480 ×); 2°T (C, 30 ×; D, 480 ×), and ET (E, 30 ×; F, 480 ×) patients. (G) A healthy megakaryocytic colony composed of 3 megakaryocytes (480 ×).
Specificity of the hypersensitivity of megakaryocytic progenitor cells to PEG-rHu MGDF in essential thrombocythemia
The sensitivities of progenitor cells to each of the 9 cytokines were determined. Table 4 shows the results.
When the basal serum-free medium had no IL-3 added, no megakaryocytic colonies developed. When PBMNCs were exposed to IL-3 (10 ng/mL) alone in the same medium, a very small number of megakaryocytic colonies developed, and their numbers were slightly greater in ET than in healthy volunteers. At low IL-3 concentrations, even in ET, the number of colonies was extremely low. Thus, titrations of IL-3 alone could not provide sufficient numbers of megakaryocytic colonies to allow determination of the relative IL-3 sensitivities of their progenitor cells from ET patients. IL-3 therefore had to be evaluated by titration in the presence of PEG-rHu MGDF. PEG-rHu MGDF (100 ng/mL) alone gave few or no megakaryocytic colonies, and any differences between ET patients and healthy subjects were not statistically significant. When PEG-rHu MGDF was included in the cultures in which IL-3 was being titrated, megakaryocytic colony numbers were found to increase with increasing concentration of IL-3 and were higher in ET patients than in healthy subjects (Figure7). However, the concentration of IL-3 at half-maximum number of megakaryocytic colonies remained the same for ET patients as for healthy subjects (Tables 4 and 5). Thus, although MGDF and IL-3 evidently interacted with respect to colony formation, megakaryocytic progenitor cells showed no evidence of hypersensitivity to IL-3 in this myeloproliferative disorder.
Comparison of sensitivities to PEG-rHu MGDF of megakaryocytic progenitor cells among total PBMNC and nonadherent PBMNC from an ET patient (RB).
Total PBMNC, ▪; nonadherent PBMNC, ▴. (A) Number of megakaryocytic colonies per 4 × 104 PBMNCs in 0.1 mL medium as a function of PEG-rHu MGDF concentration. (B) Same relationship expressed as percentage maximum of megakaryocytic colonies. Values shown are mean ± SEM colony numbers derived from quintuplicate cultures (experiment. 230).
Comparison of sensitivities to PEG-rHu MGDF of megakaryocytic progenitor cells among total PBMNC and nonadherent PBMNC from an ET patient (RB).
Total PBMNC, ▪; nonadherent PBMNC, ▴. (A) Number of megakaryocytic colonies per 4 × 104 PBMNCs in 0.1 mL medium as a function of PEG-rHu MGDF concentration. (B) Same relationship expressed as percentage maximum of megakaryocytic colonies. Values shown are mean ± SEM colony numbers derived from quintuplicate cultures (experiment. 230).
Effect of IL-3 on megakaryocytic colony formation in presence of different concentrations of PEG-rHu MGDF.
(A) Healthy PBMNCs. (B) ET PBMNCs. ▪, 0 MGDF; ▴, 4 ng/mL MGDF; ▾, 20 ng/mL MGDF; ♦, 100 ng/mL MGDF. Values shown are mean ± SEM colony numbers derived from quintuplicate cultures seeded with 4 × 104 PBMNCs in 0.1 mL medium (experiment 255).
Effect of IL-3 on megakaryocytic colony formation in presence of different concentrations of PEG-rHu MGDF.
(A) Healthy PBMNCs. (B) ET PBMNCs. ▪, 0 MGDF; ▴, 4 ng/mL MGDF; ▾, 20 ng/mL MGDF; ♦, 100 ng/mL MGDF. Values shown are mean ± SEM colony numbers derived from quintuplicate cultures seeded with 4 × 104 PBMNCs in 0.1 mL medium (experiment 255).
GM-CSF (10 ng/mL) alone gave very few colonies. In the presence of IL-3 or PEG-rHu MGDF, it yielded sensitivity ratios of less than 3-fold greater than normal.
