Abstract
We analyzed the effect of aspirin, salicylate, and other nonsteroidal antiinflammatory drugs (NSAIDs) on the viability of B-chronic lymphocytic leukemia (B-CLL) cells. Aspirin induced a decrease in cell viability in a dose- and time-dependent manner. The mean IC50 for cells from 5 patients was 5.9 ± 1.13 mmol/L (range, 4.4 to 7.3 mmol/L). In some cases, 2.5 mmol/L aspirin produced an important cytotoxic effect after 4 days of incubation. No effect was observed with other NSAIDs, at concentrations that inhibit cyclooxygenase, such as ketorolac (10 μmol/mL), NS-398 (100 μmol/mL), or indomethacin (20 μmol/mL), thus suggesting the involvement of cyclooxygenase-independent mechanisms in aspirin-induced cytotoxicity. Salicylate also produced dose-dependent cytotoxic effects on B-CLL cells and the mean IC50 for cells from 5 patients was 6.96 ± 1.13 mmol/L (range, 5 to 7.8 mmol/L). Both aspirin and salicylate induced DNA fragmentation and the proteolytic cleavage of poly(ADP(adenosine 5′-diphosphate)-ribose) polymerase (PARP), demonstrating that both compounds induce apoptosis of B-CLL cells. Finally, inhibition of caspases by Z-VAD.fmk blocked proteolytic cleavage of PARP, DNA fragmentation, and cytotoxicity induced by aspirin. Mononuclear cells from normal donors showed a lower sensitivity than cells from B-CLL patients to aspirin as determined by analysis of cell viability. B and T lymphocytes from normal donors and T lymphocytes from CLL patients are more resistant to aspirin-induced apoptosis, as determined by analysis of phosphatidylserine exposure. These results indicate that aspirin and salicylate induce apoptosis of B-CLL cells by activation of caspases and that this activation involves cyclooxygenase-independent mechanisms.
© 1998 by The American Society of Hematology.
B-CELL CHRONIC lymphocytic leukemia (B-CLL) is characterized by the accumulation of monoclonal CD5+ B lymphocytes.1,2 Most circulating cells appear to be arrested at the G0 phase of the cell cycle and it has been suggested that the clonal excess of B cells results from decreased apoptosis rather than increased proliferation.3
Apoptosis is the physiological process whereby most cells, including B lymphocytes, are eliminated, which leads to homeostasis.4The evidence obtained in recent years indicates that many cancer chemotherapy agents induce apoptosis of tumor cells.5Glucocorticoids and other chemotherapeutic agents, such as chlorambucil, 2-chloro-2′-deoxyadenosine, fludarabine, camptothecin, and mitoxantrone,6-11 induce apoptosis in B-CLL lymphocytes, suggesting that apoptosis is one of the mechanisms of their therapeutic action.
Cysteine-proteases of the CED-3/ICE family, now named caspases, play an important role in apoptosis.12-14 Some of these proteases can cleave and inactivate poly(ADP-ribose) polymerase (PARP), and this cleavage can be used as a marker of activation of caspases and apoptosis. We recently reported that drug-induced apoptosis of B-CLL cells involves the activation of caspases.15
Nonsteroidal anti-inflammatory drugs (NSAIDs) have been shown to induce apoptosis of v-src–transformed chicken embryo fibroblasts and different human cell lines.16-20 The mechanism responsible for this apoptotic effect of NSAIDs is not clear. Aspirin and other NSAIDs directly target cyclooxygenase (COX),21 a key enzyme in the production of prostaglandins, prostacyclins, and thromboxanes.22 There are 2 COX isoenzymes, COX-1 and COX-2, which differ in their expression regulation and tissue distribution23 and could be selectively inhibited. Although COX is the only molecular target known of most NSAIDs, both COX-dependent and COX-independent mechanisms in the apoptotic action of NSAIDs have been reported.17,24 25
In the present study, we have analyzed the effect of aspirin, salicylate, and other NSAIDs on the viability of B-CLL cells and whether these drugs induce apoptosis of these leukemic cells.
MATERIALS AND METHODS
Patients.
Seven patients (1 man and 6 women) with B-CLL who had not received treatment, with a median age of 75 years (range, 63 to 78 years), were studied. B-CLL was diagnosed according to standard clinical and laboratory criteria. The median peripheral blood leukocytosis was 85 × 109 leukocytes/L (range, 34 to 194 × 109). Leukemic cells were phenotyped for cell surface markers by flow cytometry and were positive in all cases for CD5 and CD19. According to Binet’s classification,26 at the time of inclusion, 5 patients were at stage A, 1 patient was at stage B, and 1 patient was at stage C.
Isolation of B-CLL cells.
Mononuclear cells from peripheral blood samples were isolated by centrifugation on a Ficoll/Hypaque (Seromed, Berlin, Germany) gradient and cryopreserved in liquid nitrogen in the presence of 10% dimethyl sulfoxide (DMSO).
Reagents.
Aspirin (acetylsalicylic acid), sodium salicylate, indomethacin, 3,(4,5-Dimethylthiazol-2-yl)2,5-diphenyltetrazolium bromide (MTT), and propidium iodide (PI) were obtained from Sigma Chemicals Co (St Louis, MO). N-benzyloxycarbonyl-Val-Ala-Asp-fluromethyl ketone (Z-VAD.fmk) was obtained from Enzyme Systems Products (Dublin, CA). Ketorolac was kindly provided by Almirall Laboratories (Barcelona, Spain). N-(2-ciclohexyloxy-4-nitrophenyl)-methanesulfonamide (NS-398) was obtained from BIOMOL Research Laboratories, Inc (Plymouth Meeting, PA). Stock solutions of aspirin and indomethacin in absolute ethanol, NS-398 in DMSO, and sodium salicylate and ketorolac in double-distilled water were freshly prepared for each experiment. The final concentrations of ethanol and DMSO did not affect cell viability (data not shown). Anti-PARP polyclonal antibody (Vi.5) raised against the recombinant human PARP overproduced in Sf9/baculovirus was kindly provided by Dr Gilbert de Murcia (Strasbourg, France).
Cell culture.
B-CLL lymphocytes were cultured immediately after the thawing of the cells at a concentration of 5 × 106 cells/mL in RPMI 1640 culture medium (GIBCO-BRL, Paisley, UK) supplemented with 10% heat-inactivated fetal calf serum (FCS), 2 mmol/L glutamine, and 0.04 mg/mL gentamicin at 37°C in a humidified atmosphere containing 5% carbon dioxide.27 Factors were added at the beginning of the culture.
Cell viability assay.
