Key Points
This study confirms the efficacy of single-agent modakafusp alfa for patients with RRMM at both recommended phase 2 doses.
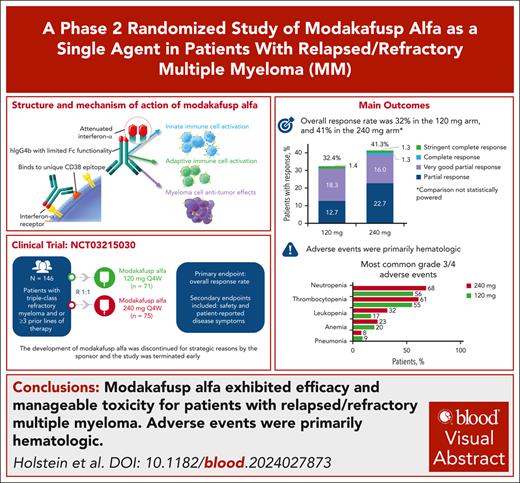
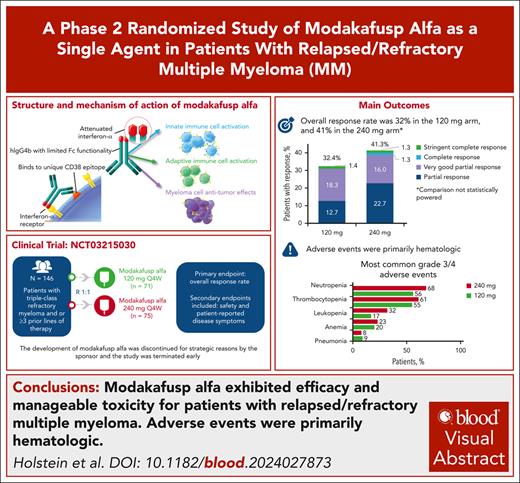
ORRs were 32% and 41% at 120 mg and 240 mg every 4 weeks, respectively, with adverse events numerically higher at 240 mg.
Visual Abstract
Modakafusp alfa is a first-in-class immunocytokine-directing interferon alfa to CD38+ cells. Our previous phase 1/2 trial identified 2 potential phase 2 doses of modakafusp alfa for patients with relapsed/refractory multiple myeloma (RRMM): 1.5 or 3 mg/kg every 4 weeks. The overall response rate (ORR) among 30 patients treated at 1.5 mg/kg was 43%. This phase 2 dose optimization study randomized 147 patients with triple-class refractory disease and ≥3 previous lines of therapy 1:1 to modakafusp alfa 120 mg (n = 71) or 240 mg (n = 75) every 4 weeks (fixed-dose equivalents of 1.5 and 3 mg/kg every 4 weeks). Patients had received a median of 6 previous lines of therapy; 66% were penta-exposed and 45% had previously been exposed to anti–B-cell maturation antigen (BCMA) therapy. Modakafusp alfa development was discontinued for strategic reasons by the sponsor and the study was terminated early. At median follow-up of 7.3 and 7.6 months in the 120- and 240-mg arms, ORRs were 32% and 41%, and median progression-free survival was 4.1 and 5.3 months, respectively. ORRs were higher in patients who had not received previous BCMA therapy (46% vs 29%). The most common treatment-related adverse events (TEAEs) in the 120- and 240-mg arms were thrombocytopenia (75% and 84%; grade ≥3, 55% and 61%; respectively) and neutropenia (68% and 73%; grade ≥3, 56% and 68%; respectively); 90% and 96% of patients, respectively, experienced grade ≥3 TEAEs; 39% and 44%, respectively, experienced serious TEAEs. Our results confirm the efficacy of single-agent modakafusp alfa for patients with RRMM. This trial was registered at www.clinicaltrials.gov as #NCT03215030.
Introduction
Multiple myeloma (MM) is considered an incurable plasma cell malignancy. Although there have been significant advances in the treatment of MM, the demand for new, effective, and well-tolerated targeted therapies remains, especially for patients who have received and have disease refractory to all or most effective classes of antimyeloma agents, including immunomodulatory drugs (IMiDs), proteasome inhibitors (PIs), anti-CD38 monoclonal antibodies (mAbs), and B-cell maturation antigen (BCMA)–directed therapies.1-4
Modakafusp alfa is a novel immunocytokine that comprises 2 attenuated interferon alfa 2b moieties fused to a humanized anti-CD38 immunoglobulin G4 (IgG4) mAb and binds with high affinity to a unique epitope on CD38.5 The proposed mode of action of modakafusp alfa involves targeted interferon alfa receptor signaling within CD38-expressing cells, eliciting direct antiproliferative and apoptotic signals to tumor cells and activation of immune cells.5-8
In our previously reported phase 1/2 study in patients with relapsed/refractory MM (RRMM), modakafusp alfa demonstrated single-agent activity, with responses in 3 of 7 patients (43%) treated at the maximum tolerated dose of 3 mg/kg every 4 weeks and in 13 of 30 patients (43%) treated at 1 dose level below the maximum tolerated dose, 1.5 mg/kg every 4 weeks.5 The most notable toxicities were hematologic.5 Responses were observed among patients who had triple-class refractory disease (38%) or were penta-exposed (41%). The response rate for patients refractory to an anti-BCMA agent was 28%. Pharmacodynamic analyses revealed that modakafusp alfa binds to CD38 and upregulates the type 1 interferon gene signature score, resulting in increased CD38 receptor density in CD38+ cells (myeloma, natural killer, and T cells), and innate and adaptive immune cell activation.9
This phase 2 randomized study aimed to identify the optimal dose of modakafusp alfa by prospectively evaluating 120 and 240 mg (fixed doses equivalent to 1.5 and 3.0 mg/kg, respectively) administered every 4 weeks in patients with RRMM.