G-CSF, Epo, IGF-I, IL-6, and IL-11 gave low colony numbers in both ET patients and healthy volunteers, even in the presence of IL-3 or PEG-rHu MGDF. Progenitor cells showed no evidence of hypersensitivity to any of these cytokines.
PEG-rHu MGDF alone gave so few megakaryocytic colonies that no estimate of progenitor cell sensitivity could reliably be made. When PEG-rHu MGDF was titrated in the presence of IL-3 at plateau concentration (10 ng/mL), the results were striking. They showed clear and consistent evidence of hypersensitivity of megakaryocytic progenitors to PEG-rHu MGDF in ET patients over healthy subjects (Table 4). This observation raised the question as to whether the effect of IL-3 was specific or whether IL-3 could be replaced by another cytokine for this particular function.
SCF was found capable of replacing IL-3 as the fixed cytokine, and hypersensitivity to MGDF was still seen (data not shown). Thus, the ability to reveal the hypersensitivity to MGDF is not a property specific to IL-3. Moreover, SCF and PEG-rHu MGDF exhibited synergism.18 There was no evidence of hypersensitivity to SCF in ET patients (Tables 4 and 5).
Table 5 shows the results of an experiment in which the sensitivities of nonadherent healthy and ET megakaryocytic progenitor cells to several different cytokines were determined. It is evident under these conditions, as well as when totalmegakaryocytic progenitor cells were examined, that there was clear evidence of hypersensitivity to PEG-rHu MGDF in ET patients over healthy subjects. Thus, of the 9 cytokines examined, megakaryocytic progenitors in ET were definitely hypersensitive only to PEG-rHu MGDF. The data in Table 4 also show that the ET progenitor cells were hyperresponsive19 as well as hypersensitive to PEG-rHu MGDF. In contrast, PEG-rHu MGDF had no effect on GM or erythroid colony formation by healthy or ET progenitors at any concentration tested from 0.16 to 200 ng/mL (data not shown).
Eight patients with PV were investigated; none had cells showing hypersensitivity to PEG-rHu MGDF (Table 3).
Comparison of megakaryocytic colony formation by progenitors from essential thrombocythemia patients and healthy subjects exposed to PEG-rHu MGDF or rHu thrombopoietin
To investigate the possibility that the observed effects of PEG-rHu MGDF seen in the present experiments were due to some property of this cytokine preparation other than that resulting from its relationship to TPO, we compared megakaryocytic colony formation by healthy and ET progenitor cells exposed to either PEG-rHu MGDF or rHu TPO. The results (Figure 8) show that the behavior of the 2 cytokines under these conditions did not differ significantly from one another. Thus, data obtained with PEG-rHu MGDF regarding colony formation by megakaryocytic progenitors may be considered to reflect sensitivities of these cells to TPO as well as to MGDF.
Comparison of sensitivities to PEG-rHu MGDF and TPO of healthy and ET megakaryocytic progenitor cells.
▪, PBMNCs from healthy control subject exposed to PEG-rHu MGDF. (A) Number of megakaryocytic colonies per 4 × 104 PBMNCs in 0.1 mL medium as a function of cytokine concentration. (B) Number of megakaryocytic colonies expressed as percentage of maximum as a function of cytokine concentration. Concentration of MGDF at half-maximum colony number was 30 ng/mL. ▾, PBMNCs from healthy control subject exposed to rHu TPO. Concentration of TPO at half-maximum colony number was 27 ng/mL. ▴, PBMNC from ET patient (SW) exposed to PEG-rHu MGDF. Concentration of MGDF at half-maximum colony number was 2.3 ng/mL. MGDF sensitivity ratio (N/ET) = 13-fold greater than normal. ♦, PBMNCs from ET patient (SW) exposed to rHu TPO. Concentration of TPO at half-maximum colony number was 1.6 ng/mL. TPO sensitivity ratio (N/ET) = 16.9-fold greater than normal (experiment 262).
Comparison of sensitivities to PEG-rHu MGDF and TPO of healthy and ET megakaryocytic progenitor cells.