Cell viability was determined by the MTT assay.28 B lymphocytes (5 × 105 cells/well) were incubated in 96-well plates in the absence or in the presence of factors in a final volume of 100 μL. After 48 hours, 10 μL of MTT (5 mg/mL in phosphate-buffered saline) was added to each well for an additional 6 hours. The blue MTT formazan precipitated was dissolved in 100 μL of isopropanol:1 mol/L HCl (24:1) and the absorbance values at 550 nm were determined on a multiwell plate reader.
Western blot analysis of PARP cleavage.
Cells were lysed with Laemmli sample buffer29 and protein extracts were electrophoresed on 10% polyacrylamide gel and transferred to Immobilon-P (Millipore, Bedford, MA) membranes. After blocking for 1 hour with 5% dried skimmed milk in TBST (50 mmol/L Tris HCl, pH 8.0, 150 mmol/L NaCl, 0.5% Tween-20), the filters were incubated with Vi.5 PARP antibody diluted 1:1,000 in 5% dried skimmed milk in TBST, which recognizes both the native enzyme (116 kD) and the cleavage product (∼85 kD).15 Antibody binding was detected using a secondary antibody (swine antirabbit Ig; DAKO, Glostrup, Denmark) conjugated to horseradish peroxidase diluted 1:500 in 5% dried skimmed milk in TBST and an enhanced chemiluminescence (ECL) detection kit (Amersham, Buckinghamshire, UK). As an internal control of total cellular protein levels, blots were also probed with α-tubulin antibody (Oncogene Science, Inc, Cambridge, MA).
Analysis of DNA fragmentation.
Analysis of DNA fragmentation by agarose gel electrophoresis was performed as previously described.15 Five million cells were pelleted and lysed for 20 minutes at 4°C in 10 mmol/L Tris-HCl, pH 7.4, 0.2% Triton X-100, 1 mmol/L EDTA. After centrifugation at 14,000g for 15 minutes, the supernatant was treated with 0.2 mg/mL of proteinase K in a buffer containing 150 mmol/L NaCl, 10 mmol/L Tris HCl, pH 8.0, 40 mmol/L EDTA, and 1% sodium dodecyl sulfate (SDS) for 6 hours at 37°C. DNA was extracted twice with buffered phenol and precipitated with 140 mmol/L NaCl and 2 vol of ethanol at −20°C overnight. DNA precipitates were washed twice in 70% ethanol, dissolved in double-distilled water, and treated for 1 hour at 37°C with RNase (Boehringer Mannheim, Mannheim, Germany). Finally, DNA preparations were electrophoresed in 1% agarose gels. Gels were stained with ethidium bromide and visualized under UV light.
PI DNA staining.
Quantification of apoptosis by PI staining and fluorescence-activated cell sorting (FACS) analysis was performed as described previously.30 Briefly, cells were harvested and fixed in 70% ethanol. Cells were centrifuged, washed in phosphate-buffered saline (PBS), and resuspended in 0.5 mL PBS containing PI (5 μg/mL) and RNase (100 μg/mL). Tubes were incubated for 30 minutes at 37°C and placed at 4°C in the dark overnight before flow cytometry analysis to identify the sub-G0 peak corresponding to apoptosis.
Analysis of apoptosis by annexin binding.
Exposure of phosphatidylserine was quantified by surface annexin V staining as described previously.31 One million cells were incubated during 24 hours with the indicated factors. After that time, cells were washed with PBS and incubated with phycoerythrin-conjugated anti-CD19 (DAKO) and tri-color–conjugated anti-CD2 (Caltag Laboratories, Burlingame, CA) for 15 minutes in the dark. Cells were then washed, resuspended in 200 μL of binding buffer (10 mmol/L HEPES, pH 7.4 , 2.5 mmol/L CaCl2, 140 mmol/L NaCl), and incubated with 0.5 μg/mL of ApoAlert Enhanced Annexin V-FITC (Clontech Laboratories Inc, Palo Alto, CA) for 5 to 15 minutes in the dark, before analysis by flow cytometry (FACScan; Becton Dickinson, Mountain View, CA). Samples were acquired using Lysis-II software and data were analyzed with the Paint-a-gate Pro software (Becton Dickinson). To analyze a sufficient number of cells, a live-gate in side scatter (SSC) versus CD19 or SSC versus CD2 was drawn and at least 5,000 CD19+ cells or CD2+ cells were acquired.
Statistical analysis.