Methods
Trial design and patients
This was a global, multicenter, open-label, randomized, phase 1/2 study of modakafusp alfa for patients with RRMM (ClinicalTrials.gov identifier: NCT03215030). Parts 1 (dose escalation) and 2 (dose expansion) of this study have been reported previously.5 In the part 3 extension phase, patients were randomized 1:1 to receive modakafusp alfa 120 or 240 mg every 4 weeks.
Eligible patients were aged ≥18 years, had MM defined by International Myeloma Working Group criteria9 with evidence of disease progression, and required additional myeloma therapy as determined by the investigator. Additionally, patients were required to have received ≥3 previous lines of therapy (LoTs) and have disease that was refractory to ≥1 IMiD, ≥1 PI, and ≥1 anti-CD38 mAb. A full listing of inclusion and exclusion criteria is available in the supplemental Appendix, available on the Blood website.
Modakafusp alfa doses of 1.5 and 3.0 mg/kg were translated into 2 fixed doses of 120 and 240 mg based on the median patient body weight (∼80 kg) from parts 1 and 2 of the study. Patients received 120 mg or 240 mg modakafusp alfa via IV infusion over 1 hour (±10 minutes) every 4 weeks, and were treated until unacceptable toxicity, decision by the investigator/sponsor, patient withdrawal, or disease progression. Dexamethasone 20 mg or equivalent was used as premedication with each dose of modakafusp alfa; full details on allowed premedications and concomitant medications are listed in the supplemental Appendix.
Randomization was stratified by cytogenetic risk (high risk [del17, t(4;14), and/or t(14;16)] vs standard risk) and myeloma type (IgA vs other); the latter based on historical observations of greater efficacy of interferon therapy in patients with IgA myeloma.10,11 Cytogenetic results from samples obtained within 5 weeks before the first treatment dose were considered acceptable for stratification. If a previous result was available and the patient had high-risk disease from previous testing, they were stratified as high-risk.
End points and assessments
The primary end point was initially specified as the overall response rate (ORR; partial response or better) as assessed by an independent review committee (IRC) when the last patient had been followed up for 6 months. Dose selection was to be based on the totality of the data, that is, safety and tolerability, efficacy, and clinical pharmacokinetic and pharmacodynamic data.12 Because the further development of modakafusp alfa was discontinued by the sponsor for strategic reasons, the study was terminated early and the IRC was not used; the efficacy results presented in this report are therefore based on investigator-assessed response (originally a secondary end point per protocol). Because of the early study termination, the follow-up period was shortened to the last patient being followed up for ∼4 months, and the analysis reported here is the only analysis planned for this study. Other secondary end points included duration of response, clinical benefit rate, disease control rate, progression-free survival (PFS), time to progression, overall survival, rate of minimal (measurable) residual disease (MRD), safety, and patient-reported disease symptoms (DSs). Response was evaluated according to International Myeloma Working Group uniform response criteria.9 Assessments were conducted at every cycle during treatment and every 4 weeks during the PFS/survival follow-up period until disease progression. Bone marrow aspirate collections were required at screening, at suspected complete response (CR) based on laboratory values, and every 6 months thereafter for further evaluation of MRD. MRD was centrally evaluated for patients with a suspected CR using the clonoSEQ assay (version 2.0; Adaptive Biotechnologies) at a sensitivity of 10−5.
Pharmacokinetic sample collection and analysis
Serum samples for sparse pharmacokinetic measurements were collected at the following time points, after dosing: day 1 (before dose, end of infusion [EOI], and at 2-4 hours after dose); days 2, 8, and 15 during the clinic visit in cycles 1 to 2; day 1 (before dose, EOI, and at 2-4 hours after dose) of cycle 3 and beyond; day 2 during the clinic visit in cycles 5 and 11; and the end of treatment.
Free serum concentrations of modakafusp alfa with both CD38- and interferon alfa–binding sites intact were determined using a validated enzyme-linked immunosorbent assay. Based on the accuracy and precision results of the assay validation, the lower and upper limits of quantification were 6.25 and 200.0 ng/mL, respectively.
Patient-reported outcomes (PROs)
Patient-reported symptoms were evaluated using the EORTC QLQ-MY20 (European Organisation for Research and Treatment of Cancer quality-of-life questionnaire MM module) instrument, completed on days 1 and 8 of cycle 1, on day 8 on all cycles thereafter, and at the end of treatment. The instrument contains 20 items across 4 subscales: 2 functional and 2 symptom subscales, DSs and side effects of treatment (SEs), with the items referring to a 7-day recall period. We describe the 2 symptom subscales, DS and SE, both linearly transformed to a 0 to 100 scale, with higher scores indicating more severe problems/symptoms.13
Safety
Treatment-emergent adverse events (TEAEs) were graded per the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0.
