In this issue of Blood, Elitzur et al1 investigate the outcomes of hematologic malignancies in individuals with ataxia-telangiectasia (A-T) by conducting a retrospective analysis of an international cohort to determine risk factors associated with adverse outcomes and generate evidence-based treatment recommendations.
A-T is a multisystemic DNA repair disorder commonly manifesting in early childhood presenting with progressive neurologic deficits (ie, ataxic gait), oculocutaneous telangiectasias, humoral or cellular immunodeficiency, and pulmonary disease.2 Biallelic mutations in the ATM gene result in either absence or significant reduction of the ATM kinase activity, which is essential for repairing DNA double-stranded breaks caused by endogenous and exogenous insults.3 Qualitative and quantitative changes in ATM kinase due to pathogenic ATM mutations lead to a heterogeneous phenotypic spectrum. Classic A-T is used to describe individuals with A-T with severe symptoms due to absence of ATM protein, and variant A-T is associated with mild clinical features due to remnant functional ATM.2 As with other DNA repair disorders, individuals with A-T have excess cancer risk (10%-25% lifetime incidence), with hematologic malignancies being the most common.4-8
Survival rates for hematologic malignancies have greatly improved over time due to large-scale clinical trials.9 Current investigations in this field are focused on targeted therapeutic strategies to reduce toxic chemotherapeutic and radiation exposure with the intent of improving long-term morbidity and mortality while maintaining curative success.10 Unfortunately, individuals with A-T have not experienced this wave of success likely due to the rarity of the disease and heterogeneous cancer spectrum, which challenges attempts at a large prospective analysis. The inherent nature of cellular radiosensitivity in individuals with A-T commonly results in the development of excess organ toxicities and the need to deescalate or modify standard chemoradiotherapy treatment regimens. This frequently leaves the oncologist with the impossible task of balancing the likelihood of cure with risk of organ toxicities and even treatment-related mortality (TRM).
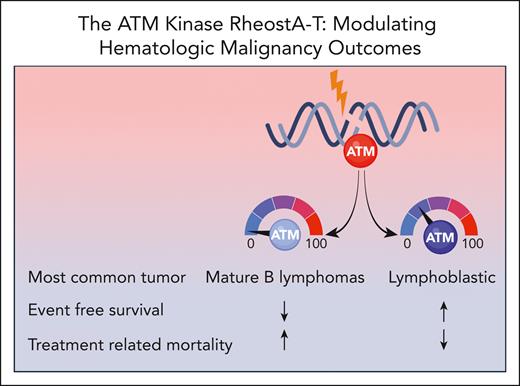
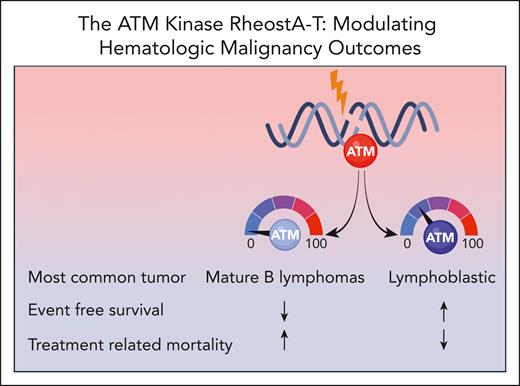
To address this gap, the authors describe the largest cohort of individuals with A-T and hematologic malignancies (n = 202) to date, involving patients from 25 different countries. The most common hematologic malignancy identified was mature B-cell lymphoma, validating findings from other A-T cancer cohorts,4-6,8 followed by acute lymphoblastic leukemia or lymphoma, Hodgkin lymphoma, and other rare blood malignancies. Interestingly, mature B-cell lymphomas were more common in patients with absent ATM kinase, and those with residual ATM kinase were mostly affected by lymphoblastic malignancies (see figure). Four-year event-free survival (EFS) and overall survival for the cohort was close to 50% with TRM being the major cause of treatment failure. TRM was defined as any death resulting from cancer therapy while being in remission or death within 3 months of therapy completion. The authors also stratified hematologic cancer outcomes based on ATM kinase activity (see figure). Among 110 patients, absent ATM kinase activity was associated with poorer EFS and higher TRM rates compared with those with residual kinase activity. Both advanced respiratory and neurologic disease correlated with poorer EFS, but only advanced respiratory disease demonstrated an increase in TRM. Older age at cancer diagnosis was associated with both decreased survival and higher TRM. Interestingly, the type of malignancy did not impact TRM but did impact EFS with the poorest outcomes observed in individuals with Hodgkin lymphoma.
The level of ATM kinase activity impacts cancer spectrum and outcomes in individuals with A-T.
The level of ATM kinase activity impacts cancer spectrum and outcomes in individuals with A-T.
The authors are to be commended for the substantial clinical impact that their findings will have for individuals with A-T and hematologic malignancies. They propose considering the functional classification of ATM mutations and severity of A-T-associated comorbidities to tailor cancer therapy, suggesting a path toward personalized cancer therapy. The practical question now is how to prospectively assess this relationship in A-T, which is a rare disorder associated with a variety of nonuniformly treated neoplasms. This underscores the need for international collaborations and novel clinical trial approaches to test ATM kinase activity-stratified treatments. Several other questions remain for treating individuals with A-T-associated malignancies. What is the solid organ cancer spectrum in A-T, and would the same approach be applicable to individuals with these cancers? What are the specific causes for TRM in A-T individuals and which organs are most chemo- and radiosensitive? Does attenuated therapy affect rates of cancer recurrence? What surveillance modality can be safely and practically utilized in this population without adding to their already substantially increased lifetime cancer risk? Systematically addressing these knowledge gaps through collaborative partnerships and prospective studies will be essential to generate meaningful impact on cancer therapy in individuals with A-T and other DNA repair disorders.
Conflict-of-interest disclosure: R.S. declares no competing financial interests.