Levels of significance between samples were determined using thet-test for nonpaired samples and the analysis of variance (ANOVA), Fisher’s protected least significant difference (PLSD). To compare the effect of aspirin on normal PBMCs and B-CLL cells, we performed a multiple analysis of variance (MANOVA), Hottelling T2.
RESULTS
Cytotoxic effect of aspirin on B-CLL lymphocytes.
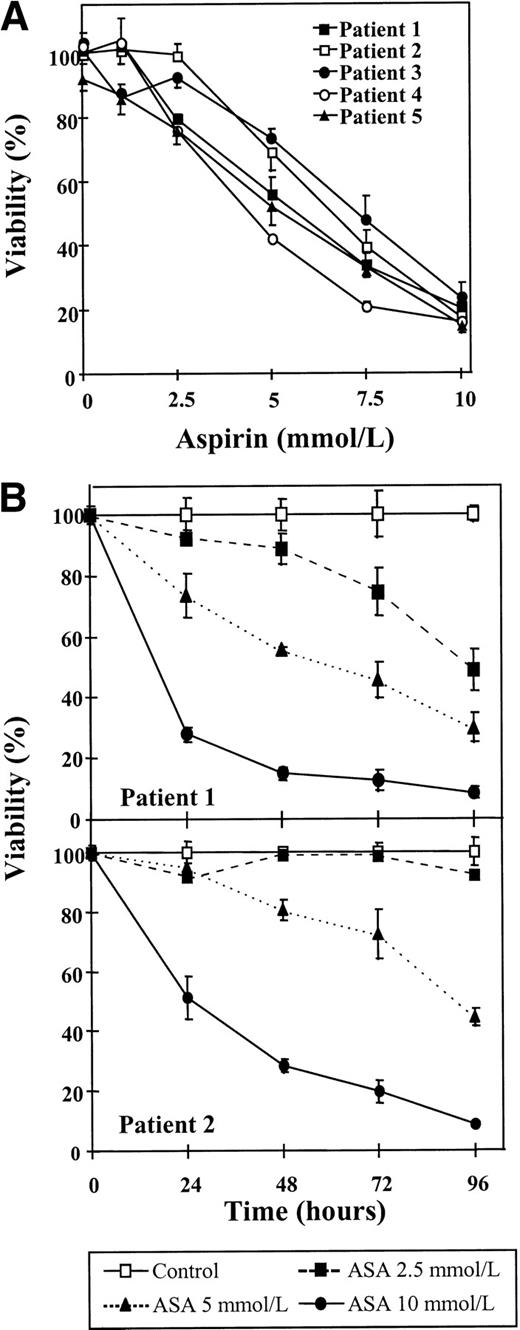
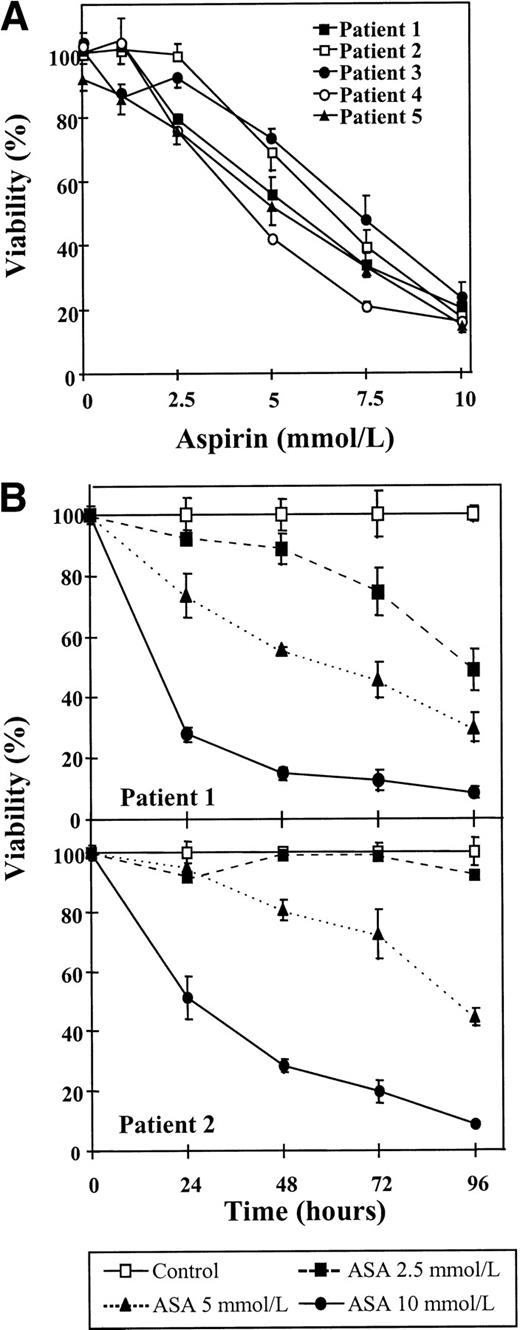
First, the effect of aspirin on the viability of B-CLL lymphocytes was studied. Cells from 5 patients were incubated for 48 hours with several doses of aspirin, ranging from 1 to 10 mmol/L, and cell viability was determined by the MTT assay. As seen in Fig 1A, a dose-dependent decrease in cell viability could be observed in all the cases studied, although cell sensitivity to aspirin differed from one patient to another. The mean IC50 of these dose-response studies was 5.9 ± 1.13 mmol/L (range, 4.4 to 7.3 mmol/L). In 3 patients (patients no. 1, 4, and 5), an effect was detected with 2.5 mmol/L aspirin.
Cytotoxic effect of aspirin on B-CLL cells. (A) Dose-response of the cytotoxic effect of aspirin on B-CLL cells. Cells from 5 patients were incubated for 48 hours with various concentrations of aspirin that ranged from 1 to 10 mmol/L. (B) Time course of aspirin-induced cytotoxicity. Cells from patients no. 1 and 2 were incubated with 2.5, 5, and 10 mmol/L aspirin (ASA) for the times indicated. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments.
Cytotoxic effect of aspirin on B-CLL cells. (A) Dose-response of the cytotoxic effect of aspirin on B-CLL cells. Cells from 5 patients were incubated for 48 hours with various concentrations of aspirin that ranged from 1 to 10 mmol/L. (B) Time course of aspirin-induced cytotoxicity. Cells from patients no. 1 and 2 were incubated with 2.5, 5, and 10 mmol/L aspirin (ASA) for the times indicated. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments.
The cytotoxicity of aspirin on B-CLL lymphocytes was further studied with time-course assays that were performed with cells from patients no. 1 and 2. Cells were incubated without or with aspirin (2.5, 5, or 10 mmol/L) for different periods of time, as indicated in Fig 1B. A significant decrease in cell viability was observed in both patients after only 24 hours of incubation with 10 mmol/L aspirin (P < .003). A dose of 5 mmol/L aspirin produced a significant decrease in cell viability of lymphocytes from patient no. 1 after 24 hours of incubation (P = .006) that increased with time of incubation. No significant effect was seen with this dose in cells from patient no. 2 after 24 hours (P = .29), and longer periods of incubation were needed to achieve an important cytotoxic effect. Incubation for 48 hours with 2.5 mmol/L aspirin induced cytotoxicity in cells from patient no. 1, but the viability of cells from patient no. 2 was not significantly decreased after 96 hours of incubation.
COX inhibition is not sufficient to induce apoptosis of B-CLL cells.
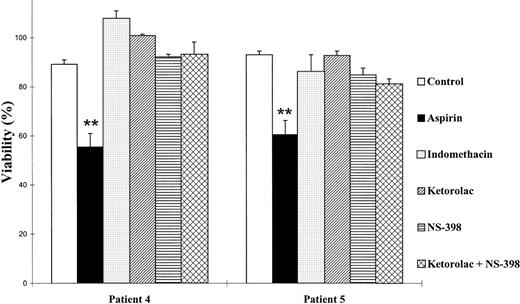
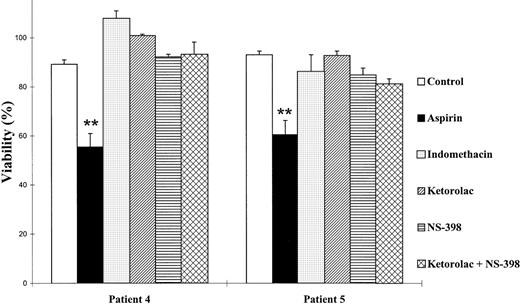
Because aspirin targets the enzyme COX, we tested whether other inhibitors of this enzyme could produce cytotoxic effects on B-CLL lymphocytes. An inhibitor of both COX-1 and COX-2 (indomethacin), a specific inhibitor of COX-1 (ketorolac), and a specific inhibitor of COX-2 (NS-398) were tested. Neither COX-1 nor COX-2 inhibitors produced any effect on cell viability with doses up to 10 μmol/L ketorolac, 20 μmol/L indomethacin, and 100 μmol/L NS-398, which have been previously described to inhibit COX32-36(Fig 2). Furthermore, we used ketorolac and NS-398 together, and no effect was observed either. All these assays were performed with cells from 5 patients and in all cases there were no effects. However, at the concentrations used in these experiments, these NSAIDs inhibited the production of prostaglandin E2(PGE2) by B-CLL cells, thus demonstrating that COX was being inhibited (data not shown). Taken together, these results indicate that inhibition of COX is not sufficient to induce the decrease in cell viability produced by aspirin on B-CLL lymphocytes.