Statistical analysis
The analyses for all efficacy end points were based on the full analysis set, which comprised all patients who received ≥1 dose of the study drug, including incomplete doses. This was a noncomparative study, with each arm treated as an independent single-arm study, so no formal statistical analysis was conducted to compare the treatment arms. The assumption for each arm for ORR was a null hypothesis of 20% and an alternate hypothesis of 35% with 1-sided type 1 error of 0.025% and 90% power. The original planned sample size was 236 patients (118 per treatment arm); after the sponsor decision to terminate the study early, enrollment was stopped at ∼75 patients per arm. Statistical assumptions were maintained, and adjustments were made for the reduced sample size at the time of data analysis. Patients with unknown or missing response status were treated as nonresponders. Safety and tolerability of modakafusp alfa was summarized using the safety analysis set, defined as all patients who received ≥1 dose of treatment, and assessed by recording TEAEs, vital signs, physical examination, laboratory analyses, electrocardiogram, and concomitant medications.
For binomial efficacy end points, frequencies, percentages, and the exact 2-sided 95% confidence intervals (CIs) were summarized. For time-to-event analysis, Kaplan-Meier survival curves and associated 2-sided 95% CIs based on the Brookmeyer and Crowley method are presented.
Descriptive PRO analysis, conducted in patients with a baseline (cycle 1, day 1) and ≥1 postbaseline measurement, was summarized with means and mean changes from baseline (with 95% CIs) at each cycle for each subscale score by dose arm without multiplicity adjustments. PRO compliance was calculated as (number of completed questionnaires)/(number of expected completed questionnaires). Differences in scores were interpreted per minimally important difference thresholds: “improved,” a score difference of −10 or less; “stable” (“maintained”), a score difference between −9 and 9; and “worsening” with a score difference of ≥10.
Ethical considerations
This study was conducted in accordance with the principles of the Declaration of Helsinki, the International Conference on Harmonised Tripartite Guideline for good clinical practice, and all applicable laws and regulations. Institutional review boards and independent ethics committees were constituted according to the applicable federal/local requirements of each participating region. All patients provided written informed consent.
Results
Patients
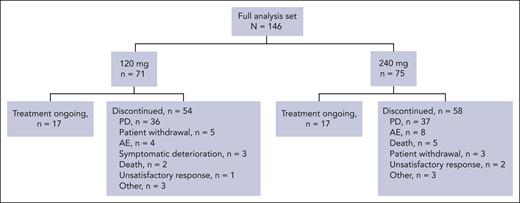
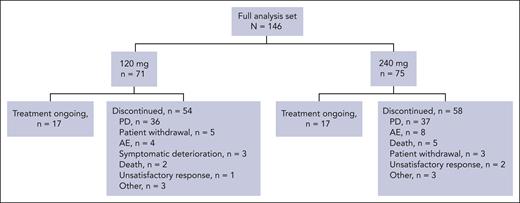
From 2 June 2022 to 27 September 2023, 147 patients were enrolled across 57 sites in 14 countries, with 146 patients treated and included in the full analysis set (120 mg arm, n = 71; 240 mg arm, n = 75; Figure 1). At data cutoff of 9 February 2024, 17 patients in each arm were still receiving ongoing treatment, with 112 patients having discontinued treatment, primarily due to progressive disease (120 mg arm, n = 36; 240 mg arm, n = 37). Median duration of exposure was 3.7 months and 3.3 months in the 120 mg and 240 mg arms, respectively. Median follow-up was 7.3 months (range, 1.1-17.3) in the 120 mg arm and 7.6 months (range, 0.4-19.8) in the 240 mg arm. Patient demographics and key baseline characteristics are included in Table 1. Overall, the median age of patients was 67 years, and median time from initial diagnosis was 7.2 years (range, 2.1-20.2) in the 120 mg arm and 6.8 years (range, 1.1-19.4) in the 240 mg arm. Median body weights were 76.9 kg and 75.3 kg in the 120 and 240 mg arms, respectively. In the 120 and 240 mg arms, 22 and 17 patients had high-risk cytogenetics features, respectively. Patients had received a median of 6 previous LoTs in each treatment arm. Previous anti-BCMA therapy had been received by 34 and 32 patients in the 120 mg and 240 mg arms, respectively. In the 120 mg arm, all patients had disease refractory to an anti-CD38 mAb and a PI, 25 patients (35%) had disease refractory to an anti-BMCA therapy, and 70 patients (99%) had disease refractory to an IMiD (1 patient was enrolled without meeting entry criteria of having IMiD refractory disease). In the 240 mg arm, all 75 patients had disease refractory to an anti-CD38 mAb, PI, and IMiD; and 29 patients (39%) had disease refractory to an anti-BCMA therapy. Across both arms, one-third of patients had penta-refractory disease, that is, refractory to 2 PIs, 2 IMiDs, and an anti-CD38 mAb; and 54 patients (37%) had disease refractory to an anti-BCMA therapy.
Efficacy
Investigator-assessed ORR was 32% (95% CI, 21.8-44.6) in the 120 mg arm, and 41% (95% CI, 30.1-53.3) in the 240 mg arm (Table 2). Overall, 14 patients in each arm had at least a very good partial response, and 27 and 36 patients had at least a minimal response in the 120 and 240 mg arms, respectively. Among 7 patients tested for MRD (120 mg, n = 6; 240 mg, n = 1), 4 patients were MRD negative at 10−5 (120 mg, n = 3; 240 mg n = 1). Of 3 MRD-negative patients in the 120 mg arm, 2 patients had a very good partial response and 1 had a stringent CR. The MRD-negative patient in the 240 mg arm had a CR.
