In this issue of Blood, Cruz-Leal et al provide important new insights into the mechanism of red blood cell (RBC) antigen loss,1 previously proposed by this group and others to be implicated in antibody-mediated immune suppression (AMIS) of erythrocyte alloimmunization independent of red cell clearance or epitope masking.2,3 Cruz-Leal et al demonstrate that some AMIS-inducing antibodies targeting RBC antigens, including anti-RhD, can trigger antigen loss through a phenomenon of membrane-bound component transfer to macrophages called trogocytosis, without necessitating RBC clearance.
Alloimmunization against RBC antigens during pregnancy or transfusion can cause serious complications, including hemolytic disease of the fetus and newborn (HDFN) and hemolytic transfusion reactions. HDFN happens when maternal alloantibodies, most frequently anti-RhD, cross the placenta and destroy incompatible fetal RBCs, potentially leading to anemia, jaundice, and even neonatal death in severe cases.4 The only prophylactic treatment currently available to prevent anti-RhD formation in RH:–1 pregnant women is the administration of human polyclonal anti-RhD. This prophylaxis is the sole example of AMIS used clinically to prevent alloimmunization and HDFN. However, alloimmunization can also be due to other non-RhD alloantigens, among which alloantibodies against MNS (U), Rh (c, C, E), Kell (K, Jsa), Kidd (Jka), and Duffy (Fya) blood group antigens can lead to clinically significant HDFN cases and for which no prophylaxis exist.5 Findings from Cruz-Leal et al unveil a novel molecular mechanism of AMIS, paving the way for developing new prophylactic strategies to prevent HDFNs.
In the current study, Cruz-Leal et al used an HOD (hen egg lysozyme [HEL]-ovalbumin-human Duffy) mouse model treated with 11 distinct antibodies, with varying immunoglobulin G (IgG) subtypes and glycosylation states, to evaluate their role in RBC clearance and antigen loss associated with AMIS. They demonstrate that multiple antibodies were capable of triggering AMIS without necessarily mediating RBC clearance. Instead, AMIS was attributed to antigen depletion through trogocytosis, as evidenced by antigen reduction in the presence of macrophages without inducing phagocytosis. In experiments with C57BL/6 mice transfused with incompatible RBCs and treated with a variety of antibodies, the authors observed that deglycosylation of CBC-512 did not impact its ability to induce AMIS. However, it did hamper its efficacy in clearing HOD-RBCs. Both forms of this antibody led to the loss of the Duffy epitope and a notable decline in RBC membrane fluorescence. Further in vitro examinations showed that antibodies targeting specific sections of the HOD molecule led to antigen and membrane loss from RBCs in the presence of macrophages, a phenomenon they identified to be macrophage dependent. More excitingly, similar results were observed with anti-RhD in human RBCs. Confocal microscopy showed macrophages containing membrane segments from sensitized RBCs, indicating trogocytosis. However, erythrophagocytosis has also been observed by the authors either concurrently with or independent of trogocytosis, confirming that both processes can underly AMIS.