Effect of COX inhibitors on the viability of B-CLL lymphocytes. Cells were incubated for 48 hours with 5 mmol/L aspirin, 10 μmol/L ketorolac, 100 μmol/L NS-398, or 20 μmol/L indomethacin. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments. Statistical significance of differences between control cells and cells treated with COX inhibitors was assayed by ANOVA, Fisher’s PLSD. **P < .0001.
Effect of COX inhibitors on the viability of B-CLL lymphocytes. Cells were incubated for 48 hours with 5 mmol/L aspirin, 10 μmol/L ketorolac, 100 μmol/L NS-398, or 20 μmol/L indomethacin. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments. Statistical significance of differences between control cells and cells treated with COX inhibitors was assayed by ANOVA, Fisher’s PLSD. **P < .0001.
Cytotoxic effect of salicylate on B-CLL lymphocytes.
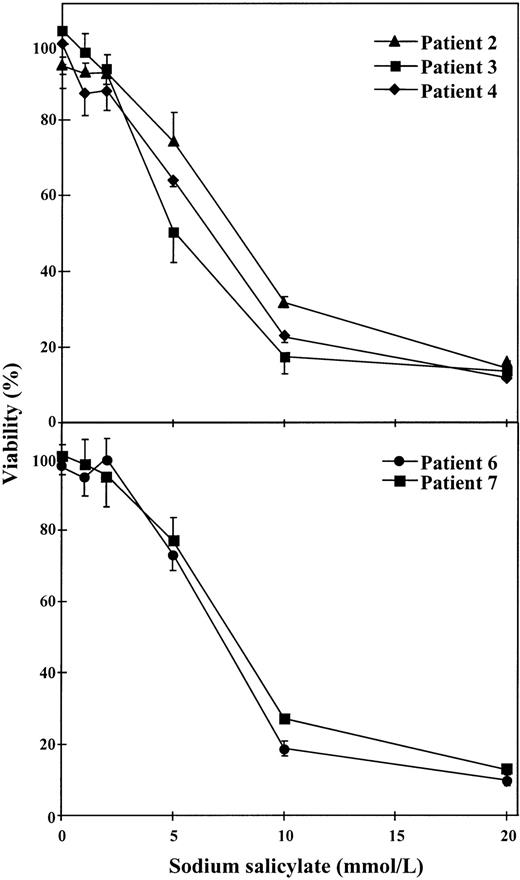
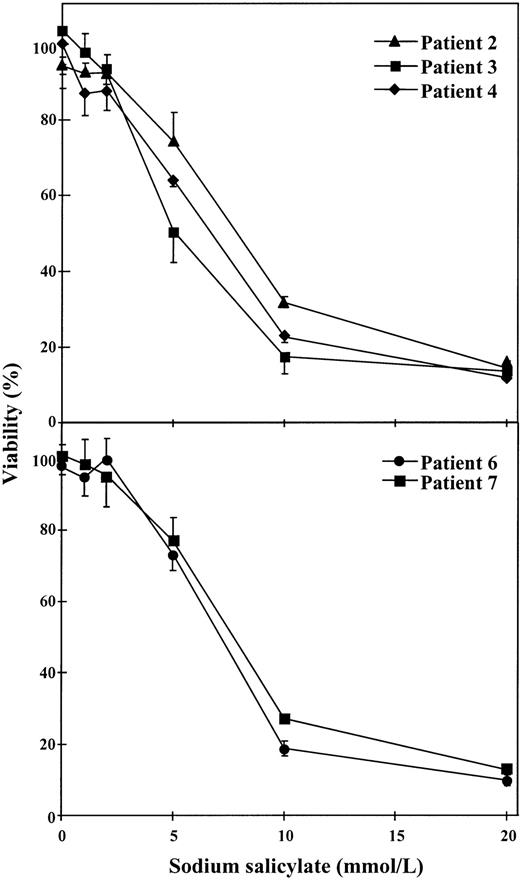
To test if salicylate, a structure-related compound of aspirin, produced any effect on cell viability, dose-response studies were performed with 1 to 20 mmol/L sodium salicylate. As seen in Fig 3, this substance produced cytotoxic effects on B-CLL cells when concentrations of 5 to 10 mmol/L were used. This effect was observed in cells from 5 patients studied. The mean IC50 for these 5 patients was 6.96 ± 1.13 mmol/L (range, 5 to 7.8 mmol/L).
Dose-response of the cytotoxic effect of salicylate on B-CLL cells. Cells from 5 patients were incubated for 48 hours with various concentrations of sodium salicylate as indicated. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments.
Dose-response of the cytotoxic effect of salicylate on B-CLL cells. Cells from 5 patients were incubated for 48 hours with various concentrations of sodium salicylate as indicated. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments.
Aspirin and salicylate induce apoptosis and activation of caspases in B-CLL lymphocytes.
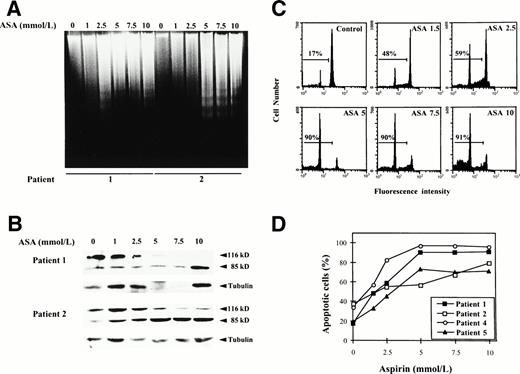
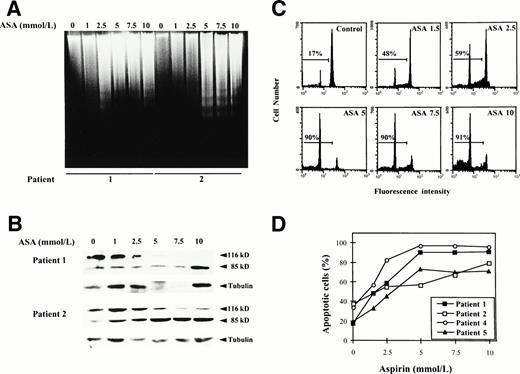
To determine whether this cytotoxic effect was due to induction of apoptosis, we analyzed whether incubation with aspirin or salicylate induced DNA fragmentation and proteolytic cleavage of PARP, a hallmark of apoptosis in most cells, including B-CLL cells.15 Cells from patients no. 1 and 2 were incubated with various doses of aspirin as described previously, ranging from 1 to 10 mmol/L. As seen in Fig 4, both DNA fragmentation (Fig 4A) and PARP cleavage (Fig 4B) were dose-dependent and a clear effect could be observed with 2.5 to 5 mmol/L aspirin. These doses corresponded to the doses that induced a cytotoxic effect as previously determined by the MTT assay. Similar results were obtained with cells from patients no. 3 and 4. The apparent decrease in the total amount of PARP in the Western blot analysis of patient no. 1 at the doses of 5 and 7.5 mmol/L is due to a decrease in the total amount of loaded protein, as demonstrated by immunoblotting of α-tubulin.