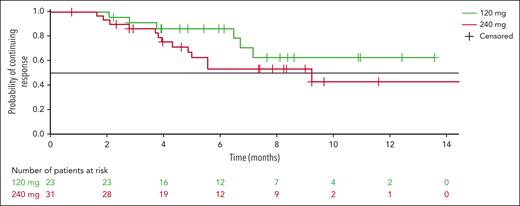
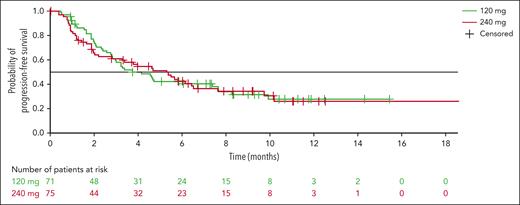
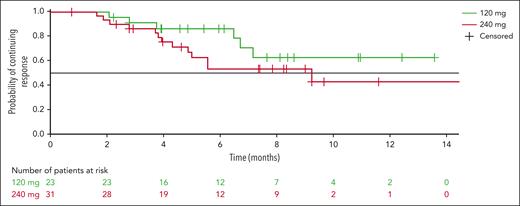
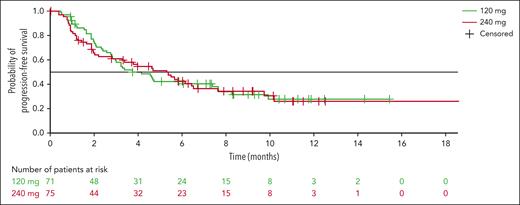
The median duration of response was not reached (95% CI, 6.7 to not evaluable) in the 120 mg arm, and was 9.2 months (95% CI, 4.9 to not evaluable) in the 240 mg arm (Figure 2). Median investigator-assessed PFS was 4.1 months (95% CI, 2.8-7.6) in the 120 mg arm and 5.3 months (95% CI, 2.8-6.5) in the 240 mg arm (Figure 3), and median time to progression was 4.7 (95% CI, 3.0-8.3) and 5.5 months (95% CI 3.4-7.6), respectively (supplemental Figure 1).
Kaplan-Meier curves of duration of response of modakafusp alfa 120 mg or 240 mg every 4 weeks (q4w).
Kaplan-Meier curves of duration of response of modakafusp alfa 120 mg or 240 mg every 4 weeks (q4w).
Kaplan-Meier curves of PFS with modakafusp alfa 120 mg or 240 mg q4w.
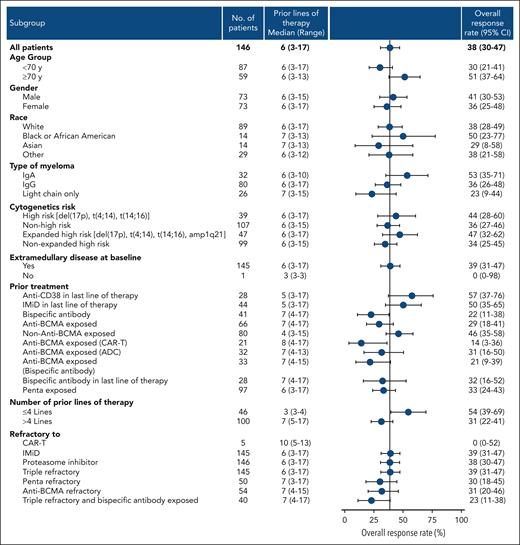
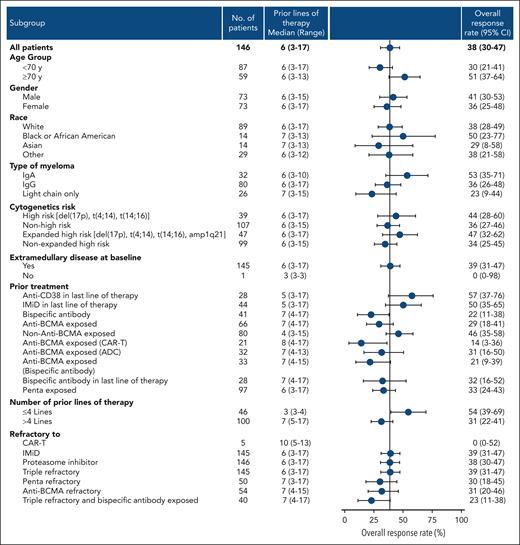
Figure 4 shows exploratory subgroup analyses of ORR for the combined study population, whereas analyses by treatment arm are shown in supplemental Figure 2. The ORR was numerically higher in patients who had received ≤4 previous LoTs than those who had >4 LoTs (54% vs 31%), and in patients who had not received previous anti-BCMA therapy compared with patients who had received previous anti-BCMA (46% vs 29%). Patients with and without previous exposure to anti-BCMA therapy had received a median of 7 and 4 previous LoTs, respectively. The ORR was 57% for patients who had received an anti-CD38 mAb in their last LoT; these patients had received a median of 5 previous LoTs (Figure 4).
Exploratory subgroup analysis full analysis set. ADC, antibody-drug conjugate; CAR-T, chimeric antigen receptor T cells.