▪, PBMNCs from healthy control subject exposed to PEG-rHu MGDF. (A) Number of megakaryocytic colonies per 4 × 104 PBMNCs in 0.1 mL medium as a function of cytokine concentration. (B) Number of megakaryocytic colonies expressed as percentage of maximum as a function of cytokine concentration. Concentration of MGDF at half-maximum colony number was 30 ng/mL. ▾, PBMNCs from healthy control subject exposed to rHu TPO. Concentration of TPO at half-maximum colony number was 27 ng/mL. ▴, PBMNC from ET patient (SW) exposed to PEG-rHu MGDF. Concentration of MGDF at half-maximum colony number was 2.3 ng/mL. MGDF sensitivity ratio (N/ET) = 13-fold greater than normal. ♦, PBMNCs from ET patient (SW) exposed to rHu TPO. Concentration of TPO at half-maximum colony number was 1.6 ng/mL. TPO sensitivity ratio (N/ET) = 16.9-fold greater than normal (experiment 262).
Megakaryocytic colony formation by essential thrombocythemia progenitor cells in the absence of added PEG-rHu MGDF
Some of the cell cultures from ET patients and controls developed small numbers of megakaryocytic colonies, although their progenitor cells had not been exposed to exogenous PEG-rHu MGDF. In this regard, it needs to be said first that in our cultures of healthyPBMNCs to which no PEG-rHu MGDF had been added, we regularly observed the development of small numbers of megakaryocytic colonies. These colonies are probably due to the stimulation of megakaryocytic progenitor cell growth by IL-3 and ATRA (which were included in our complete serum-free medium to improve colony-forming efficiencies), because such background colonies never developed in our basal serum-free medium, which lacks these 2 growth factors (D.E., A.A., unpublished observations). The number of colonies that developed in the absence of added PEG-rHu MGDF was significantly greater in ET cultures than in normal cultures (.01 > P > .001, Table6). This phenomenon could therefore not have been related to the culture medium. Because megakaryocytic progenitor cells from ET patients were found to be markedly hypersensitive to PEG-rHu MGDF (Figures 2 and 3, Tables 4 and 5), the presence in ET cultures of even minute amounts of this growth factor could have been responsible for inducing their development. A plausible interpretation would be that the progenitor cells were responding to minute amounts of endogenously produced MGDF. This could have come from stromal cells,20,21 or perhaps from megakaryocytes.22 23
Discussion
Properties of the hypersensitivity to PEG-rHu MGDF in essential thrombocythemia
The main finding in this work is that circulating megakaryocytic progenitor cells in ET, when assayed in vitro, were hypersensitive to PEG-rHu MGDF. The statistically pronounced distinction between ET patients on the one hand, and 2°T patients and healthy volunteers on the other, with respect to sensitivity to PEG-rHu MGDF, was striking. Median sensitivity of megakaryocytic progenitors to PEG-rHu MGDF in ET was approximately 53 times greater than that of healthy volunteers or 2°T patients, but it could reach as high as more than 700 times normal. The 2°T is an umbrella diagnosis covering a number of different diseases that have in common only the fact that patients all have increases in platelet numbers in their blood.8 Thus, despite the heterogeneity of their diseases of origin (Table 1), the 2°T patients constituted a remarkably homogenous group with respect to their having normal PEG-rHu MGDF sensitivity. Hypersensitivity to PEG-rHu MGDF was not seen in any of the 22 healthy volunteers. In addition, among 8 patients with PV, none showed hypersensitivity to PEG-rHu MGDF. Among the 9 cytokines investigated, pronounced hypersensitivity of megakaryocytic progenitors in ET was confined to PEG-rHu MGDF (Tables 4 and 5). However, PEG-rHu MGDF did not influence GM or erythroid colony formation by progenitor cells from ET patients (data not shown). These observations provide strong evidence that the PEG-rHu MGDF hypersensitivity of megakaryocytic progenitors in ET is highly specific with respect to disease, to cytokine, and to cell lineage. This, as far as we know, is the first observation made under stringent serum-free conditions of specific hypersensitivity to PEG-rHu MGDF in ET. Moreover, the hypersensitivity was shown to be equally a function of the normal cytokine, TPO (Figure 7); it was not an artifact of the recombinant cytokine preparation.