Induction of apoptosis by aspirin on B-CLL cells. (A) Effect of aspirin on DNA fragmentation in B-CLL cells. Cells from patients no. 1 and 2 were incubated for 24 hours with 1 to 10 mmol/L aspirin (ASA). DNA was extracted and subjected to agarose gel electrophoresis as described in the Materials and Methods. (B) Effect of aspirin on PARP cleavage. Cells from patients no. 1 and 2 were incubated for 48 hours with 1 to 10 mmol/L aspirin (ASA). PARP cleavage was analyzed on protein extracts from these cells by Western blot as described in the Materials and Methods. The position of the native PARP (116 kD) and the proteolytic fragment (85 kD) is indicated. As an internal control, blots were also probed with -tubulin antibody. (C) Effect of aspirin on DNA content in a representative CLL patient. Cells from patient no. 1 were incubated for 48 hours with 1 to 10 mmol/L aspirin (ASA). DNA content was quantified by PI staining and flow cytometry analysis as described in the Materials and Methods. (D) Quantification of aspirin-induced apoptosis in cells from 4 patients by PI staining and FACS analysis after 48 hours of incubation with 1 to 10 mmol/L aspirin.
Induction of apoptosis by aspirin on B-CLL cells. (A) Effect of aspirin on DNA fragmentation in B-CLL cells. Cells from patients no. 1 and 2 were incubated for 24 hours with 1 to 10 mmol/L aspirin (ASA). DNA was extracted and subjected to agarose gel electrophoresis as described in the Materials and Methods. (B) Effect of aspirin on PARP cleavage. Cells from patients no. 1 and 2 were incubated for 48 hours with 1 to 10 mmol/L aspirin (ASA). PARP cleavage was analyzed on protein extracts from these cells by Western blot as described in the Materials and Methods. The position of the native PARP (116 kD) and the proteolytic fragment (85 kD) is indicated. As an internal control, blots were also probed with -tubulin antibody. (C) Effect of aspirin on DNA content in a representative CLL patient. Cells from patient no. 1 were incubated for 48 hours with 1 to 10 mmol/L aspirin (ASA). DNA content was quantified by PI staining and flow cytometry analysis as described in the Materials and Methods. (D) Quantification of aspirin-induced apoptosis in cells from 4 patients by PI staining and FACS analysis after 48 hours of incubation with 1 to 10 mmol/L aspirin.
Furthermore, we corroborated these results measuring apoptosis by PI staining and FACS analysis. As shown in Fig 4C and D, aspirin induced in a dose-dependent manner an increase in the percentage of subdiploid cells in 4 patients studied.
As seen in Fig 5, salicylate also induced DNA fragmentation and cleavage of PARP when cells were incubated with the doses that produced cytotoxic effects. Similar results to that shown in Fig 5 were obtained with cells from 2 additional patients (results not shown). These results demonstrate that the cytotoxicity of both aspirin and salicylate is due to apoptosis induction.
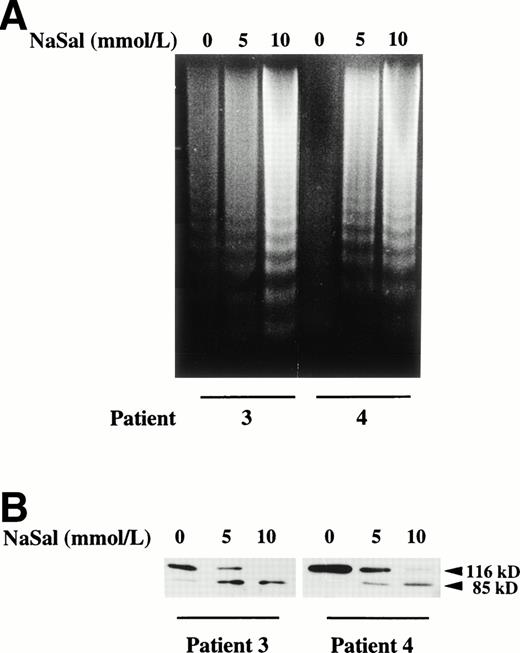
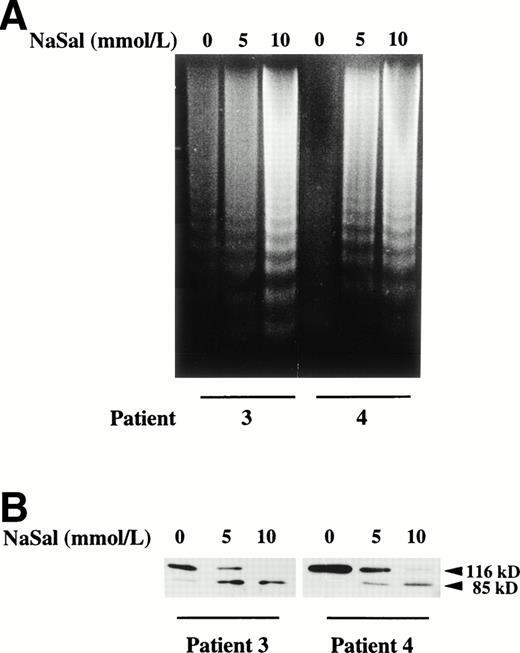
Induction of apoptosis by salicylate on B-CLL cells. (A) Effect of salicylate on DNA fragmentation in B-CLL cells. Cells from patients no. 3 and 4 were incubated for 24 hours with 5 and 10 mmol/L sodium salicylate (NaSal). DNA was extracted and subjected to agarose gel electrophoresis as described in the Materials and Methods. (B) Effect of salicylate on PARP cleavage. Cells from patients no. 3 and 4 were incubated for 48 hours with 5 and 10 mmol/L sodium salicylate (NaSal). PARP cleavage was analyzed on protein extracts from these cells by Western blot as described in the Materials and Methods. The position of the native PARP (116 kD) and the proteolytic fragment (85 kD) is indicated.