Exploratory subgroup analysis full analysis set. ADC, antibody-drug conjugate; CAR-T, chimeric antigen receptor T cells.
Safety
In the 120 mg and 240 mg treatment arms, the most common TEAEs (occurring in >10% of the overall population) included thrombocytopenia (74.6% and 84.0%), neutropenia (67.6% and 73.3%), anemia (43.7% and 44.0%), and fatigue (29.6% and 37.3%; Table 3). The most common nonhematologic TEAEs with ≥10% difference between the 120 mg and 240 mg treatment arms were cough (16.9% vs 28.0%), nausea (12.7% vs 28.0%), dyspnea (9.9% vs 21.3%), and pyrexia (8.5% vs 18.7%). Infections and infestations were reported in 34 patients (47.9%) in the 120 mg arm, and 39 (52.0%) in the 240 mg arm. Hematologic TEAEs occurred more frequently in the 240 mg arm and during the first cycles of treatment (Table 4). Rates of platelet and red blood cell transfusions, and other hematologic supportive measures are shown in supplemental Table 1. Platelets and red blood cell support were more common in the 120 mg than the 240 mg arm (80.3% vs 65.3%), whereas granulocyte colony-stimulating factor administration was more frequent in the 240 mg arm than the 120 mg arm (50.7% vs 39.4%).
Grade ≥3 TEAEs were reported in 64 patients (90.1%) and 72 patients (96.0%) in the 120 mg and 240 mg arms, respectively. The most common grade ≥3 TEAEs were hematologic and occurred more frequently with 240 mg compared with 120 mg modakafusp alfa (neutropenia, 68.0% vs 56.3%; and thrombocytopenia, 61.3% vs 54.9%; Table 4). Grade ≥3 infections and infestations (21.1% vs 14.7%), renal and urinary disorders (8.5% vs 2.7%), and vascular disorders (5.6% vs 2.7%) were reported more frequently in the 120 mg arm vs the 240 mg arm.
Rates of serious TEAEs were 39.4% in the 120 mg arm and 44.0% in the 240 mg arm. Serious bleeding occurred in 4 patients (1 in the 120 mg arm, 3 in the 240 mg arm). Four patients (5.6%) in the 120 mg arm and 5 patients (6.7%) in the 240 mg arm died on study (associated TEAEs: 120 mg: infection, pulmonary hemorrhage, alteration of general condition, sepsis, and myeloma progression; 240 mg: respiratory failure, sepsis, worsening anemia, cardiac arrest, and sudden death).
In total, 30 patients (20.5%) experienced infusion-related reactions (IRRs); the most common symptoms across both treatment groups were chills (6.2%); flushing (4.8%); and back pain, nausea, and pruritus (each 4.1%; Table 5). A greater proportion of patients reported IRRs in the 120 mg arm (18 patients [25.4%]) than the 240 mg arm (12 patients [16.0%]). Most (87%) IRRs occurred during, or <2 hours after, infusion in both the treatment arms and the number of patients experiencing IRRs decreased with each treatment cycle. Four patients had grade 3 IRR events (2 in each arm), and 7 had serious IRRs (4 in the 120 mg arm; 3 in the 240 mg arm).
In total, 85 patients (58.2%) had a modakafusp alfa dose modification during the study, mostly because of TEAEs (in 78 patients [53.4%]). Of these, drug delays and reductions were slightly more common in the 240 mg arm, whereas infusion interruptions were more frequent in the 120 mg arm (Table 6). Overall, dose delays occurred in 45.1% of patients in the 120 mg arm and 50.7% of patients in the 240 mg arm; dose delays by treatment cycle and modakafusp alfa relative dose intensity are presented in supplemental Table 2, and TEAEs resulting in dose delay are shown in supplemental Table 3. Rates of study drug discontinuations because of AEs were 9.9% in the 120 mg arm and 13.3% in the 240 mg arm; TEAEs leading to drug discontinuation are shown in supplemental Table 4.
Pharmacokinetics
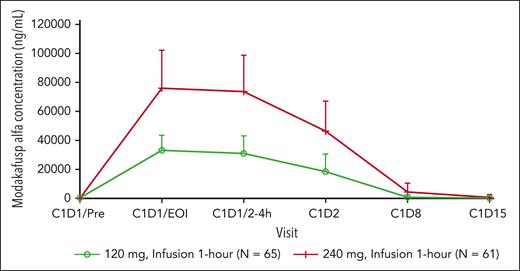
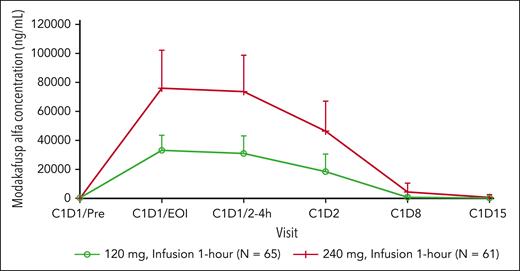
After single-dose administration, the geometric mean serum concentrations of modakafusp alfa at the EOI (ie, the expected maximum exposure level) were 31 μg/mL (n = 62) and 72 μg/mL (n = 60), in the 120 and 240 mg arms, respectively (Figure 5). The serum concentration declined in a biphasic manner after the EOI. At day 15 after dosing, serum concentrations of modakafusp alfa declined to below measurable levels in 89% of patients in the 120 mg arm and 51% of patients in the 240 mg arm. In general, the increase in overall exposure was approximately or slightly greater than twofold as the dose increased from 120 to 240 mg.