When we compared the sensitivities to PEG-rHu MGDF of total ET PBMNCs with those of ET PBMNCs from which the vast majority of adherent cells had been removed, we found very similar sensitivities to the cytokine (Figure 4). This strongly suggests, but does not prove, that the high level of sensitivity to PEG-rHu MGDF in ET is a property of the megakaryocytic progenitor cells themselves and is not determined by accessory cells. Nichol et al18 showed that rHu MGDF stimulates the production of megakaryocytic colonies from healthy CD34+ cells. Experiments underway currently in our laboratory with CD34+ cells isolated24 from patients may help to settle the question as to whether PEG-rHu MGDF acts directly or indirectly in ET.
Definite hypersensitivity to PEG-rHu MGDF in ET could be demonstrated only when this cytokine was titrated in the presence of IL-3, because PEG-rHu MGDF alone gave very poor megakaryocytic colony growth.
We detected no hypersensitivity to IL-3 itself. This finding is in disagreement with that of Kobayashi et al25 who reported that nonadherent cells from patients with ET had maximal megakaryocytic colony growth at IL-3 concentrations considerably lower than those for healthy subjects or those with 2°T. However, the conditions under which the 2 experiments were carried out were not identical: the Kobayashi experiment used ASF 101 medium26 with BSA from the Sigma Co, but with no mention of which of their BSA preparations was used; our experiment used SCM , with Sigma fatty acid-free and globulin-free BSA,13,10,14 which we had previously shown not to contain growth factor(s) capable of stimulating megakaryocytic colony formation. Zauli et al27 also failed to confirm hypersensitivity to IL-3 in ET in fibrin clot assays of either CFU-MK or BFU-MK from bone marrow, although the numbers of both progenitors were increased in response to this cytokine.
We observed the hypersensitivity to PEG-rHu MGDF in ET at both 14 and 21 days of culture. Therefore, the stage of differentiation at which the alteration leading to the PEG-rHu MGDF hypersensitivity occurred in this disorder must have been earlier than either the CFU-MK28 or the BFU-MK.29 Despite this, the hypersensitivity phenomenon exhibited striking specificity with respect to cell lineage: MGDF hypersensitivity was seen in relation to megakaryocytic but not granulocyte/macrophage or erythroid colony formation. Thus, the decision to become hypersensitive must have been taken after differentiation into the various myeloid cell lineages had occurred.
Recently, it has been found30 31 that, in hematopoietic stem and progenitor cells self-renewing in the presence of IL-3 before commitment to a particular cell lineage is made, receptors specific to each of the various cell lineages are expressed at a low level. During unilineal commitment and differentiation, inappropriate gene expression is silenced by down-regulation, whereas genes for the appropriate cell lineage are up-regulated. It is tempting to speculate that the time when hypersensitivity to specific cytokines develops may coincide with the time when these selective developmental decisions are made.
Some hypotheses regarding possible mechanism(s) of MGDF/TPO hypersensitivity
Theoretically, the hypersensitivity to MGDF/TPO that we have observed in vitro in progenitor cells of patients with ET could reflect an elevated level of TPO in these patients before the cells were cultured. However, TPO levels are generally not significantly elevated in ET. They have been reported in a number of studies as being either in the normal range (in most) or only slightly elevated (in a minority).32-39 Serum TPO levels in ET do not appear to be appropriately down-regulated as are their counterparts in other myeloproliferative disorders such as PV and CML. In ET, these levels are not decreased even in the presence of high platelet counts.39
It is also possible that not the level of TPO but its rate of metabolism could be elevated in the ET cells, and this might give rise to MGDF/TPO hypersensitivity. In keeping with such a notion is the observation that circulating TPO levels showed a tendency toward an increase after therapy in some ET patients,36 but this did not reach statistical significance.