Induction of apoptosis by salicylate on B-CLL cells. (A) Effect of salicylate on DNA fragmentation in B-CLL cells. Cells from patients no. 3 and 4 were incubated for 24 hours with 5 and 10 mmol/L sodium salicylate (NaSal). DNA was extracted and subjected to agarose gel electrophoresis as described in the Materials and Methods. (B) Effect of salicylate on PARP cleavage. Cells from patients no. 3 and 4 were incubated for 48 hours with 5 and 10 mmol/L sodium salicylate (NaSal). PARP cleavage was analyzed on protein extracts from these cells by Western blot as described in the Materials and Methods. The position of the native PARP (116 kD) and the proteolytic fragment (85 kD) is indicated.
To demonstrate the involvement of activation of caspases in the apoptotic effect of aspirin, we studied whether the caspase inhibitor Z-VAD.fmk prevented aspirin-induced apoptosis. We incubated B-CLL cells with aspirin in the presence or absence of 200 μmol/L Z-VAD.fmk, a concentration that inhibits apoptosis of B-CLL cells.15This concentration of Z-VAD.fmk had no cytotoxic effects (results not shown) and prevented the cytotoxic effect of both 5 and 10 mmol/L aspirin in the 3 cases studied, 2 of which are shown in Fig 6. Furthermore, Z-VAD.fmk completely blocked aspirin-induced DNA fragmentation (Fig 7A) and PARP cleavage (Fig 7B).
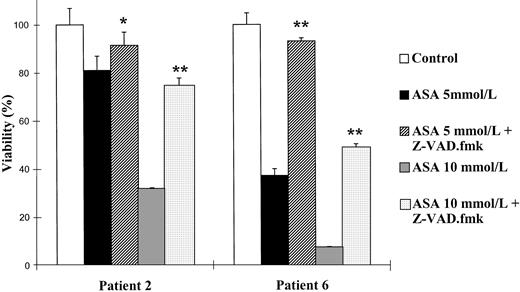
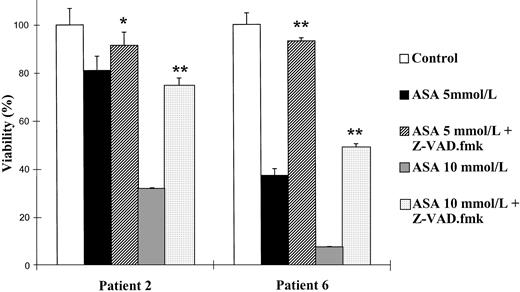
Effect of the caspase inhibitor Z-VAD.fmk on the viability of B-CLL cells. Cells from patients no. 2 and 6 were seeded in 96-microwell plates and incubated for 48 hours either alone or in the presence of aspirin (ASA; 5 or 10 mmol/L) or aspirin with 200 μmol/L Z-VAD.fmk. Z-VAD.fmk was added 1 hour before aspirin administration. Cytotoxicity was analyzed by the MTT method as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments. Statistical significance of differences between treatment with aspirin alone or in combination with Z-VAD.fmk was assayed ANOVA (Fisher’s PLSD). *P < .03; **P < .0001.
Effect of the caspase inhibitor Z-VAD.fmk on the viability of B-CLL cells. Cells from patients no. 2 and 6 were seeded in 96-microwell plates and incubated for 48 hours either alone or in the presence of aspirin (ASA; 5 or 10 mmol/L) or aspirin with 200 μmol/L Z-VAD.fmk. Z-VAD.fmk was added 1 hour before aspirin administration. Cytotoxicity was analyzed by the MTT method as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. Data are shown as the mean value ± SD of 3 independent experiments. Statistical significance of differences between treatment with aspirin alone or in combination with Z-VAD.fmk was assayed ANOVA (Fisher’s PLSD). *P < .03; **P < .0001.
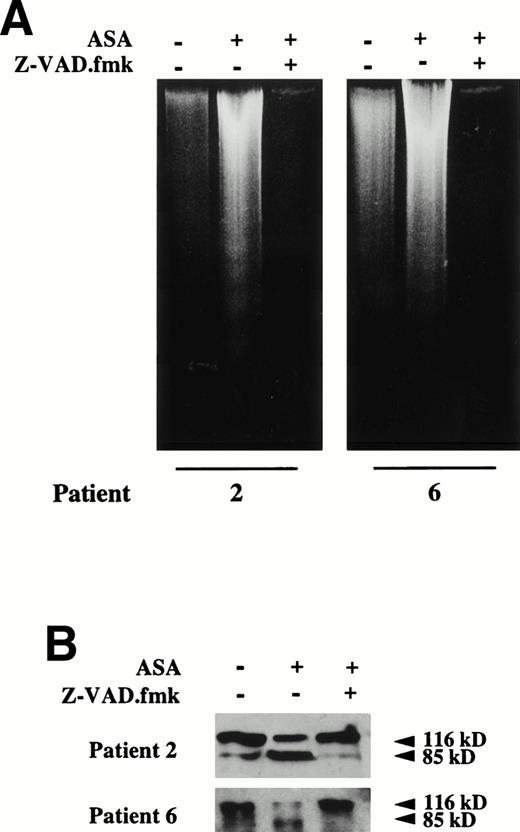
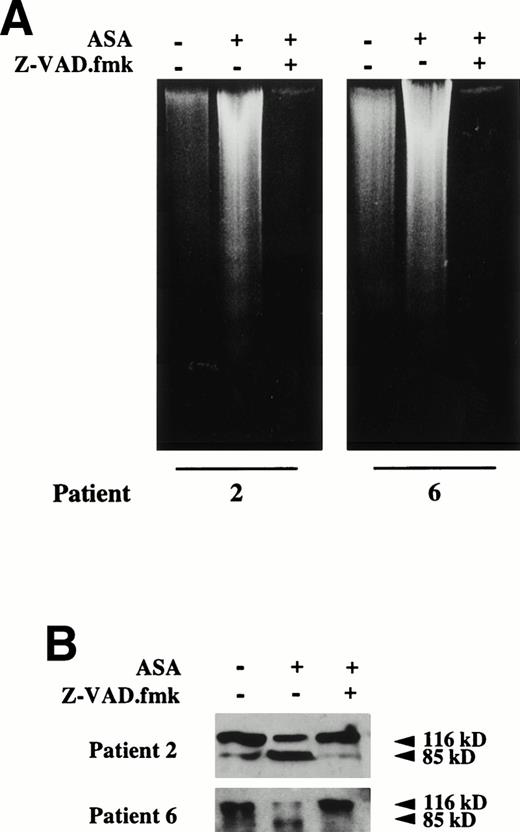
Effect of the caspase inhibitor Z-VAD.fmk on aspirin-induced apoptosis in B-CLL cells. (A) Effect of Z-VAD.fmk on aspirin-induced DNA fragmentation. B lymphocytes from 2 B-CLL patients were incubated for 24 hours with 10 mmol/L aspirin (ASA) in the presence or absence of 200 μmol/L Z-VAD.fmk. DNA was extracted and subjected to agarose gel electrophoresis as described in the Materials and Methods. (B) Effect of Z-VAD.fmk on aspirin-induced PARP cleavage. Cells from patients no. 2 and 6 were incubated for 24 hours with 10 mmol/L aspirin (ASA) in the presence or absence of 200 μmol/L Z-VAD.fmk. Cells were lysed and analyzed by Western blot as described in the Materials and Methods. Z-VAD.fmk was added 1 hour before aspirin administration.