Mean serum concentrations of modakafusp alfa vs time after single-dose administration. C1, cycle 1.
Mean serum concentrations of modakafusp alfa vs time after single-dose administration. C1, cycle 1.
PROs
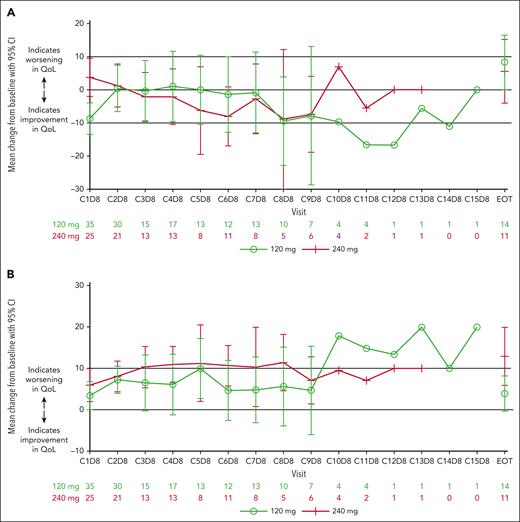
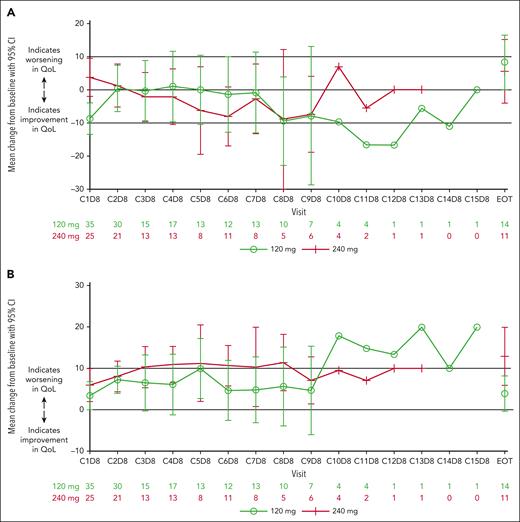
A total of 36 (120 mg arm) and 30 patients (240 mg arm) had PRO measurements at both baseline and a postbaseline visit. EORTC QLQ-MY20 compliance during treatment was 45% and 34% in the 120 and 240 mg arms, respectively. At baseline, mean DS scores were 26.4 and 22.8, and mean SE scores were 16.7 and 17.7 in the 120 and 240 mg arms, respectively. In the 120 mg arm, mean DS scores remained relatively unchanged compared with baseline over time with improvements at cycles 8 and 9; in the 240 mg arm, improvements in scores compared with baseline appeared more consistently from cycle 3 through to cycle 9 (Figure 6A). Scores for patients in the 240 mg arm trended below those for the 120 mg arm at most cycles, suggesting slightly poorer DSs in patients in the 120 mg arm (although 95% CIs crossed). Mean SE scores worsened at all cycles compared with baseline in both arms, with scores appearing slightly poorer for patients in the 240 mg arm (minimally important difference reached at most cycles) vs the 120 mg arm (although 95% CIs crossed; Figure 6B). For the DS subscale, generally a higher proportion of patients in the 240 mg arm vs 120 mg arm were classified as having “improved or stable” disease from baseline at each cycle; whereas, for the SE subscale, typically a higher proportion of patients in the 240 mg arm vs the 120 mg arm were classified as having “worsening” disease from baseline at each cycle (except for cycles 2, 5, and 9; supplemental Figure 3). Beyond cycle 9, only 7 and 6 patients in the 120 and 240 mg arms, respectively, remained in the analysis, and therefore no interpretations were made for these data points.
Patient-reported outcomes. Mean change from baseline and 95% CIs of EORTC QLQ-MY20 scores in DS subscale (A) and side effects of treatment subscale (B) over time with modakafusp alfa 120 mg or 240 mg q4w. CIs were truncated if outside of (−100, 100) and suppressed if there were >5 patients in either treatment arm. Lines at y = 10 and y = −10 were plotted to aid interpretation of subscale trends based on minimally important difference thresholds.14 EOT, end of treatment; QoL, quality of life.
Patient-reported outcomes. Mean change from baseline and 95% CIs of EORTC QLQ-MY20 scores in DS subscale (A) and side effects of treatment subscale (B) over time with modakafusp alfa 120 mg or 240 mg q4w. CIs were truncated if outside of (−100, 100) and suppressed if there were >5 patients in either treatment arm. Lines at y = 10 and y = −10 were plotted to aid interpretation of subscale trends based on minimally important difference thresholds.14 EOT, end of treatment; QoL, quality of life.