Biologic responses are often mediated by receptor trans-signaling mechanisms involving 2 or more receptor systems. Hypersensitivity to MGDF/TPO could possibly occur as a result of different signaling pathways merging through receptor trans-signaling.40 Stem cell factor and MGDF/TPO signaling pathways, for instance, could theoretically merge and give rise to a hypersensitive response, given that stem cell factor and MGDF/TPO act synergistically.18
Spontaneous megakaryocytic colonies
Spontaneous megakaryocytic colony formation has been previously reported in blood and bone marrow cell cultures from patients with ET,41,42 and c-Mpl, the receptor for TPO, has been implicated: c-Mpl messenger RNA (mRNA) was detected in cultured cells from ET patients who showed spontaneous megakaryocyte colony formation in semisolid culture, but not in the cells of those who showed no such colonies, and antisense oligonucleotide to c-Mpl decreased spontaneous megakaryocyte growth.42 Spontaneous megakaryocyte colony formation disappeared, presumably because of a lack of ligand, when serum-free cultures were used with highly purified CD34+cells.42 Antibody to IL-3, IL-6, or GM-CSF did not eliminate spontaneous megakaryocyte colony formation.43Recently, Taksin et al44 have found that in ET, soluble Mpl receptor markedly and specifically inhibited spontaneous megakaryocytic growth, but neutralizing antibody against Mpl ligand did not, suggesting an intracellular site of the interaction. The inhibition was reversed by the addition of PEG-rHu MGDF. The authors concluded that spontaneous megakaryocytic growth in ET is not related to autocrine stimulation by Mpl ligand. However, our findings of striking hypersensitivity of ET megakaryocytic progenitors to PEG-rHu MGDF under strictly serum-free conditions, together with the development of megakaryocytic colonies in the absence of added ligand under the same conditions (Table 6), make it likely that MGDF and its receptorare implicated in the pathogenesis of ET, presumably through a response to endogenously produced ligand.
Serum-free megakaryocytic colony culture
We believe that the use of a strictly serum-free culture medium was critical for showing that megakaryocytic progenitor cells in ET are hypersensitive to PEG-rHu MGDF. Serum is known to contain TPO in variable amounts. Had TPO not been eliminated in the present experiments, there could have been a constant risk of confusion between the concentration of exogenous TPO added to the cultures (the basis of our assay method for sensitivity of megakaryocyte progenitor cells to this cytokine) and the amount of endogenous TPO already present in the cultures before the exogenous TPO was added. Ensuring that the culture medium was stringently serum-free guaranteed that this variable amount of endogenous TPO was eliminated, and with it the confusion it could have engendered.
It is not unusual in publications to see culture media referred to as serum-free when certain of the medium components, eg, albumin, may be known to be contaminated with serum-derived growth-promoting factors. Judgement as to whether a medium is in fact serum-free requires that the complete recipe for the medium and the sources of its constituents are explicitly disclosed. Our basal medium (which contains Sigma fatty acid-free, globulin-free BSA) does not promote megakaryocytic colony formation, and thus all growth observed in the cultures could be related directly to the cytokines added.
Hypersensitivity to different cytokines in the different clonal myeloproliferative disorders
This study brings to 3, of 3 so far investigated, the number of clonal chronic myeloproliferative disorders that have in common the property of hypersensitivity to a cytokine that is capable of stimulating proliferation, differentiation, and survival of normal progenitors in their respective lineages. First, in juvenile chronic myelogenous/myelomonocytic leukemia (JCML/JMML),34 a rare, clonal,5 fatal, pediatric disease, arising at the myeloid stem cell level or earlier, Emanuel et al9 found that dose-response curves relating GM colony number to concentration of ligand in culture showed hypersensitivity to GM-CSF around 10 times normal, whereas sensitivities to IL-3 and G-CSF remained unchanged. Activation of p21RAS is an essential component of the proliferative response to many hematopoietic growth factors,46 and the GM-CSF hypersensitivity seen in JCML/JMML is believed to be a direct consequence of Ras activation in a susceptible myeloid progenitor.47,48 This was the first demonstration of a myeloid leukemia in which hypersensitivity to a specific growth factor appeared to be involved in the pathogenesis of the disease.9
Second, Correa et al10 found that in PV, erythroid progenitor cells were hypersensitive to IGF-I, but not to Epo. IGF-I, like Epo, stimulates erythropoiesis.49 IGF-I sensitivities were at least 100 times,10 and as high as more than 10 000 times greater than normal (D.E., A.A., unpublished observations). Under conditions that were not strictly serum-free, sensitivities of purified blood and marrow BFU-E to IL-3 of 117 and 38 times normal, respectively,50,51 and of purified marrow BFU-E to GM-CSF of 48 times normal51 have been reported in PV by the Krantz group.