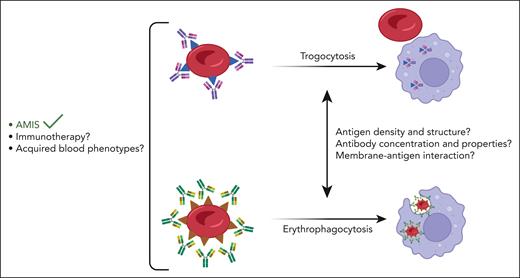
Why do macrophages preferentially induce trogocytosis vs erythrophagocytosis depending on the antigen-antibody interaction? The findings from Cruz-Leal et al using anti-RhD suggest that RBC phagocytosis may require either more antibody or a higher antigen density than trogocytosis. Although the process triggering trogocytosis is poorly studied, it is well known that phagocytosis requires high levels of antibody-opsonized targets or tight ligand clustering to enhance FcγR phosphorylation and trigger phagocytosis.6 The capability of an antibody to either clear RBCs or induce antigen modification can depend on multiple factors such as the density of the targeted antigen on the RBC, the molecular structure of the antigen, the binding affinity of the corresponding antibody, and the specific mechanisms driving each process (see figure).2 In the current work, the use of HEL polyclonal IgG was unable to induce significant phagocytosis of HOD-RBCs although it triggered phagocytosis of HEL RBCs. The phagocytosis behavior differs between HOD RBCs and HEL RBCs due to varying HEL antigen copy numbers. This is in line with previous work showing that the relationship between antigen copy number and antibody concentration influences the outcome of RBC alloimmunization in HEL RBC mice.7
Anti-erythrocyte antibodies, for AMIS and potentially in the context of immunotherapies or acquired blood phenotypes, bind their corresponding RBC antigens and either induce trogocytosis-mediated antigen loss or lead to phagocytic clearance of IgG-sensitized RBCs by macrophages. This balance may be controlled by several factors including antigen density and structure at the membrane, antibody concentration and biological properties, and the interaction of the antigen with membrane proteins. The figure was adapted from the visual abstract in the article by Cruz-Leal et al that begins on page 807.
Anti-erythrocyte antibodies, for AMIS and potentially in the context of immunotherapies or acquired blood phenotypes, bind their corresponding RBC antigens and either induce trogocytosis-mediated antigen loss or lead to phagocytic clearance of IgG-sensitized RBCs by macrophages. This balance may be controlled by several factors including antigen density and structure at the membrane, antibody concentration and biological properties, and the interaction of the antigen with membrane proteins. The figure was adapted from the visual abstract in the article by Cruz-Leal et al that begins on page 807.
Cruz-Leal et al propose trogocytosis as a potential mechanism for AMIS in anti-RhD prophylaxis; however, this could also be a plausible mechanism by which RBC antigen loss might occur with other human anti-erythrocyte antibodies. For instance, immunotherapy treatment using some monoclonal antibodies that recognize antigens also present on RBCs are known to induce loss of these antigens from RBC surface. The anti-CD38 monoclonal antibody daratumumab (DARA) offers a targeted therapeutic approach against plasma cells in multiple myeloma patients. Interestingly, many patients treated with DARA unexpectedly show a negative direct antiglobulin test, and the antibody screen in their plasma indicates the presence of anti-erythrocyte antibody.8 The agglutination potential of DARA in indirect antiglobulin test is a commonly seen phenomenon that complicates blood transfusion tests. Interestingly, RBCs from DARA-treated individuals exhibit reduced CD38 levels, even after weeks of treatment, without causing hemolysis or altering other RBC antigens. The mechanism by which DARA induces CD38 loss from the RBC membrane remains unknown.8 CD38 is weakly expressed on the RBC surface, and it would be of great interest to assess the implication of trogocytosis in DARA-induced loss of CD38 in patients treated with DARA. Another immunohematological enigma in which trogocytosis-based antigen loss may be involved is the acquired blood phenotypes. These result from the spontaneous loss of blood group antigens at the RBC surface and are frequently detected in patients with hematological disorders in the absence of previous transfusion or transplantation. In the acquired AnWj-negative and JMH:–1 blood phenotypes, the expression of both AnWj and JMH1 antigens is reduced or completely lost through unknown mechanisms,9,10 with the concomitant appearance of their corresponding autoantibodies. The involvement of trogocytosis in these acquired blood phenotypes is worth investigation.
Overall, Cruz-Leal et al convincingly demonstrate that trogocytosis plays a major role in mediating RBC antigen loss in AMIS independently from RBC phagocytic clearance. Future studies should assess the impact of trogocytosis on RBC integrity and life span. Additionally, further investigations to assess the balance between trogocytosis and erythrophagocytosis in mediating antibody-induced RBC antigen loss should consider the tissue where these phenomena might occur (ie, the spleen), the inflammatory state or other clinical background of patients, and the different types of macrophages involved.
Conflict-of-interest disclosure: The authors declare no competing financial interests.