Discussion
This randomized phase 2 extension study was intended to define the single-agent dose of modakafusp alfa for further clinical development, including as a component of combination therapy, given the encouraging findings of the phase 1/2 study in patients with RRMM.5 The investigator-assessed ORRs of 32% in the 120 mg arm and 41% in the 240 mg arm are encouraging, given enrolled patients had triple-class-refractory disease and had received a median of 6 LoTs (range, 3-17). ORRs tended to be lower in subgroups of patients with a higher number of previous LoTs. Response rates for patients previously exposed to anti-BCMA therapy and those not previously exposed were 26% (95% CI, 13%-44%) and 41% (95% CI, 25%-58%), respectively, in the 120 mg arm and 31% (95% CI, 16%-50%) and 51% (95% CI, 35%-67%), respectively, in the 240 mg arm; the anti-BCMA–exposed subgroup are a population in whom there are limited options for therapy.15 Conversely, patients who had received an anti-CD38 in their last LoT had an ORR of 57% (95% CI, 37%-76%). This could be because of a more favorable biological state of these patients, reflected by their lower number of previous LoTs but may also be because of a synergistic effect between modakafusp alfa and any circulating anti-CD38 mAbs, which have a long half-life.16,17 Furthermore, modakafusp alfa was shown to upregulate CD38 expression on myeloma cells in patients in the first-in-human (FIH) phase 1/2 study5; and so it is possible that CD38 upregulation combined with remnant anti-CD38 mAbs may be resulting in a higher efficacy in these patients, driven by early responses. This is further supported by results from a separate phase 1 study of modakafusp alfa in combination with daratumumab showing an encouraging ORR of 53% in 15 patients with ≥3 previous LoTs and/or triple-refractory myeloma (K. Kim, S. Manier, M. A. Schroeder, R. Zuniga, W. DeRosa, J. M. Schiano de Colella, M. Schuster, T. Chen, C. Li, S. C. Collins, C. Argueta, A. Nelson, S. Wang, X. Parot, S. Wu, S. A. Holstein, unpublished data, May 2025).
Although caution should be exercised when performing cross-trial comparisons, our results appear favorable relative to phase 2 trials of therapies in similar patient populations (despite the inclusion of heavily pretreated patients including those who had received previous BCMA-targeted therapies), including daratumumab (ORR, 29%),18 belantamab mafodotin (ORR, 31%-34%),19 selinexor in combination with dexamethasone (ORR, 20%-26%),20,21 and mezigdomide (ORR, 41%).22
Consistent with the results from our phase 1/2 study,5 most TEAEs observed were hematologic. Such events were more common in the 240 mg arm, most notably grade 3 and 4 neutropenia, which was reflected in the greater use of colony-stimulating factor in the 240 mg arm. It is possible that these toxicities are related to interferon receptor engagement by modakafusp alfa on CD38-expressing hematopoietic cells. Preclinical studies have shown that modakafusp alfa causes maturational arrest of megakaryocyte precursors (unpublished). Nevertheless, hematologic AEs were generally manageable with a combination of supportive therapies and dose delays with few patients discontinuing study treatment because of hematologic toxicities. Rates of grade ≥3 infections were numerically lower in the 240 mg arm (15% vs 21% in the 120 mg arm). There were no reports of cytokine release syndrome or immune effector cell–associated neurotoxicity syndrome symptoms typically seen with T-cell–redirecting drugs,23-26 or neuropsychiatric side effects reported with systemic interferon alfa therapy.27,28 Of note, the rate of discontinuation because of AEs was low (12%).
After single-dose administration, the increase in overall serum exposure appeared to be approximately or slightly greater than twofold as the dose increased from 120 to 240 mg. Although differences between doses were not evaluated, this finding of greater exposure with 240 mg suggests clinical significance given the observed higher rates of both response and toxicities with 240 mg.
Because higher rates of efficacy and toxicity were observed with 240 mg modakafusp alfa, the dose choice for any potential future trials would likely be dependent on the patient population and whether modakafusp alfa is used as a single agent or as part of combination therapy (120 mg is the suggested starting dose for combination therapy) especially when combined with agents that have overlapping toxicities. Regarding possible combination partners for future trials, pharmacodynamic data from our FIH phase 1/2 study showed an increase in CD38 density on T cells and myeloma cells, and activation of CD8 T cells after treatment with modakafusp alfa.5 Based on this, the manageable safety profile with no reports of immune effector cell–associated neurotoxicity syndrome and cytokine release syndrome, and on data from the separate study of modakafusp alfa in combination with daratumumab (Kim et al, manuscript in preparation), we hypothesize that both T-cell–redirecting therapies and anti-CD38 mAbs may be appropriate for further investigation, with efficacy most likely to be seen in earlier LoTs. Although exercising caution would be necessary because of the potential for increased hematologic toxicities, other potential combination partners include IMiDs and PIs. Evaluation of PROs in this study revealed that patient-reported DSs somewhat improved from baseline across treatment cycles in the 240 vs 120 mg arms, with a generally higher proportion of patients in the 240 mg arm vs those in the 120 mg arm classified as having “improved or stable” disease at each cycle relative to baseline. Conversely, patient-reported SEs of treatment appeared slightly poorer during treatment relative to baseline for the 240 vs 120 mg arms, with a higher proportion of patients in the 240 mg arm vs those in the 120 mg arm classified as having disease “worsening” from baseline at most cycles. The PRO findings are consistent with, and supportive of, the efficacy and safety results observed for the 2 arms.
The main limitation of this study relates to its premature termination and the consequent impact on data collection, such as potentially missed late deepening of response, missed bone marrow evaluations for confirmation of CRs, limited collection of toxicity data, and limited long-term follow-up. Furthermore, investigator assessment was used for the primary end point of ORR, instead of the protocol-specified IRC assessment. Finally, the study was noncomparative, thus, an optimal dose could not be statistically ascertained.