We have provided evidence that an aberration in the IGF-I receptor signal transduction pathway is somehow involved in PV, because monoclonal antibody to the receptor inhibited the hypersensitive response to ligand,10 and tyrosine phosphorylation of the IGF-I receptor β subunit occurred earlier, reached greater intensity, and responded to substantially lower ligand concentration in PV than it did in healthy volunteers.52 Moreover, enhanced tyrosine phosphorylation of the IGF-I receptor β subunit in PV could be demonstrated even in the absence of added ligand.52However, in a systematic examination of cDNA sequence, we found no evidence of mutation or deletion in the β- or α-subunit of the IGF-I receptor gene in this disorder (W.Y. Jia, L. Zhou, T. Zhang, A.A. Axelrad, unpublished observations).
Third, in ET, megakaryocytic progenitor cells appeared to be approximately 53 times as sensitive as healthy volunteers to PEG-rHu MGDF, and this hypersensitivity was highly specific (Tables 4 and 5). Mutation or deletion in the c-Mpl receptor gene appears also to have been ruled out as a basis for the cytokine hypersensitivity in this disorder.53 44
As one mechanism underlying the selective amplification of different progenitor cells in the different myeloproliferative disorders, despite their origin in a single pluripotential stem cell, we propose a lineage-restricted hypersensitivity of hematopoietic progenitor cells to normal/endogenous cytokines. This notion could provide an attractive explanation for the unique clinicopathologic emphasis seen in each of these disorders: Acquisition of a lineage-restricted hypersensitivity to cytokine could bring about amplification of those cells whose progenitors had already become committed to a particular lineage; it could therefore be responsible for the specific emphasis in the pathologic differentiation that emerged.
Essential thrombocythemia, an exclusion diagnosis
At the current time, the diagnosis of ET is based essentially on exclusion, ie, other conditions giving rise to intense thrombocytosis must be ruled out before this diagnosis can be made. This is often difficult to achieve, and thus a positive test for distinguishing between ET and 2°T would be useful. The current findings could form the basis for development of such a test, provided that the reproducibility, sensitivity, and specificity of the PEG-rHu MGDF and TPO hypersensitivity of megakaryocytic progenitor cells seen during this work are confirmed in blind experiments with a larger series of patients having a variety of hematological conditions. This would greatly facilitate the diagnosis of ET.
Acknowledgments
We appreciate the patients' consenting to have their blood taken for these studies. We thank Drs D. Dotten and P. Anglin for providing some of the patients from their practices at St Michael's/Wellesley and Scarborough Centenary Hospitals, respectively. Our thanks go also to Mrs Maureen A. Anderson for arranging to make the patients available to us. The advice and assistance of Dr B. Fernandes during the study and in his critical reading of the manuscript are very much appreciated.
Portions of this work were presented at the American Society of Hematology Meeting, Miami Beach, FL, December 1998.
Supported by grant numbers MT 3969 and 14347 of the Medical Research Council of Canada.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 U.S.C. section 1734.
References
Author notes
Arthur A. Axelrad, Departments of Anatomy and Cell Biology and Medical Biophysics, Medical Sciences Bldg, #1 King's College Circle, Toronto, Ontario, Canada M5S 1A8; e-mail:a.axelrad@utoronto.ca.