Effect of the caspase inhibitor Z-VAD.fmk on aspirin-induced apoptosis in B-CLL cells. (A) Effect of Z-VAD.fmk on aspirin-induced DNA fragmentation. B lymphocytes from 2 B-CLL patients were incubated for 24 hours with 10 mmol/L aspirin (ASA) in the presence or absence of 200 μmol/L Z-VAD.fmk. DNA was extracted and subjected to agarose gel electrophoresis as described in the Materials and Methods. (B) Effect of Z-VAD.fmk on aspirin-induced PARP cleavage. Cells from patients no. 2 and 6 were incubated for 24 hours with 10 mmol/L aspirin (ASA) in the presence or absence of 200 μmol/L Z-VAD.fmk. Cells were lysed and analyzed by Western blot as described in the Materials and Methods. Z-VAD.fmk was added 1 hour before aspirin administration.
Aspirin effect on normal peripheral blood mononuclear cells (PBMCs).
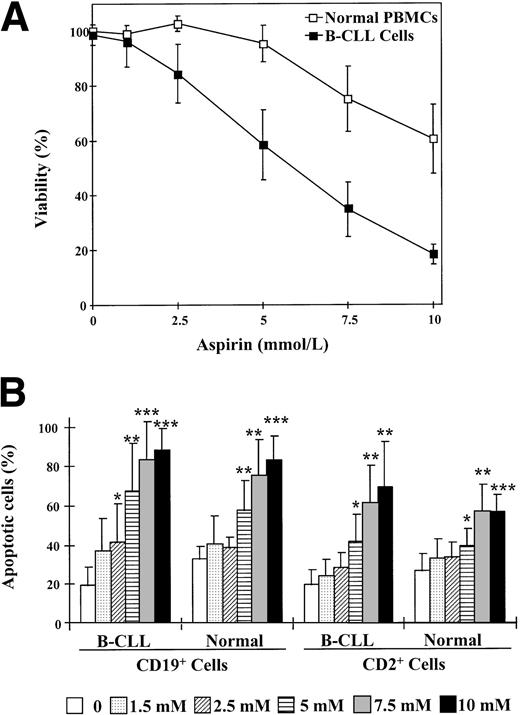
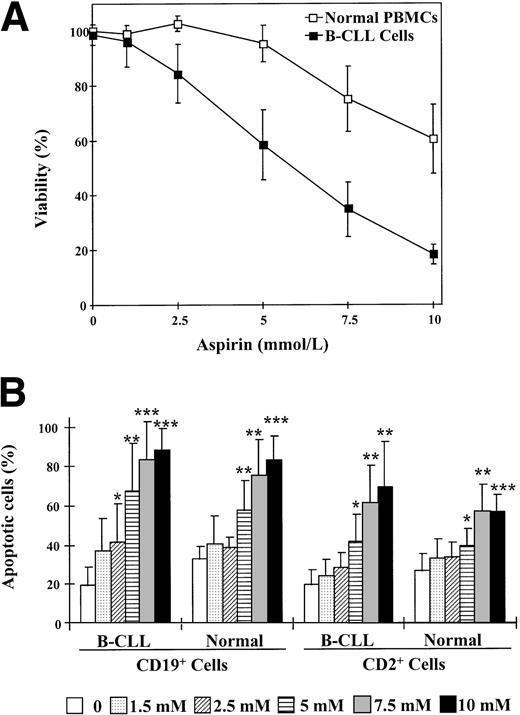
Finally, we studied the cytotoxic effects of aspirin in normal PBMCs. Incubation of PBMCs with aspirin produced no significant cytotoxic effect (P > .12) with doses up to 7.5 mmol/L (Fig 8A). The mean aspirin IC50value for normal cells was 15.8 ± 6.2 mmol/L (range, 9.8 to 26.3 mmol/L). The comparison between the dose-responses of the cytotoxic effect of aspirin on B-CLL cells and normal PBMCs indicates that normal PBMCs are more resistant to aspirin (P = .003). A similar dose-dependency was found when DNA fragmentation and PARP cleavage were analyzed. The concentration of aspirin or salicylate necessary to induce detectable DNA fragmentation and PARP cleavage was 7.5 mmol/L (results not shown).
Effect of aspirin on normal PBMCs and B cells. (A) Dose-response of the cytotoxic effect of aspirin on normal PBMCs. Cells from 6 normal donors were incubated with various concentrations of aspirin as indicated for 48 hours. Data are presented as the means ± SD of 3 experiments each for all 6 normal donors and compared with the means of the 5 B-CLL patients represented in Fig 1A. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. (B) Comparison between the induction of apoptosis in B cells and T cells from B-CLL patients and normal donors. Cells were incubated with 1 to 10 mmol/L aspirin for 24 hours and phosphatidylserine exposure was measured by binding of annexin V to CD19+ or CD2+ cells as described in the Materials and Methods. Statistical significance was determined using the t-test for nonpaired samples. *P < .05; **P < .01; ***P < .001.
Effect of aspirin on normal PBMCs and B cells. (A) Dose-response of the cytotoxic effect of aspirin on normal PBMCs. Cells from 6 normal donors were incubated with various concentrations of aspirin as indicated for 48 hours. Data are presented as the means ± SD of 3 experiments each for all 6 normal donors and compared with the means of the 5 B-CLL patients represented in Fig 1A. Cell viability was determined by the MTT assay as described in the Materials and Methods and is expressed as a percentage of the viability of control cells at the beginning of the culture. (B) Comparison between the induction of apoptosis in B cells and T cells from B-CLL patients and normal donors. Cells were incubated with 1 to 10 mmol/L aspirin for 24 hours and phosphatidylserine exposure was measured by binding of annexin V to CD19+ or CD2+ cells as described in the Materials and Methods. Statistical significance was determined using the t-test for nonpaired samples. *P < .05; **P < .01; ***P < .001.