Because of the early discontinuation of this study, there is no plan for formal selection of the optimal dose from these data, because no further studies are planned by the sponsor. However, because of the novel mechanism of action of the drug and the clinically meaningful results seen in heavily pretreated patients, almost half of whom have received the most recently available targeted therapies of different modalities (eg, BCMA-directed antibody-drug conjugates, bispecific antibodies, and chimeric antigen receptor T-cell therapies), these results are highly relevant for the myeloma community because they inform future drug development efforts centered on novel strategies involving targeted delivery of interferon alpha and potentially immunotherapy more broadly.
To conclude, our results describe the efficacy and safety of modakafusp alfa, a first-in-class immunocytokine, for patients with RRMM. Modakafusp alfa exhibited efficacy and manageable toxicity in heavily pretreated patients, including those who had received BCMA-directed therapy. Further development of modakafusp alfa has been discontinued by the sponsor for strategic reasons including the rapidly evolving MM treatment landscape and the long development timelines. Although this highlights the complexity of drug development for RRMM in the era of rapid advances in immunotherapies, the need for innovative treatment strategies persists given the incurable nature of the disease.
Acknowledgments
The authors thank the patients and their caregivers, as well as the physicians, nurses, trial coordinators, and research staff for participation in the trial. The authors acknowledge and thank Erin Goodman (employee of Takeda Development Center Americas, Inc) for her technical contribution to the manuscript. Medical writing support for the development of this manuscript, under the direction of the authors, was provided by Advaitaa Haripershad and Luisa Madeira of Ashfield MedComms, an Inizio Company, funded by Takeda Pharmaceuticals USA, Inc, Cambridge, MA, and complied with good publication practice guidelines.29
This trial was funded by Takeda Development Center Americas, Inc, Cambridge, MA.
Authorship
Contribution: H. Mian, C.B.G., K.S., C.L., D.C., X.P., and D.T.V. made substantial contributions to the conception or design of this study; S.A.H., S.A., M.A.D., F.S., R.P., M.E.G., C.B.G., C.T., M.B., H. Magen, O.N., K.S., C.L., S.L., A.N., D.C., X.P., and D.T.V. made substantial contributions to the acquisition, analysis, or interpretation of data for this study; and all authors were responsible for drafting the work or revising it critically for important intellectual content, providing their final approval of the version to be published, and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Conflict-of-interest disclosure: S.A.H. reports serving on an advisory council or committee for Takeda; and received consulting fees from Janssen, Sanofi, and Takeda. S.A. has served on an advisory council or committee for, and received honoraria from, Bristol Myers Squibb (BMS), Johnson & Johnson (J&J), and GlaxoSmithKline (GSK); received consulting fees from Sanofi and Pfizer; and received grants or funds from GSK and Amgen. H. Mian has been on an advisory council or committee for J&J, GSK, Sanofi, Pfizer, Takeda, and AbbVie; has received honoraria from Janssen; and received grants or funds from Janssen and Pfizer. M.A.D. reports honoraria from, and an advisory council or committee position for, Amgen, Sanofi, Regeneron, Menarini, Takeda, GSK, BMS, Janssen, BeiGene, Swixx, and AstraZeneca. F.S. has served on an advisory council or committee for AbbVie, Celgene, GSK, Janssen, Oncopeptides, Sanofi, and Takeda; and received honoraria from AbbVie, Amgen, BMS, Daiichi Sankyo, GSK, Janssen, Novartis, Oncopeptides, Pfizer, Sanofi, SkylineDx, Takeda, and Regeneron. R.P. has received honoraria from J&J, GSK, AbbVie, BMS, Pfizer, and Roche; has received consulting fees from GSK and J&J; and has received grants or funds from Pfizer and GSK. L.F. received consultancy fees from Sanofi, Janssen, Amgen, AbbVie, and Pfizer. M.B. reports serving on an advisory council or committee for BMS, Janssen, GSK, Takeda, Menarini, Sanofi, and Pfizer; has received honoraria from Amgen, BMS, Menarini, Janssen, Takeda, and GSK; and has received grants or funds from Janssen. S.M. is a consultant for AbbVie, Adaptive Biotechnology, Amgen, Celgene/BMS, GSK, Janssen, Novartis, Regeneron, Roche, Sanofi, and Takeda. O.N. has served on an advisory council or committee for J&J, BMS, Sanofi, Takeda, and GPCR Therapeutics; and has received grants or funds from JNJ, BMS, and Takeda. K.S., C.L., S.L., and D.C. report employment and the ownership of stock/shares with Takeda. X.P. is employed by Takeda. D.T.V. reports consulting fees from Takeda, AbbVie, Janssen, BMS, GSK, Genentech, and Karyopharm; and has received grants or funds from Takeda and Active Biotech. The remaining authors declare no competing financial interests.
The current affiliation for A.N. is PMV Pharmaceuticals.
Correspondence: Sarah A. Holstein, University of Nebraska Medical Center, 986840 Nebraska Medical Center, Omaha, NE 68198; email: sarah.holstein@unmc.edu.
References
Author notes
The data sets, including the redacted study protocols, redacted statistical analysis plans, and individual participant data supporting the results of the completed study will be made available after the publication of the final study results within 3 months from initial request to researchers who provide a methodologically sound proposal. The data will be provided after their deidentification, in compliance with applicable privacy laws, data protection, and requirements for consent and anonymization.
The online version of this article contains a data supplement.
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.