Because most normal PBMCs are T lymphocytes, it was interesting to distinguish the effects of aspirin on B and T cells. To specifically analyze the induction of apoptosis in normal B cells by aspirin, we quantified phosphatidylserine exposure in response to aspirin in CD19+ cells. As seen in Fig 8B, aspirin did not induce a significant increase in the apoptosis of normal B cells with doses up to 2.5 mmol/L (P > .13); in contrast, aspirin induced apoptosis of B-CLL cells at 2.5 mmol/L, indicating that B cells from normal donors have a lower degree of sensitivity to aspirin than that of B-CLL cells. Furthermore, T cells from both normal donors and B-CLL patients appeared to be more resistant to aspirin-induced apoptosis than B-CLL cells (Fig 8B).
DISCUSSION
The results shown in this report demonstrate that aspirin and its metabolite salicylate induce apoptosis of B-CLL cells. This is the first report showing apoptotic action of NSAIDs in human primary tumor cells.
NSAIDs have been shown to induce apoptosis of human colon carcinoma and in vitro transformed colon adenoma cell lines.17-20Furthermore, different NSAIDs cause apoptosis in v-src–transformed chicken embryo fibroblasts16 and sulindac sulfide induces apoptosis of promyelocytic leukemia cell line HL-60.18 It has been reported that aspirin, in contrast to other NSAIDs, does not induce apoptosis in either HT-29 colon carcinoma cells or v-src–transformed chicken fibroblasts.16,20 The discrepancy between these results and our results could be explained by the fact that the doses used in previous studies were too low (150 μmol/L and 1.5 mmol/L). Very recently, it has been reported that aspirin induces apoptosis of HT-29 cells at concentrations higher that 1 mmol/L.37
The mechanisms by which NSAIDs induce apoptosis are not clear. In agreement with the hypothesis that the apoptotic action of NSAIDs is mediated by the inhibition of COX, overexpression of COX-2 in rat epithelial intestinal cells inhibits butyrate-induced apoptosis and this inhibition was reversed by the NSAID sulindac sulfide.24 However, the sulfone metabolite of sulindac, which does not inhibit cyclooxygenases,38 also induces apoptosis of HT-29 cells,17 and treatment with different prostaglandins failed to reverse the effects of NSAIDs on apoptosis.25 Considering the results herein reported, two different arguments indicate the involvement of cyclooxygenase-independent mechanisms in the apoptotic action of aspirin and salicylate on B-CLL cells. (1) Neither COX-1 nor COX-2 inhibitors induced cytotoxicity in B-CLL cells. (2) Although aspirin is a much more potent inhibitor of COX than salicylate, both drugs show similar potency as inducers of apoptosis. Taken together, these results suggest the existence of COX-dependent and COX-independent mechanisms in the apoptotic action of NSAIDs. Interestingly, we recently demonstrated that aspirin inhibits DNA synthesis in Swiss 3T3 fibroblasts by both COX-dependent and COX-independent mechanisms, depending on the dose used.39
COX-independent mechanisms have been proposed to explain the inhibition of the transcription factor NF-κB by aspirin and salicylate.40-43 It is noteworthy that inhibition of NF-κB causes apoptosis of B lymphocytes44,45 and that NF-κB has an essential role in preventing tumor necrosis factor-α (TNF-α)– and cancer therapy-induced apoptosis.46-49Another COX-independent mechanism induced by aspirin and salicylate is the inhibition of AP-1 activity, which may involve the elevation of H+ concentration.50 In addition, it was very recently reported that salicylate induces apoptosis of fibroblasts via p38 mitogen-activated protein kinase,51 and this kinase has been involved in mIgM-induced apoptosis of human B lymphocytes.52 The results presented in this report demonstrate that aspirin-induced apoptosis of B-CLL cells involves the activation of caspases, as previously described for glucocorticoid-, fludarabine-, and mitoxantrone-induced apoptosis.11 15 The elucidation of the mechanisms involved in the apoptotic action of aspirin and salicylate in B-CLL cells needs further investigation.
The finding that salicylate also induces apoptosis of B-CLL cells is important, because aspirin is substantially hydrolyzed to salicylate on first pass through portal circulation. Consequently, plasma concentrations of salicylate rapidly exceed plasma concentrations of aspirin after oral administration of aspirin, and the half-life of salicylate in the plasma is considerably longer than that of aspirin.53 Although we only observe effects of aspirin and salicylate on apoptosis of B-CLL cells at relatively high concentrations (1.5 to 10 mmol/L), the lower range of such concentrations is achieved and even maintained in the treatment of chronic inflammatory states such as rheumatoid arthritis, in which the therapeutic plasma concentration is 150 to 300 mg/L (0.94 to 1.88 mmol/L).54-57 Unfortunately, not all patients are able to tolerate the adequate dosage to maintain these plasma levels for a long time.57 On the other hand, normal peripheral blood mononuclear cells are more resistant to aspirin than B-CLL cells, thus suggesting that clinical use of aspirin at concentrations that induce apoptosis of B-CLL cells could have only minor effects on normal lymphocytes in certain patients.
Several epidemiological studies have demonstrated an association between the long-term consumption of aspirin and a reduced risk of colon cancer.58-61 Although no association with reduced risk of other types of cancer has been clearly demonstrated,61 the incidence of B-CLL in cohorts of individuals taking aspirin on a regular basis has not been analyzed. This issue, as well as whether there is a potential role for aspirin in B-CLL management, deserves investigation. Finally, the elucidation of the mechanisms underlying the induction of the apoptosis of B-CLL cells by aspirin and salicylate would provide important information to understand the biology of B-CLL and to design new strategies for its therapy.
ACKNOWLEDGMENT
The authors are indebted to Dr G. de Murcia for the kind gift of PARP antibodies. We thank Drs V. Jiménez and J. Ros for the determinations of PGE2 levels. We thank Drs R. Bartrons, C. Pastor, and P. Feliz for their help and suggestions and R. Rycroft for language assistance.
B.B., M.P., M.B., and E.C. are recipients of a research fellowship from Fundación de la Asociación Española Contra el Cáncer, CIRIT, Fundació August Pi i Sunyer, and Universitat de Barcelona, respectively. This work was supported by “Fondo de Investigaciones Sanitarias de la Seguridad Social” (FIS 95/0873), by Quı́mica Farmacéutica Bayer S.A. (Division Consumer Care), and by the “Generalitat de Catalunya” (1995 SGR 00427).
Address reprint requests to Joan Gil, PhD, Departament de Ciències Fisiològiques II, Campus de Bellvitge, Universitat de Barcelona, Pavelló de Govern, 4a planta, 08907 L’Hospitalet, Spain; e-mail: joangil@bellvitge.bvg.ub.es.
The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked "advertisement" is accordance with 18 U.S.C. section 1734 solely to indicate this fact.